| Research Article | ||
Open Vet. J.. 2026; 16(3): 1488-1495 Open Veterinary Journal, (2026), Vol. 16(3): 1488-1495 Research Article ELISA-based assessment of cholinesterase levels in different animal groups: Insights into neurological healthReem M. Mousa1, Azhar Adil Mohammad1* and Ayman Albanna21Basic Nursing Sciences Branch, Nursing College, Ninevah University, Mosul, Iraq 2Department of Environmental Science, College of Environmental Sciences, University of Mosul, Mosul, Iraq *Corresponding Author: Azhar Adil Mohammad. Basic Nursing Sciences Branch, Nursing College, Ninevah University, Mosul, Iraq. Email: azhar.mohammed [at] uoninevah.edu.iq Submitted: 10/11/2025 Revised: 11/02/2026 Accepted: 21/02/2026 Published: 31/03/2026 © 2026 Open Veterinary Journal
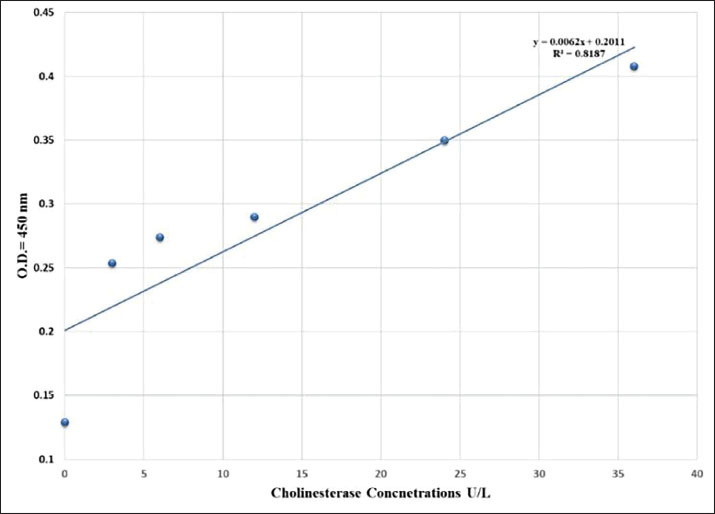
AbstractBackground: Neurological health is a relevant area of animal public health, as perturbations in the activity of neural enzymes have a tendency to mirror animal welfare and environmental stress that will ultimately affect human populations. Aim: The aim was to measure serum cholinesterase (ChE) activity in different animal species using a 96-well Enzyme-Linked Immunosorbent Assay (ELISA) assay, with an emphasis on its application as a biomarker of animal welfare within the frame of public health. Methods: A linear calibration curve (y=0.0062x + 0.2011, R²=0.8187) with nice linearity was achieved, and validated the assay to be trustworthy for quantitative enzyme assay. Nine groups of animal serum samples, including rabbit, sheep, cow, fish, cat, chicken, rat, dog, and goat, were collected and assayed to determine variation in ChE levels. Results: The findings demonstrated distinct interspecies variations, revealing the highest mean enzyme activity in chickens (6.99 U/l), followed by cats and fish, and the lowest in sheep, goats, and dogs (1.6–1.7 U/l). The results show that the activity of ChE is directly related to the physiological status of animals and may be a highly sensitive biochemical parameter for measuring animal welfare and neurophysiological homeostasis. There could be reduced activity of ChE could be indicative of exposure to neurotoxic chemicals, stress, or starvation, while consistent activity would indicate health and environmental stability. Conclusion: From the public health point of view, the study highlights the importance of monitoring ChE activity as being integral to welfare monitoring and environmental watch programs. The ELISA-based assay was shown to be a high-throughput, robust method for determining neurological and welfare status in animals, reaffirming its usefulness under the One Health umbrella that links animal and human well-being. Keywords: Animal public health, Cholinesterase levels, ELISA. IntroductionNeurological health of domestic and wildlife animals is essential in ecological balance, productivity, and welfare (Beaulieu, 2024). Among all biomarkers used in ascertaining neurological health, cholinesterase (ChE) enzymes, and particularly acetylcholinesterase (AChE) and butyrylcholinesterase (BChE), have generated enormous interest as sensitive markers of neurotoxic chemicals and as playing a fundamental role in synaptic transmission (Santarpia et al., 2013). ChEs catalyze the hydrolysis of the neurotransmitter acetylcholine to choline and acetate, a reaction that is essential to the termination of nerve impulses in cholinergic synapses (Tougu, 2001). Any alteration in their activity, whether through genetic, physiological, or toxicological mechanisms, can lead to profound disruptions of neuromuscular function and mental capacity (Siciliano et al., 2020). Consequently, measurement of ChE levels provides an extremely sensitive indicator of animal population neurological health and environmental exposure history (Lionetto et al., 2013). In the past decades, exposure to neurotoxic chemicals, including organophosphates, carbamates, heavy metals, and certain natural toxins, has been identified as a main cause of ChE inhibition (Colovic et al., 2013). Ecological monitoring studies increasingly employed ChE activity as a neurotoxicity biomarker for estimating ecosystem health, occupational hazard, and pesticide contamination (Chambers et al., 2002). Despite its long-standing diagnostic application, however, comparative measurements of ChE activity across different animal phyla are few (Jiang et al., 2022). Interspecies comparison can illustrate evolutionary adaptations in neurophysiology and reveal species that are atypically vulnerable to neurotoxic stressors (Spurgeon et al., 2020). A very valuable and potent instrument for immunodiagnosis, with which to identify biomolecules of high specificity and sensitivity, is the Enzyme-Linked Immunosorbent Assay (ELISA) (Kondo et al., 1995). ELISA is thus highly suited for large-scale comparative studies across animal species. The knowledge of baseline and variant profiles of ChE activity in animal species serves multiple purposes. It enables us to establish reference ranges that are needed for the interpretation of deviations occurring due to disease, aging, or exposure to xenobiotics (Nunes, 2011). Second, comparisons across species can uncover differences in functional and ecological responses—e.g., herbivorous versus carnivorous versus omnivorous species, or aquatic versus terrestrial species—that are a manifestation of their respective evolved strategies of neural modulation and detoxification (Pezzementi et al., 2011). Third, these findings are of translational relevance to human health because, in most cases, animals serve as sentinels for environmental neurotoxicity, mirroring exposures that may, in the long run, affect human populations within the same environment. AChE in mammals is located mainly in neural and muscle tissue, while BChE occurs in tremendous quantity in plasma and liver as a reserve scavenger enzyme (Ord and Thompson, 1950). In birds, both the localization and activity of these enzymes are highly variable with respect to migratory pattern and dietary exposure to agrochemicals (Layer, 1983). Amphibians and reptiles, which primarily occupy contaminated aquatic environments, possess high ChE sensitivity and are valuable bioindicators of pesticide pollution. Fish species, too, have been extensively studied for water pollutant ChE response because enzyme activity changes mirrored behavioral and reproductive impairment (Khan and Law, 2005). However, complexity in interspecies variation demands standardized procedures such as ELISA for credible cross-taxonomic comparison to be possible. Outside of toxicological measurement, the ChE enzymes play roles in larger processes of neurological health and disease (Ballard et al., 2005). Changed levels of AChE and BChE in humans and in animal models are linked to neurodegenerative diseases such as Alzheimer's, Parkinson's, and hepatic encephalopathy Changed levels of AChE and BChE in humans and in animal (Bawaskar et al., 2015). Study of similar enzymatic adaptations in animals provides a model comparison for the understanding of neurochemical control, oxidative stress adaptations, and adaptive response in different physiological conditions (Akki et al., 2022). As global attention to one health—integrated human, animal, and environmental health—grows, biomarker surveillance of neurological integrity along the biological continuum assumes greater importance. Overall, these studies provide input to predictive models of neurotoxic risk, refine risk assessment practices, and guide conservation efforts for susceptible wildlife species. This article, "ELISA-Based Measurement of ChE Activities in Various Animal Groups: Implications for Neurological Welfare," aims to provide a general comparative evaluation of ChE activities across various animal taxon using the ELISA assay. The idea is aimed at interspecies different identification, and the foundation of baseline reference values, and examining relationships between enzyme activity and ecological or physiological factors. Through the application of biochemical data coupled with neurophysiological interpretation, the study tries to account for how ChE modulation reflects adaptive and pathological processes within animal nervous systems. Material and MethodsThe idea was focused on evaluating ChE enzyme activity in diverse groups of animals using a quantitative sandwich ELISA provided by Sunlong Biotech Co., which reference number (SL004Ani) (Kondo et al., 1995). Ninety blood samples were collected from young male animals representing nine species commonly found in Iraq. Animals were selected based on defined age-specific criteria to ensure uniformity among young individuals. Breed or species subtype, nutritional status, housing conditions, and geographic and environmental exposure were documented, and all sampling procedures were performed in compliance with recognized animal welfare and ethical standards, i.e., rabbit (Oryctolagus cuniculus), sheep (Ovis aries), cow (Bos taurus), fish (Cyprinus carpio), cat (Felis catus), chicken (Gallus gallus domesticus), rat (Rattus norvegicu), dog (Canis lupus familiaris), and goat (Capra hircus) according to consideration blood collection techniques. Rabbits were considered at 6 months of age, and sheep were chosen at 5 months. Cattle considered in this study included cows aged 8 months. Samples of fish were obtained at 3 months, depending on the growth rate of each species. Cats were considered at 5 months and chickens at 4 months. Lab rats were chosen at 5 weeks with consideration of their early sexual maturity. Dogs were considered at 6 months and goats at 4 months. All animals were housed under standardized management practices appropriate for each species for their nutritional, housing, as well as environmental needs. Procedures for animal handling and tissue collection were handled in a way that was in line with ethical requirements for animal research. Ten random samples from healthy adult animals for each group were used to reduce biological variation due to age, sex, or physiological status. Blood was collected directly from the jugular or caudal vein and placed in plain tubes. Then all samples were kept at room temperature, and centrifuged at 3,000 rpm for 10 minutes to obtain the serum, which was subsequently stored at –20°C prior to analysis (Al-Hafidh and Albanna, 2023). All procedures were conducted in standardized and aseptic conditions to preserve sample integrity as well as enzyme stability.Serum ChE activity was measured using a (96-well ELISA kit) obtained from SUNLONG BIOTECH CO. The technique is based on antigen–antibody binding principle, and all procedures were followed as per the manufacturer's guidelines. Optical density was used at 450 nm in a microplate reader (BioTek ELx808), and ChE concentrations were figured out via using a calibration curve prepared from standard enzyme solutions included with the kit. A standard curve was plotted using six standards of the ChE standard (0, 3, 6, 12, 24, and 36 U/l) with their corresponding absorbance values of 0.129, 0.254, 0.274, 0.280, 0.305, and 0.408. Linear regression provided the equation y=0.0062x + 0.2011 with the correlation coefficient (R²=0.8187) indicating higher linearity over the assay range. Serum enzyme activity in all samples was measured off this standard curve, and the results were expressed as units per liter (U/l). All determinations were performed in duplicate, and the mean was taken for statistical analysis to minimize experimental variability. Data for all the animal groups were compared employing **GraphPad Prism version 9.0** software. Mean and standard deviation (SD) of ChE activity for each species were calculated. Statistical comparison between the nine groups was performed by **one-way analysis of variance** followed by **Tukey's post-hoc test** for multiple comparisons. Statistical differences were noted at *p* < 0.05. Graphical illustrations were plotted to depict the mean enzyme activity (± SD) per animal group. This method provided a reproducible and accurate estimation of the activity of ChE in different animal species. The use of a reproducible 96-well ELISA platform provided uniformity and analytical precision to all the samples. By comparing enzyme activity patterns between species, the study aimed to illuminate potential differences in physiology and how these might influence neurological health and environment (Zeyad et al., 2024). Ethical approvalThe Ethical Committee of the College of Veterinary Medicine, University of Mosul, Iraq (Approval No. UM.VET.2025.088) approved all animal procedures. These procedures were performed in accordance with accepted ethical standards. The data that support the findings of this study are available from the corresponding author upon reasonable request and are subject to institutional regulation. Animals were used humanely, and ethical standards were complied with. No funding was sourced from an external body. ResultsA calibration curve was subsequently constructed by the standard serial dilutions of ChE standards at the concentrations of 0, 3, 6, 12, 24, and 36 U/l. The corresponding absorbance was 0.129 to 0.408 at 450 nm. The relationship between the enzyme concentration and optical density was linear in this range and provided the regression equation y=0.0062x + 0.2011 with a correlation coefficient of R²=0.8187 (Fig. 1). This strong positive correlation indicates consistent linearity and demonstrates that the ELISA kit was accurate and sensitive for quantitative determination of serum ChE activity in the concentration range used. The calibration curve derived was used to calculate enzyme concentrations in serum samples from all animal groups.
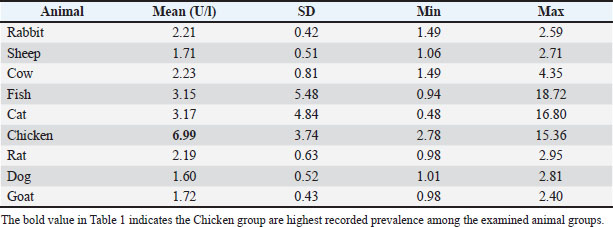
Fig. 1. Standard ChE ELISA curve illustrating linear correlation between enzyme concentration (U/l) and optical density at 450 nm (R²=0.8187). All serum samples from nine animal groups included rabbit, sheep, cow, fish, cat, chicken, rat, dog, and goat that were analyzed using the ELISA method. Quantitated ChE values showed significant interspecies differences, based on physiological activity and metabolism. The highest mean ChE activity was observed in chickens (6.99 U/l), followed by cats (3.17 U/l) and fish (3.15 U/l). All other animals but sheep, goats, and dogs had moderate activity of the enzyme (≈ 2.1–2.3 U/l), whereas sheep, goats, and dogs had the lowest enzyme activity (1.6–1.7 U/l). The results are as given in Table 1. Table 1. Animal groups of ChE activity levels (Mean ± SD).
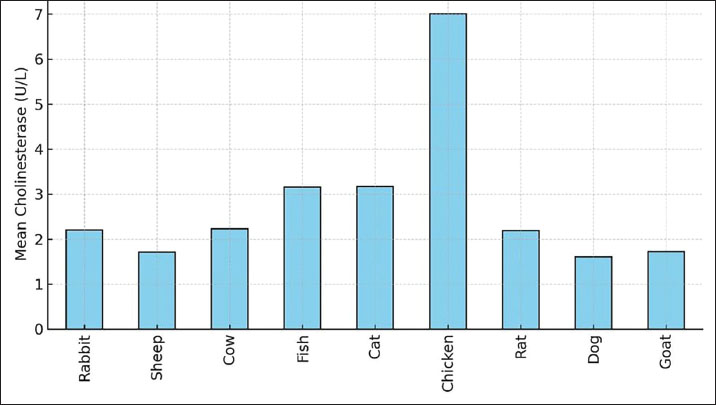
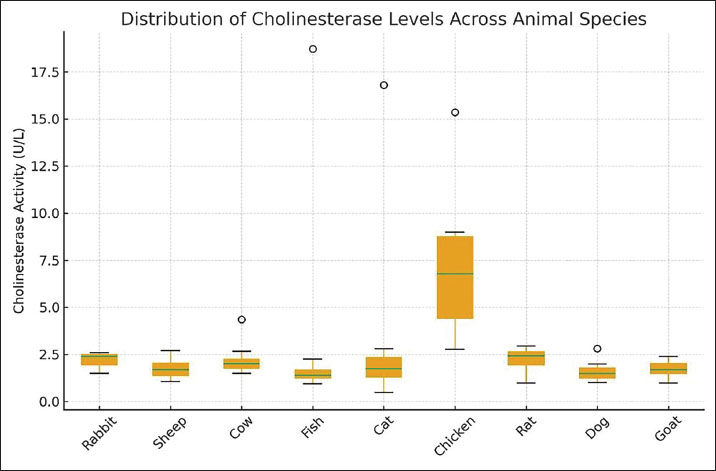
The average species distribution of ChE activity is given by Figure 2, and intra-group variability by Figure 3. From the results, it can be seen that chickens contained much greater enzyme activity than any of the other animals, and the least was in the case of dogs and sheep. Extensive inter-individual range in cats and fish, and less uniform, but narrower enzyme ranges in rabbits and goats, are further pointed out by the boxplot.The chicken group had the highest and most variable levels, followed by sheep, goats, and dogs with the lowest. Outliers are symbolized by circles.
Fig. 2. Histogram illustrating mean of serum ChE levels across nine animal species. Chickens displayed the highest activity, while sheep, goats, and dogs showed the lowest.
Fig. 3. Boxplot of serum ChE activity in nine animal species. Overall, there were unequivocal interspecies differences in serum ChE activity. The most active of the bird species was the chicken, presumably because it is a high-metabolism species with much neuromuscular activity required. Cats and fish evidenced high, though extremely variable levels of the enzyme, implicating ChE regulation by either environment or physiology. In contrast, Rats, cows, and rabbits have got moderate and steady levels characteristic of mammalian cholinergic homeostasis, whereas sheep, goats, and dogs possessed low activities, which were presumably due to reduced enzymatic turnover or metabolic demand. DiscussionThe present study has been planned as an explorative and comparative analysis of ChE activity in a variety of different animal species and intends to address the issue of interspecific variability and the potential use of ChE as a general bioindicator of neurophysiological condition and animal welfare (Hayashi et al., 2004). ChE activity has been generally accepted as a highly sensitive bioindicator for cholinergic activity and exposure to neurotoxic substances of the organophosphate and carbamate groups in terrestrial and aquatic species for a variety of years past. However, the analysis of ChE activities among species demands a cautious view of analytical performance, biological variability, and statistical strength (Miao et al., 2010). The approach using ELISA, as applied in this study, showed a positively proportional relationship between concentrations of ChE and optical density, as can be seen in the calibration curve equation (y=0.0062x + 0.2011). Although the coefficient of determination shows moderate linearity between variables (R²=0.8187), it cannot be fully considered as an ultimate bioanalytical calibration following recent guidelines (Massoulié et al., 1993). This was supported by recent studies, considering that accurate quantitative analysis for biomarkers should combine multiple assessments for bioanalytical validation, such as linearity, accuracy, precision, specificity, relatedness, and reproducibility for multiple assay analysis. In this way, it can be stated that this particular calibration curve was only useful for semi-quantitative analysis support rather than ultimate analytical calibration (Xing et al., 2021). The observed differences in ChE activity between rabbits, sheep, cows, fish, cats, chickens, rats, dogs, and goats are in line with earlier published data on the inter-species variability in the expression levels and regulation mechanisms of the cholinergic enzyme (Baldwin, 1966). ChE s, such as AChE and BChE, are critical in terminating a synaptic transmission event and play a significant role in xenobiotic detoxification by plasma BChE produced in the liver (Patočka et al., 2004). The mean activity of ChE enzymes observed in chickens was comparatively higher than those measured in most mammalian species and supports earlier views on higher cholinergic turnover activity and hist metabolic rate in avian species related to rapid neuromuscular activity and flying metabolism. However, without kinetic validation and physiological considerations, these results can at most provide evidence of a possible activity trend and do not immediately support functional supremacy of cholinergic physiological activity in avians (Nunes, 2011). This stability also serves as evidence of the ELISA methods being reliable in their reflection of true physiological enzyme levels (Khetarpal and Kumar, 2017). The level of ChE activity seen in intermediates in rats, rabbits, and cows is in keeping with that in mammals that have fairly steady metabolic needs. In mammals, AChE and BChE are localized in the nervous and muscular tissue and blood plasma, respectively. This anatomy reflects the detoxification process rather than involvement in synaptic transmission (Szotáková et al., 2004). Perhaps because of its similarity, it would be difficult to completely discount the effects of the environment in reaching the conclusion that the group was free from ChE-inhibiting chemicals (Ward and Glicksberg, 1971). The lower mean activities of ChE in sheep, goats, and dogs are also in accordance with the observed species-specific trends. The ruminants exhibit unique patterns of liver metabolism based on dietary and microbial factors and enzyme production rates, which could influence ChE concentrations in body fluids. The decreased activity of plasma BChE in dogs has been found to relate to species-specific expression levels rather than cholinergic dysfunction. These variations emphasize the advisability of species-specific reference ranges prior to the use of ChE for its proposed applications (Chang and Kam, 1999). Fish and cats had high mean values combined with high SDs due to isolated high values. Such high individual variation is well known in aquatic animals, where enzyme activity is known to be highly affected by water quality, season, stress, and toxicant exposure. There may also be compensatory enzyme induction due to inhibition of ChE activity in fish exposed to pesticides and heavy metals, making the single-point estimate complex to interpret. In domestic cats, individual variation of enzyme levels might be due to enzyme production in the liver, stress responses, or metabolic variations, as BChE is of mainly hepatocyte origin (Koelle, 1950; Balajee et al., 1994). In a comparative manner, the interspecific variation of ChE activity supports the hypothesis of a relationship between ChE activity and the specific needs of an organism, as well as ecological adaptation. Nevertheless, it is not possible, based upon these data, to draw conclusions about evolutionary optimization and environmental exposure, and thus, it only constitutes a preliminary comparative investigation that can help to elaborate a hypothesis in neurotoxicology and physiology (Petrichev, 2021). ELISA provides a number of benefits when considering traditional colorimetric assays, such as Ellman’s test, which include a high level of specificity, insusceptibility to sample hemolysis or turbidity, and the ability to perform high-throughput analysis (Yang et al., 2018). However, it is important to note that these benefits will in no way supersede the need for a high level of validation, especially when multiple assays are carried out on different species (Khetarpal and Kumar, 2017). On the other hand, within the context of the One Health approach, baseline information on the ChE activities within different animal species may play a role within integrated environmental and veterinary surveillance activities (Roy et al., 2025). Inasmuch as the inhibition of ChE activity remains the fundamental biomarker indicating the effects of neurotoxic pesticides, the need to establish species-specific reference values cannot be overemphasized (Fukuto, 1990). Fish species, birds, because of their sensitivity and particular roles within the ecosystem, may act as sentinel species, whereas mammals may act as indicators within agricultural or occupational settings (Yang et al., 2018). In closing, it is clear that the current study represents the demonstration of interspecific levels of ChE activity discernible by current technology, and in line with physiological diversity (Tedersoo et al., 2019; Roy et al., 2025). Although the results of the study represent a confirmatory indication of the relevance of ChE as a comparative marker, it must be recognized that the results contain several caveats in terms of study power in relation to validation of assays, sample size, as well as the availability of adequate statistical parameters (Fukuto, 1990). Despite the differences observed in ChE activities of species, the present study certainly did not examine the metabolic rate, detoxification pathways, or evolutionary adaptation. As such, any association between ChE activity and physiological demands, environmental exposure, or functional trait characteristic of a species should be considered descriptive rather than mechanistic. The study was conducted as a comparative and exploratory study that aimed to document interspecific variation rather than test causal hypotheses. Therefore, confirmation of conclusions involving metabolism, detoxification efficiency, or adaptive significance will require special experimental designs, kinetic studies, and controlled exposures. ConclusionThe present study sufficiently established measuring serum ChE activity across different male animal species. Interspecies variations were significantly reflected, signifying the vast metabolic and neurological variations among each group. Chickens showed the highest enzyme activity, while sheep, goats, and dogs showed the lowest activities. These differences highlight the physiological adaptations of both species and the potential for ChE to be an effective sensitive biomarker of neurophysiological health and environmental exposure. In general, ELISA-based measurement of ChE represents a very specific and useful method for the assessment of neurological health and environmental safety in the One Health context. AcknowledgmentsWe present our acknowledgments to Ninevah University for providing their labs and scientific supports also to the laboratories of the University of Mosul. Conflict of interestThe authors declare no conflicts of interest FundingNone. Data availability- All data supporting the findings of this study are available within the manuscript. - The data that support the findings of this study are not openly available due to reasons of sensitivity and are available from the corresponding author upon reasonable request. Authors' contributionsReem M. Mousa and Azhar Adil Mohammad developed the concept and design of the study. Ayman Albanna carried out the experimental work in the laboratory, including the handling of the experimental animals, the collection of blood samples, and the laboratory testing of the cholinesterase activity, and prepared the first draft of the manuscript. Reem M. Mousa and Azhar Adil Mohammad carried out the statistical analysis of the study and the formatting of the references using EndNote software. ReferencesAkki, R., Siracusa, R., Cordaro, M., Remigante, A., Morabito, R., Errami, M. and Marino, A. 2022. Adaptation to oxidative stress at cellular and tissue level. Arch. Physiol. Biochem. 128(2), 521–531. Al-Hafidh, Z.A. and Albanna, A. 2023. Environmental health risks of antibiotic residue in sewage discharged from Mosul city: an Elisa-based investigation. IOP Conf. Ser.: Earth Environ. Sci. 1259(1), 012036. Balajee, A.S., Oh, H.J. and Natarajan, A.T. 1994. Analysis of restriction enzyme-induced chromosome aberrations in the interstitial telomeric repeat sequences of CHO and CHE cells by FISH. Mutat. Res/Fundam. Mol. Mech. Mutagen. 307(1), 307–313. Baldwin, R.L. 1966. Enzymatic activities in mammary glands of several species. J. Dairy Sci. 49(12), 1533–1542. Ballard, C.G., Greig, N.H., Guillozet-Bongaarts, A.L., Enz, A. and Darvesh, S. 2005. Cholinesterases: roles in the brain during health and disease. Curr. Alzheimer Res. 2(3), 307–318. Bawaskar, H.S., Bawaskar, P.H. and Bawaskar, P.H. 2015. RBC acetyl cholinesterase: a poor man's early diagnostic biomarker for familial alzheimer's and Parkinson's disease dementia. J. Neurosci. Rural Pract. 6(1), 33. Beaulieu, M. 2024. Capturing wild animal welfare: a physiological perspective. Biolo. Rev. 99(1), 1–22. Chambers, J.E., Boone, J.S., Carr, R.L., Chambers, H.W. and Straus, D.L. 2002. Biomarkers as predictors in health and ecological risk assessment. Hum. Ecol. Risk Assess. 8(1), 165–176. Chang, G.W.M. and Kam, P.C.A. 1999. The physiological and pharmacological roles of cytochrome P450 isoenzymes. Anaesthesia 54(1), 42–50. Colovic, M.B., Krstic, D.Z., Lazarevic-Pasti, T.D., Bondzic, A.M. and Vasic, V.M. 2013. Acetylcholinesterase inhibitors: pharmacology and toxicology. Curr. Neuropharmacol. 11(3), 315–335. Fukuto, T.R. 1990. Mechanism of action of organophosphorus and carbamate insecticides. Environ. Health Perspect. 87, 245. Hayashi, Y., Matsuda, R., Maitani, T., Imai, K., Nishimura, W., Ito, K. and Maeda, M. 2004. Precision, limit of detection and range of quantitation in competitive ELISA. Anal. Chem. 76(5), 1295–1301. Jiang, Y., Alam, J.J., Gomperts, S.N., Maruff, P., Lemstra, A.W., Germann, U.A., Stavrides, P.H., Darji, S., Malampati, S., Peddy, J. and Bleiwas, C. 2022. Preclinical and randomized clinical evaluation of the p38α kinase inhibitor neflamapimod for basal forebrain cholinergic degeneration. Nat. Commun. 13(1), 5308. Khan, M.Z. and Law, F.C. 2005. Adverse effects of pesticides and related chemicals on enzyme and hormone systems of fish, amphibians and reptiles: a review. Proc. Pak. Acad. Sci. 42(4), 315–323. Khetarpal, R.K. and Kumar, C.A. 2017. ELISA methodology. In Molecular methods in plant pathology. Boca Raton, FL: CRC Press, pp. 329–341. Koelle, G.B. 1950. The histochemical differentiation of types of cholinesterases and their localizations in tissues of the cat. J. Pharmacol. Exp. Ther. 100(2), 158–179. Kondo, M., Hada, T., Fukui, K., Iwasaki, A., Higashino, K. and Yasukawa, K. 1995. Enzyme-linked immunosorbent assay (ELISA) for Aleuria aurantia lectin-reactive serum cholinesterase to differentiate liver cirrhosis and chronic hepatitis. Clin. Chim. Acta. 243(1), 1–9. Layer, P.G. 1983. Comparative localization of acetylcholinesterase and pseudocholinesterase during morphogenesis of the chicken brain. Proc. Nat. Acad. Sci. 80(20), 6413–6417. Lionetto, M.G., Caricato, R., Calisi, A., Giordano, M.E. and Schettino, T. 2013. Acetylcholinesterase as a biomarker in environmental and occupational medicine: new insights and future perspectives. BioMed Res. Int. 2013(1), 321213. Massoulié, J., Sussman, J., Bon, S. and Silman, I. 1993. Structure and functions of acetylcholinesterase and butyrylcholinesterase. Prog. Brain Res. 98, 139–146. Miao, Y., He, N. and Zhu, J.J. 2010. History and new developments of assays for cholinesterase activity and inhibition. Chem. Rev. 110(9), 5216–5234. Nunes, B. 2011. The use of cholinesterases in ecotoxicology. Rev. Environ. Contam. Toxicol. 212, 29–59. Ord, M.G. and Thompson, R.H.S. 1950. The distribution of cholinesterase types in mammalian tissues. Biochem. J. 46(3), 346. Patočka, J., Kuča, K. and Jun, D. 2004. Acetylcholinesterase and butyrylcholinesterase–important enzymes of human body. Acta Med. (Hradec Kralove). 47(4), 215–228. Petrichev, M. 2021. Some important biochemical parameters in clinical veterinary toxicology appears as a journal article published in Tradition and Modernity in Veterinary Medicine (TMVM). Sofia, Bulgaria. Pezzementi, L., Nachon, F. and Chatonnet, A. 2011. Evolution of acetylcholinesterase and butyrylcholinesterase in the vertebrates: an atypical butyrylcholinesterase from the Medaka Oryzias latipes. PLoS One 6(2), e17396. Roy, S., Malla, S., Dekari, D. and Choudhury, T.G. 2025. One Health approach: human, environment, and animal health. In Management of fish diseases. Eds., Sahoo, P.K. and Saha, K. Singapore: Springer Nature Singapore, pp: 281–297. Siciliano, G., Chico, L., Lo Gerfo, A., Simoncini, C., Schirinzi, E. and Ricci, G. 2020. Exercise-related oxidative stress as mechanism to fight physical dysfunction in neuromuscular disorders. Front. Physiol. 11, 451. Spurgeon, D., Lahive, E., Robinson, A., Short, S. and Kille, P. 2020. Species sensitivity to toxic substances: evolution, ecology and applications. Front. Environ. Sci. 8, 588380. Szotáková, B., Baliharová, V., Lamka, J., Nožinová, E., Wsól, V., Velık, J., Machala, M., Neča, J., Souček, P., Šusová, S. and Skálová, L. 2004. Comparison of in vitro activities of biotransformation enzymes in pig, cattle, goat and sheep. Res. Vet. Sci. 76(1), 43–51. Tedersoo, L., Drenkhan, R., Anslan, S., Morales‐Rodriguez, C. and Cleary, M., 2019. High‐throughput identification and diagnostics of pathogens and pests: overview and practical recommendations. Mol. Ecol. Res. 19(1), 47–76. Tougu, V. 2001. Acetylcholinesterase: mechanism of catalysis and inhibition. Curr. Med. Chem-Central Nerv. Syst. Agents, 1(2), 155–170. Ward, F.P. and Glicksberg, C.L. 1971. Effects of dichlorvos on blood cholinesterase activity in dogs. J. Am. Vet. Med. Assoc. 158(4), 457–461. Xing, S., Li, Q., Xiong, B., Chen, Y., Feng, F., Liu, W. and Sun, H. 2021. Structure and therapeutic uses of butyrylcholinesterase: application in detoxification, Alzheimer's disease, and fat metabolism. Med. Res. Rev. 41(2), 858–901. Yang, M., Zhao, Y., Wang, L., Paulsen, M., Simpson, C.D., Liu, F., Du, D. and Lin, Y. 2018. Simultaneous detection of dual biomarkers from humans exposed to organophosphorus pesticides by combination of immunochromatographic test strip and ellman assay. Biosens. Bioelectron. 104, 39–44. Zeyad , H., Albanna, A. and Alomari, A. 2024. Combating dioxin and furan pollution in Mosul City's industrial water effluents: the efficacy of ELISA detection and tannic acid treatment. Egypt. J. Aquat. Biol. Fish. 28(4), 1039–1047. | ||
| How to Cite this Article |
| Pubmed Style Mousa RM, Mohammad AA, Albanna A. ELISA-based assessment of cholinesterase levels in different animal groups: Insights into neurological health. Open Vet. J.. 2026; 16(3): 1488-1495. doi:10.5455/OVJ.2026.v16.i3.8 Web Style Mousa RM, Mohammad AA, Albanna A. ELISA-based assessment of cholinesterase levels in different animal groups: Insights into neurological health. https://www.openveterinaryjournal.com/?mno=295712 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.8 AMA (American Medical Association) Style Mousa RM, Mohammad AA, Albanna A. ELISA-based assessment of cholinesterase levels in different animal groups: Insights into neurological health. Open Vet. J.. 2026; 16(3): 1488-1495. doi:10.5455/OVJ.2026.v16.i3.8 Vancouver/ICMJE Style Mousa RM, Mohammad AA, Albanna A. ELISA-based assessment of cholinesterase levels in different animal groups: Insights into neurological health. Open Vet. J.. (2026), [cited March 31, 2026]; 16(3): 1488-1495. doi:10.5455/OVJ.2026.v16.i3.8 Harvard Style Mousa, R. M., Mohammad, . A. A. & Albanna, . A. (2026) ELISA-based assessment of cholinesterase levels in different animal groups: Insights into neurological health. Open Vet. J., 16 (3), 1488-1495. doi:10.5455/OVJ.2026.v16.i3.8 Turabian Style Mousa, Reem M., Azhar Adil Mohammad, and Ayman Albanna. 2026. ELISA-based assessment of cholinesterase levels in different animal groups: Insights into neurological health. Open Veterinary Journal, 16 (3), 1488-1495. doi:10.5455/OVJ.2026.v16.i3.8 Chicago Style Mousa, Reem M., Azhar Adil Mohammad, and Ayman Albanna. "ELISA-based assessment of cholinesterase levels in different animal groups: Insights into neurological health." Open Veterinary Journal 16 (2026), 1488-1495. doi:10.5455/OVJ.2026.v16.i3.8 MLA (The Modern Language Association) Style Mousa, Reem M., Azhar Adil Mohammad, and Ayman Albanna. "ELISA-based assessment of cholinesterase levels in different animal groups: Insights into neurological health." Open Veterinary Journal 16.3 (2026), 1488-1495. Print. doi:10.5455/OVJ.2026.v16.i3.8 APA (American Psychological Association) Style Mousa, R. M., Mohammad, . A. A. & Albanna, . A. (2026) ELISA-based assessment of cholinesterase levels in different animal groups: Insights into neurological health. Open Veterinary Journal, 16 (3), 1488-1495. doi:10.5455/OVJ.2026.v16.i3.8 |