| Review Article | ||
Open Vet. J.. 2026; 16(2): 768-787 Open Veterinary Journal, (2026), Vol. 16(2): 768-787 Review Article Ebola virus disease: A persistent challenge in global health securityMuniroh Muniroh1,2*, Luluk Hermawati3, Fathurrohim Fathurrohim4, Siti Darifah5, Lola Febriana Dewi6, Neshya Ruriana Putri7, Yessy Andriani Fauziah8, Eveline Yulia Darmadi8, Dwi Setianingtyas9, Aswin Rafif Khairullah10, Abdul Hadi Furqoni11 and Bima Putra Pratama121Graduate School Student, Universitas Islam Negeri (UIN) Syarif Hidayatullah Jakarta, South Tangerang, Indonesia 2Department of Clinical Pathology, Faculty of Medicine, Universitas Islam Negeri (UIN) Syarif Hidayatullah Jakarta, South Tangerang, Indonesia 3Department of Medical Biology, Faculty of Medicine and Health Sciences, Universitas Sultan Ageng Tirtayasa, Serang, Indonesia 4Department of Microbiology, Faculty of Medicine, Universitas Sriwijaya, Palembang, Indonesia 5Department of Community Medicine, Faculty of Medicine and Health Sciences, Universitas Sultan Ageng Tirtayasa, Serang, Indonesia 6Department of Oral Biology, Dentistry Study Program, Faculty of Medicine, Universitas Sriwijaya, Palembang, Indonesia 7Department of Clinical Pathology, Faculty of Medicine, Universitas Sultan Ageng Tirtaysa, Serang, Indonesia 8School of Dental Medicine, Universitas Ciputra, Surabaya, Indonesia 9Department of Oral Medicine, Faculty of Dentistry, Universitas Hang Tuah, Surabaya, Indonesia 10Research Center for Veterinary Science, National Research and Innovation Agency (BRIN), Bogor, Indonesia 11Center for Biomedical Research, National Research and Innovation Agency (BRIN), Bogor, Indonesia 12Research Center for Process Technology, National Research and Innovation Agency (BRIN), South Tangerang, Indonesia *Corresponding Author: Muniroh Muniroh. Universitas Islam Negeri (UIN) Syarif Hidayatullah Jakarta, South Tangerang, Indonesia. Email: muniroh.sps.uinjkt [at] gmail.com Submitted: 22/11/2025 Revised: 01/01/2026 Accepted: 09/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
ABSTRACTEbola virus disease (EVD) is a highly fatal zoonotic disease caused by a negative-stranded RNA virus of the genus Ebolavirus in the family Filoviridae. Since its first discovery in 1976 in Central Africa, the disease has triggered several large outbreaks with a mortality rate that can reach 90%. Ebola is transmitted through direct contact with the blood, body fluids, or tissues of infected individuals, including corpses, and can also spread in healthcare facilities if infection control procedures are not properly implemented. Fruit bats of the family Pteropodidae are thought to be the natural reservoir of the virus, playing a role in maintaining the ecological cycle and facilitating zoonotic transmission to humans through intermediary animals such as non-human primates. Clinically, EVD typically begins with high fever, muscle pain, vomiting, diarrhea, and extensive bleeding that can progress to multiple organ failure. Barriers to early detection, limited diagnostic laboratories, and weak health system preparedness in endemic areas contribute to accelerating the spread of the disease. The major outbreak that occurred in West Africa in 2014–2016 provided a crucial momentum for improving the global health system, particularly in terms of surveillance, rapid detection, and coordination between countries. Current EVD prevention and control efforts focus on a One Health approach, the implementation of stringent biosafety standards, the use of the rVSV-ZEBOV vaccine, and the development of monoclonal antibody-based antiviral therapies. This review reviews the latest developments in EVD pathogenesis, epidemiology, and mitigation strategies to strengthen preparedness for potential future outbreaks. Keywords: EVD, Zoonosis, Filoviridae, Vaccination, Virus. IntroductionEbola virus disease (EVD) is a highly dangerous zoonotic disease caused by a single-stranded RNA virus of the genus Ebolavirus in the family Filoviridae (Jacob et al., 2020). The disease was first recognized in 1976 in Yambuku, Democratic Republic of the Congo, and in Nzara, South Sudan, following two large outbreaks with very high mortality rates (Breman et al., 2016). Since its initial discovery, EVD has remained a major threat to global public health due to its rapid transmission, high case fatality rate, and significant socioeconomic impact in affected areas (Omoleke et al., 2016). Biologically, the Ebola virus belongs to the filovirus family, a filamentous virus of varying lengths and a negative-stranded RNA genome (Hume and Mühlberger, 2019). This genome encodes seven key structural proteins, including glycoprotein (GP), nucleoprotein (NP), and RNA-dependent RNA polymerase (L) (Jain et al., 2021). The complexity of its structure and replication processes makes this virus highly virulent and able to adapt rapidly to various mammalian hosts (Rivera and Messaoudi, 2016). Fruit bats from the Pteropodidae family are thought to be its natural reservoir, playing a role in maintaining the virus’s presence in the environment and enabling its spread to other wildlife, such as non-human primates, before ultimately infecting humans (Bhatia et al., 2024). EVD transmission to humans occurs through direct contact with the blood, bodily fluids, or tissue of an infected person, including corpses (Rewar and Mirdha, 2014). Transmission in healthcare facilities (nosocomial) often occurs when infection prevention and control protocols are not properly implemented, particularly in areas with limited resources (Baller et al., 2022). The incubation period of the disease ranges from 2 to 21 days, and initial symptoms are generally nonspecific, such as fever, muscle aches, and fatigue (Hasan et al., 2019). As the disease progresses, patients can experience vomiting, diarrhea, internal and external bleeding, and even multiorgan failure (Hussein, 2023). The mortality rate from this infection varies between 50% and 90%, depending on the virus strain and the quality of medical care available (Izudi and Bajunirwe, 2024). The large EVD outbreak in West Africa in 2014–2016 was a landmark event in the history of the disease, with over 28,000 cases and approximately 11,000 deaths (Collier et al., 2024). This event exposed the weaknesses of global health systems in dealing with high-risk infectious diseases and emphasized the need for more effective international coordination in outbreak management (Stephens et al., 2024). Furthermore, the recurrence of Ebola cases in the post-outbreak period demonstrated that the virus could persist in survivors and potentially trigger new infections, highlighting the importance of further research into viral persistence in immunoprivileged tissues (Den Boon et al., 2019). In an era of globalization with high human mobility, the potential for disease spread between countries is increasing, necessitating the strengthening of surveillance systems, early detection, and public health emergency preparedness (Woolhouse et al., 2015). Given that EVD is a zoonotic disease with human–animal–environment interfaces, applying the One Health approach is important to improve timely detection, coordinated response, and prevention strategies across sectors. This approach—widely emphasized in the context of neglected zoonotic diseases—highlights the need for integrated surveillance, intersectoral collaboration, and joint risk mitigation strategies spanning clinical care, veterinary health, and environmental monitoring (Meurer, 2025). EVD control and prevention require a multidisciplinary approach encompassing the interaction between human, animal, and environmental health, in accordance with the One Health concept (Rodriguez, 2024). Furthermore, the development of vaccines and antiviral therapies is a top priority in modern research to reduce mortality and prevent virus transmission in potential future outbreaks (Marzi and Feldmann, 2014). Therefore, this review aims to provide a comprehensive understanding of the biological characteristics of the Ebola virus, its pathogenesis mechanisms, epidemiological patterns, diagnostic strategies, and recent advances in prevention and control efforts. By synthesizing the latest scientific data, this article is expected to serve as a reference for researchers, healthcare professionals, and policymakers in strengthening response capacity to high-risk infectious diseases such as EVD. EtiologyEVD is a zoonotic disease caused by the Ebola virus, which belongs to the genus Ebolavirus, family Filoviridae, and order Mononegavirales (Mirazimi, 2015). This virus is a single-stranded, negative-sense RNA virus (ssRNA) with a lipid envelope and a distinctive filamentous shape, which are the main characteristics of members of the Filoviridae (Emanuel et al., 2018). This fiber-like or thread-like shape is the basis for the name of the family, derived from the Latin word filum, meaning “thread” (Kuhn et al., 2010). Classificationally, the Ebolavirus genus consists of five main species that have been identified: Zaire ebolavirus (EBOV), Sudan ebolavirus (SUDV), Bundibugyo ebolavirus (BDBV), Taï Forest ebolavirus, and Reston ebolavirus (RESTV) (Jun et al., 2015). Among these five species, EBOV is the most virulent and is responsible for most major outbreaks with a high mortality rate of 70%–90%. SUDV and BDBV are also capable of causing disease in humans, but with generally lower fatality rates (Matson et al., 2025). In contrast, RESTV is found circulating in Southeast Asia, primarily in non-human primates and pigs, and has so far not been shown to cause disease in humans although it can cause subclinical infections (Peñas et al., 2019). The Ebolavirus genome is a linear RNA approximately 19 kilobases long that encodes seven major structural and functional proteins: NP, viral protein 35 (VP35), VP40, GP, VP30, VP24, and RNA-dependent RNA polymerase (L) (Shu et al., 2019). Each protein has a specific function in the viral life cycle. NP forms a nucleocapsid complex that protects the genomic RNA from degradation, while VP35 and VP30 are involved in replication and transcription (Almeida-Pinto et al., 2024). VP40 and VP24 proteins play a role in virion assembly and the release of new virus particles (Licata et al., 2004). GP plays a central role in the binding and entry of the virus into host cells through interactions with surface receptors such as C-type lectins, TIM-1, and Niemann–Pick C1 (NPC1) (Wang et al., 2016). Ebolavirus infection begins when the virus particle binds to specific receptors on the surface of the host cell (Falasca et al., 2015). It then enters the cell through endocytosis and releases its genome into the cytoplasm (Lai et al., 2014). The entire replication process occurs in the cytoplasm, with the enzyme RNA-dependent RNA polymerase (L) responsible for producing mRNA and duplicating new viral genomes (Fang et al., 2022). Newly formed virions are then assembled at the plasma membrane and released from the cell through budding, incorporating the lipid layer of the host cell membrane as their envelope (Husby et al., 2022). From a molecular perspective, phylogenetic studies have revealed that Ebolavirus exhibits a high degree of genetic diversity, both between species and between outbreaks (Judson and Munster, 2023). These differences are primarily found in the GP, VP35, and L genes, which may influence virulence, tissue preference, and the resulting immune response (Misasi and Sullivan, 2014). A thorough understanding of the genome structure, genetic organization, and replication mechanisms of Ebola virus is crucial for the development of effective diagnostic methods, vaccines, and antiviral therapies against the various circulating strains (Ghosh et al., 2021). ReservoirIdentifying the natural reservoir of Ebolavirus is a key element in understanding the epidemiology and ecological dynamics of the disease (Shen et al., 2025). Although numerous studies have been conducted since the first outbreak in 1976, there is still no consensus on the exact host species that maintains the virus in the natural environment (Kadanali and Karagoz, 2015; Sivanandy et al., 2022). Nevertheless, serological, molecular, and ecological evidence consistently indicate that fruit bats of the Pteropodidae family are the most likely candidate natural reservoirs of the virus (Vetter et al., 2016). Several bat species, such as Hypsignathus monstrosus, Epomops franqueti, and Myonycteris torquata, have been found to carry Ebolavirus RNA or antibodies without showing symptoms of disease (De Nys et al., 2018). This indicates that these bats are capable of carrying the virus in a latent or subclinical form, an important characteristic of a natural reservoir (Garry, 2019). The successful isolation of EBOV from healthy bat tissue in Gabon and the Democratic Republic of the Congo further confirms the hypothesis that bats are the primary reservoir of the virus in their natural habitat (Leendertz et al., 2016). Fruit bats possess ecological characteristics that allow Ebolavirus to persist and circulate in the natural environment (Weinberg and Yovel, 2022). They are nocturnal, capable of long-distance travel, and live in large colonies, facilitating both individual-to-individual transmission and the spread of the virus across geographic areas (Judson et al., 2015). Their habit of leaving fruit scraps or contaminating them with saliva and excreta can also provide an indirect source of transmission to other animals, including nonhuman primates and antelopes, which could potentially serve as intermediate hosts (Mendez-Rodríguez, 2015). This interspecies transfer of the virus is thought to be the primary cause of initial human infections in endemic areas, primarily through wildlife hunting and consumption of contaminated meat (Beeching et al., 2014). In addition to bats, several studies have also found antibodies to Ebolavirus in various other wildlife, such as nonhuman primates (e.g., gorillas and chimpanzees), duikers, and several rodents (Caron et al., 2018; Schmidt et al., 2019; Rojas et al., 2020). However, because infection in these species usually causes fatal disease and they are unable to maintain long-term viral circulation, these animals are more appropriately viewed as incidental hosts or hosts that only play a role in temporary amplification, rather than as primary reservoirs of the virus (Hayman et al., 2022). From an ecological perspective, the existence of natural reservoirs of Ebolavirus is significantly influenced by environmental changes and human activities (Judson et al., 2016). Activities such as deforestation, habitat fragmentation, and agricultural expansion increase the frequency of contact between humans and virus-carrying wildlife (Banik and Basu, 2025). These anthropogenic activities create the potential for spillover, the transfer of viruses from wild animals to humans, which can trigger local epidemics (Lee-Cruz et al., 2021). Therefore, a One Health approach, which emphasizes the interconnectedness of human, animal, and environmental health, is crucial for understanding reservoir dynamics and preventing interspecies transmission (Ramirez-Plascencia et al., 2025). EpidemiologyThe geographic distribution of Ebolavirus shows a consistent endemic pattern in tropical Sub-Saharan Africa, particularly in Central and West Africa (Omoleke et al., 2016). Since its first identification in 1976 in Yambuku, Democratic Republic of the Congo (Breman et al., 2016), and in Nzara, South Sudan (Stein, 2015), outbreaks have occurred in several African countries, suggesting a stable ecological cycle between the virus, its natural reservoir, and intermediate hosts (Dewan and Khan, 2025). The majority of outbreaks have occurred in tropical rainforest areas, the primary habitat of fruit bats (Pteropodidae), considered the primary natural reservoir of the virus (Hussein, 2023). Table 1 summarizes the chronology and geographic distribution of Ebolavirus outbreaks from 1976 to 2023. Table 1. Chronology and geographic distribution of Ebolavirus outbreaks.
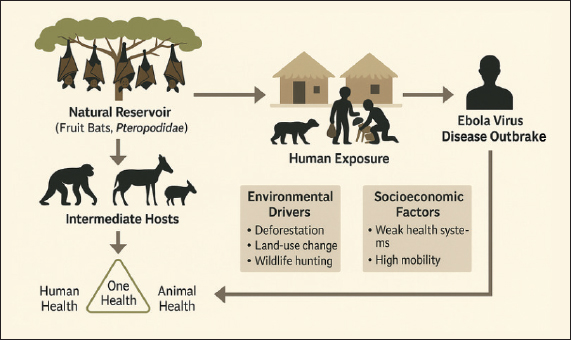
Historically, large-scale Ebolavirus outbreaks have been recorded in several Central African countries, including the Democratic Republic of the Congo (Breman et al., 2016), South Sudan (Stein, 2015), Uganda (Branda et al., 2023), Gabon (Wauquier et al., 2009), and the Republic of the Congo, as well as in West Africa, including Guinea (Raab et al., 2021), Liberia (Fall, 2019), and Sierra Leone (Fitzgerald et al., 2016). The West African outbreak of 2014–2016 was the largest and most complex in history, with over 28,000 cases and over 11,000 deaths (Den Boon et al., 2019). This marked a significant shift in the distribution of Ebolavirus, which had previously been largely confined to the forests of Central Africa (Jacob et al., 2020). Factors such as cross-border transmission and high population mobility played a significant role in accelerating the spread of the virus to new regions (Rojas et al., 2020). Following previous large outbreaks, several unusual cases of EVD recurred in the Democratic Republic of the Congo (2018–2020, 2022, and 2023) (Guetiya Wadoum et al., 2021; Hangi et al., 2024; Milambo and Businge, 2025), Uganda (2022) (Rai et al., 2022), South Sudan (2021) (Kabami et al., 2024), and Guinea (2021) (Pare et al., 2023). This repeated pattern of outbreaks in the same regions indicates that Ebolavirus has adapted ecologically to tropical environments, enabling long-term persistence through a zoonotic cycle involving bats and other wildlife (Pigott et al., 2014). Outside Africa, RESTV, which is not pathogenic to humans, has been found in non-human primates and domestic pigs in the Philippines and China, suggesting a broader geographic distribution of the virus even though the clinical risk to humans remains low (Pan et al., 2014; Peñas et al., 2019). Environmental factors, climate, and human activity play a significant role in determining the spread of Ebolavirus (Redding et al., 2019). Deforestation, wildlife hunting, and agricultural expansion in tropical forests increase the potential for contact between humans and wildlife carrying the virus, facilitating zoonotic spillover (Esposito et al., 2023). Furthermore, global climate change, which affects rainfall patterns and food availability for bats, may alter the distribution of natural reservoirs, ultimately impacting the geographic distribution of the disease (Olivero et al., 2020). Spatial analysis and ecological risk modeling, combining information on environmental conditions, bat populations, and human mobility patterns, indicate that areas at high risk for Ebola outbreaks extend from the equatorial forest belt of Central Africa to tropical West Africa (Baluma Didier et al., 2024). This approach provides a valuable tool for designing evidence-based mitigation strategies to predict potential locations for new outbreaks. The pathway of Ebola virus emergence at the forest–village interface, including environmental and socioeconomic drivers of human exposure, is summarized in Figure 1.
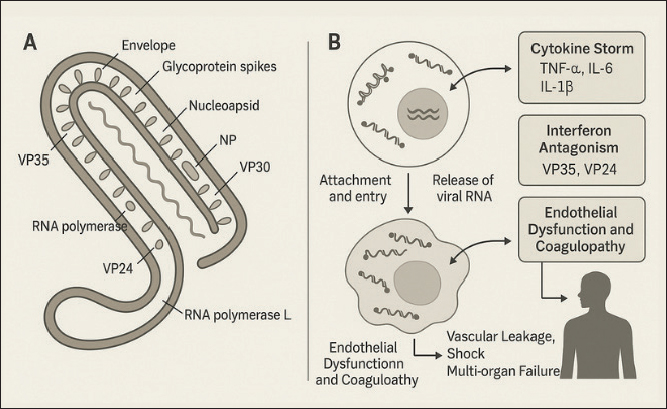
Fig. 1. Ebola virus spillover pathway at the forest–village interface. PathogenesisThe pathogenesis of EVD is a complex process, involving a dynamic interaction between viral virulence factors and host immune defense mechanisms (Muñoz-Fontela and McElroy, 2017). After entering the body through direct contact with contaminated blood, body fluids, or tissue, Ebolavirus rapidly infects key target cells, such as macrophages, monocytes, and dendritic cells (Vine et al., 2017). These cells play a crucial role in the innate immune response, so early infection significantly disrupts the host’s immune system (Ndayambaje et al., 2025). Infection begins when the viral GP binds to receptors on the cell surface, including C-type lectins, T-cell immunoglobulin and mucin domain 1 (TIM-1), and Niemann–Pick C1 (NPC1) (Kuroda et al., 2015). After the virus enters the cell via endocytosis, fusion of the viral membrane and the endosome releases the RNA genome into the cytoplasm (Winter et al., 2023). The Ebolavirus genome is a negative-polarity, single-stranded RNA (-ssRNA) that encodes seven structural and nonstructural proteins (Jain et al., 2021). Transcription and replication of the genome are carried out by the enzyme RNA-dependent RNA polymerase (L protein), which results in the synthesis of large amounts of viral proteins and the formation of new virions in the cytoplasm (Donahue et al., 2025). Massive infection of mononuclear phagocytes triggers the release of proinflammatory cytokines, including tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), and interleukin-1β (IL-1β) (Obeagu and Ezeala, 2025). This excessive cytokine production causes a cytokine storm, which contributes to increased vascular permeability and systemic tissue damage (Escudero-Pérez et al., 2014). The virus also exerts immunopathological effects by inducing apoptosis in T and B lymphocytes, resulting in severe lymphopenia and loss of control over viral replication (Zampieri et al., 2007). Furthermore, Ebolavirus has an effective strategy to evade the immune response. The VP35 protein acts as an interferon antagonist by inhibiting the detection of viral RNA by the RIG-I-like receptor, while VP24 interferes with the phosphorylation and translocation of the STAT1 transcription factor into the nucleus, thereby suppressing the type I interferon signaling pathway (Leung et al., 2011). Consequently, activation of the innate immune response is impaired, and the production of endogenous interferons, such as interferon-α and interferon-β, becomes less effective (Cárdenas et al., 2006). This gives the virus time to replicate extensively before the adaptive immune system can be activated (Dyall et al., 2017). Endothelial damage caused by both direct viral infection and inflammatory mediators results in increased vascular permeability, plasma leakage, and hypovolemia (Wahl-Jensen et al., 2006). This condition, combined with activation of the coagulation system due to tissue factor released from infected macrophages, triggers disseminated intravascular coagulation, internal bleeding, and circulatory shock (Mariappan et al., 2021). Furthermore, hepatocyte infection impairs clotting factor synthesis, worsening coagulopathy (Obeagu and Ezeala, 2025). Involvement of other organs such as the kidneys, spleen, and heart accelerates the development of multiorgan failure, which is the leading cause of death in severe EVD cases (Singh et al., 2017). In patients who survive, clinical recovery is usually associated with the development of an effective adaptive immune response (Ndayambaje et al., 2024). The production of neutralizing antibodies against the GP and the activation of CD8+ cytotoxic T cells play a crucial role in clearing the virus (Perez-Valencia et al., 2023). Conversely, patients with a slow or inadequate adaptive immune response tend to experience rapid disease progression toward the terminal phase (Zhang et al., 2023). Immune responseThe immune response to Ebolavirus infection is the result of a complex interaction between the innate and adaptive immune systems (Kumar, 2016). The virus has a remarkable ability to evade detection and suppression by the host immune system through a variety of sophisticated molecular mechanisms (Falasca et al., 2015). The success or failure of the host immune response is a major determinant of disease severity and the likelihood of clinical recovery (Muñoz-Fontela and McElroy, 2017). In the early stages of infection, the innate immune system plays a crucial role in recognizing viruses through pattern recognition receptors, such as Toll-like receptors and RIG-I-like receptors (Thompson et al., 2011). Activation of these receptors should stimulate the production of type I interferons (IFN-α and IFN-β), which suppress viral replication (Locke et al., 2021). However, Ebolavirus has an effective evasion mechanism. The VP35 protein acts as an interferon antagonist by inhibiting viral RNA detection by RIG-I and MDA5 and suppressing the activation of the transcription factors IRF3 and NF-κB (Cárdenas et al., 2006). Furthermore, VP24 interferes with the phosphorylation and translocation of STAT1 to the nucleus, thereby suppressing the expression of interferon-stimulated genes, which are essential for antiviral defense (Kühl and Pöhlmann, 2012). Disruption of this interferon pathway reduces the ability of immune cells to limit viral spread (Ndayambaje et al., 2025). Infected macrophages and dendritic cells become primary sites of viral replication and trigger the excessive release of pro-inflammatory cytokines such as TNF-α, IL-6, and IL-1β (Olukitibi et al., 2019). This condition triggers a cytokine storm, which causes vascular endothelial damage, increased vascular permeability, and systemic hemodynamic disturbances (Vine et al., 2017). Furthermore, Ebolavirus suppresses dendritic cell activation, thus inhibiting antigen processing and delivery to T cells, resulting in a weakened adaptive immune response (Lubaki et al., 2016). Successful viral clearance depends heavily on the timely activation of the adaptive immune system (Jacob et al., 2020). Cytotoxic CD8+ T cells play a key role in destroying infected cells, while CD4+ T cells support B cell differentiation and the production of specific antibodies (LaVergne et al., 2020). Serological studies have shown that patients who survive EVD generally have a rapid and robust adaptive immune response, characterized by effective T cell activation and the production of neutralizing antibodies against surface GP (Longet et al., 2021). These antibodies prevent the virus from attaching to and fusing with the target cell membrane and mark viral particles for destruction through antibody-dependent cellular cytotoxicity and complement-mediated lysis (Singh et al., 2018). In contrast, patients who experience fatal infections tend to have a delayed or inadequate adaptive immune response, characterized by low antibody levels and impaired T-cell function (Longet et al., 2021). Furthermore, massive apoptosis of lymphocytes due to the indirect effects of viral infection leads to severe lymphopenia, further compromising the host’s ability to control viral replication (Zampieri et al., 2007). An uncontrolled immune response can have pathological effects. Excessive cytokine release and persistent activation of macrophages and neutrophils can lead to extensive tissue damage and multiorgan failure (Obeagu and Ezeala, 2025). Therefore, the balance between a protective antiviral response and control of excessive inflammation is a critical factor in determining the clinical outcome of infection (Mahanty et al., 2003). Recovered patients typically develop long-term immunity that provides protection against reinfection by the same strain (Gupta et al., 2004). Antibodies against the GP and NP can persist for years, indicating the development of effective immune memory (McLean et al., 2023). Understanding the mechanisms by which this protective immunity develops has led to the development of a modern vaccine based on a recombinant adenovirus vector (rVSV-ZEBOV), which is capable of inducing robust humoral and cellular immune responses (Pinski and Messaoudi, 2020). The schematic overview of Ebola virion structure and its main steps of infection and immunopathogenesis in human immune cells is shown in Figure 2.
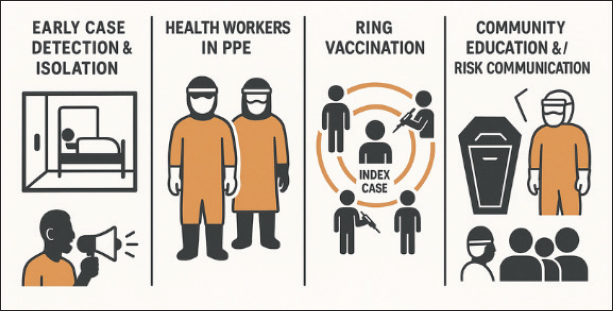
Fig. 2. Ebola virus structure (A) and pathogenesis (B). Clinical manifestationsThe clinical manifestations of EVD reflect a progressive disease, affecting multiple systems, and are strongly influenced by the interaction between viral virulence, the infectious dose, and the host’s immune response (Chiappelli et al., 2015). The incubation period lasts between 2 and 21 days (average 7–10 days), during which the infected individual is not infectious (Do and Lee, 2016). Initial symptoms are usually acute and nonspecific, often mimicking other tropical diseases, such as malaria, typhoid fever, or dengue fever in the early stages (Hasan et al., 2019). In the early stages of infection (days 1–3 of symptoms), patients typically experience a sudden onset of high fever, chills, severe fatigue, muscle aches (myalgia), severe headache, and back pain (Hussein, 2023). Gastrointestinal symptoms, such as nausea, vomiting, diarrhea, and abdominal pain, typically appear on days 3–5, which can lead to significant dehydration and electrolyte imbalance (Martínez et al., 2015). These manifestations reflect systemic spread of the virus through the bloodstream (viremia) and visceral organ involvement (Richards et al., 2025). Entering the advanced stage (days 5–10), clinical symptoms become more characteristic due to endothelial damage and impaired hemostasis (Choi and Croyle, 2013). The virus infects vascular endothelial cells, hepatocytes, and macrophages, leading to vascular leakage, hypovolemia, and impaired blood clotting (Rivera and Messaoudi, 2016). Consequently, some patients exhibit signs of bleeding, such as petechiae, ecchymoses, conjunctival hemorrhage, epistaxis, hematemesis, melena, or bleeding from the injection site (Perry et al., 2012). Although only about 30%–50% of patients experience overt bleeding, this symptom is often an indicator of a poor prognosis (Ji et al., 2016). Liver and kidney dysfunction begin to manifest at this stage, characterized by elevated liver enzyme levels (AST and ALT) and serum creatinine (Warren et al., 2020). In severe cases, encephalopathy can occur due to cerebral hypoperfusion and metabolic disturbances (Rojek et al., 2017). The combination of hypovolemia, coagulation disorders, and multiple organ damage can lead to circulatory shock, leading to multiorgan failure and death, typically occurring between the 7th and 12th day after symptom onset (Rivera and Messaoudi, 2016). Patients who survive typically begin to show clinical improvement between days 10 and 14 (Kadanali and Karagoz, 2015). Signs of recovery include a decrease in fever, normalization of organ function, and return of appetite (Jacob et al., 2020). However, the reconvalescent phase is often accompanied by post-infection complications, such as arthralgia, chronic myalgia, uveitis, conjunctivitis, hearing loss, and general weakness, which can persist for several weeks to months (Mandizadza et al., 2024). Furthermore, Ebolavirus can persist in immunoprivileged locations, such as the eyeball, testes, and central nervous system, for several months (Lyon et al., 2017). This allows for sexual transmission from recovered men, as viral RNA can remain detectable in semen for more than 6 months after recovery (Schindell et al., 2018). This phenomenon has significant implications for outbreak control strategies and public health policy (Paterson et al., 2025). The mortality rate for EVD varies between 25% and 90%, depending on the virus species, health system capacity, and the timing of medical care (Izudi and Bajunirwe, 2024). EBOV is known to be the most virulent and has the highest mortality rate, while SUDV and BDBV have lower fatality rates (Kadanali and Karagoz, 2015). Prognostic factors associated with mortality include high viral load, rapid onset of gastrointestinal symptoms, liver failure, and the absence of an antibody response early in the infection (Yeabah et al., 2025). DiagnosisDiagnosis of EVD requires a comprehensive, multi-step approach, including clinical assessment, epidemiological history, and laboratory confirmation using molecular and immunological methods (Broadhurst et al., 2016). This approach is crucial because early symptoms are nonspecific and often resemble other tropical infections, such as malaria, typhoid fever, or dengue hemorrhagic fever (Martínez et al., 2015). The initial diagnosis of EVD is made by recognizing typical clinical symptoms, such as sudden high fever, severe muscle pain, weakness, nausea, vomiting, diarrhea, and signs of bleeding, along with a history of close contact with a confirmed patient or a wild animal that could potentially act as a reservoir for the virus, such as a bat or a nonhuman primate (Hussein, 2023). A history of travel to an endemic area in Sub-Saharan Africa within the past 21 days is also an important factor in the initial clinical evaluation (Jarrett, 2015). However, because early symptoms are nonspecific, a definitive diagnosis must always be confirmed through safe and standardized laboratory testing (Martínez et al., 2015). Routine hematology tests in EVD patients typically show leukopenia, thrombocytopenia, and an elevated hematocrit, reflecting dehydration due to fluid loss (Koenig et al., 2014). Blood biochemistry may reveal elevated liver enzymes (AST and ALT), hypokalemia, hyponatremia, and elevated urea and creatinine levels, indicating liver and kidney involvement (Kjaldgaard et al., 2022). While these test results are not specific to EVD, they are useful in assessing the severity of the disease and monitoring its progression (Dembek et al., 2024). The primary method for confirming EVD is reverse transcription polymerase chain reaction (RT-PCR) (Coarsey et al., 2017). This technique can detect viral RNA in blood, serum, or other body fluids with high sensitivity and specificity, especially in the acute phase of infection (Ward et al., 2020). RT-PCR can detect the virus from the first day of symptoms and is the gold standard for rapid diagnosis in the field (Muzembo et al., 2022). Additionally, quantitative real-time RT-PCR (qRT-PCR) is used to measure viral load, which is important in assessing patient prognosis (Matson et al., 2022). In situations where molecular facilities are limited, Loop-mediated Isothermal Amplification can be an alternative, because this method does not require complex PCR equipment and is able to provide results quickly (Bettini et al., 2023). Antigen and antibody detection are also used for diagnostic purposes and epidemiological surveillance (Ravichandran and Khurana, 2022). Antigen-based Enzyme-Linked Immunosorbent Assay (ELISA) is used to detect viral proteins, such as GPs and NPs, during the acute phase of infection, while IgM and IgG antibody-based ELISAs are used to assess the immune response after infection (Paweska et al., 2019). IgM antibodies typically appear by the end of the first week of infection, while IgG antibodies are detected after recovery and can persist for years (Broadhurst et al., 2016). In addition, immunohistochemistry techniques are also used to detect viral antigens in postmortem tissue or biopsy samples (Oumarou Hama et al., 2022). Ebolavirus isolation is performed by growing clinical samples, such as blood, serum, or tissue, in cell culture media such as Vero E6 or Huh7 (Logue et al., 2019). While this method is definitively confirmatory, it can only be performed in a biosafety level 4 laboratory due to the high risk to personnel (Broadhurst et al., 2016). Histopathological analysis of tissue from deceased patients revealed diffuse hepatocellular necrosis, necrosis of the spleen and kidneys, and vascular endothelial degeneration, consistent with filovirus infection (Wang et al., 2024). As the need for rapid detection in outbreak areas increases, various point-of-care diagnostic tools have been developed, such as the GeneXpert Ebola assay and the ReEBOV Antigen Rapid Test, which can detect viral antigens in less than an hour with high sensitivity (Wang et al., 2023). Furthermore, next-generation sequencing-based approaches are now being used for phylogenetic analysis and transmission tracking between outbreaks, supporting mapping of viral evolution and epidemic control strategies (Di Paola et al., 2020). TransmissionEVD is a zoonotic disease with a complex transmission pattern, involving interactions between natural reservoirs, intermediary animals, and humans (Hussein, 2023). Transmission occurs through direct contact with the blood, body fluids, or tissues of infected individuals or animals, or through equipment or surfaces contaminated with the virus (Jacob et al., 2020). Although Ebolavirus is not airborne like influenza viruses, it can be transmitted through large droplets containing viral particles from symptomatic patients (Osterholm et al., 2015). The initial transmission of EVD to humans (index cases) generally results from contact with infected wild animals (Rewar and Mirdha, 2014). Epidemiological and molecular evidence suggests that fruit bats of the Pteropodidae family, particularly the species Hypsignathus monstrosus, Epomops franqueti, and Myonycteris torquata, serve as the primary natural reservoir of Ebolavirus (Lacroix et al., 2021). The virus is likely transmitted to humans through consumption of bushmeat, handling carcasses of animals such as nonhuman primates and duikers, or contact with bodily fluids of infected animals (Alexander et al., 2015). Ecologically, human activities such as deforestation and poaching increase the frequency of interactions with the reservoir, thus increasing the risk of zoonotic spillover (Wegner et al., 2022). After primary infection, Ebolavirus can be transmitted efficiently between humans through direct contact with the bodily fluids of symptomatic patients, including blood, saliva, sweat, urine, vomit, feces, breast milk, and genital fluids (Hussein, 2023). The risk of transmission increases especially in the later stages of the disease, when the viral load peaks and hemorrhagic symptoms appear (Vetter et al., 2016). The virus can also survive on inanimate objects (fomites), such as contaminated clothing, syringes, or medical equipment, especially in humid, low-temperature environments (Nikiforuk et al., 2017). Nosocomial transmission is a major factor in the spread of large outbreaks, particularly in healthcare facilities where infection prevention measures are inadequate (Wong and Wong, 2015). Healthcare workers are at high risk of infection if they do not properly use personal protective equipment (PPE), particularly when performing invasive procedures or caring for terminally ill patients (Lyon et al., 2017). Furthermore, traditional burial practices involving direct contact with the remains of Ebola patients have been shown to be a significant source of community transmission, as was the case during the West African outbreak (2014–2016) (Park, 2020). In addition to being transmitted through bodily fluids, evidence suggests that Ebolavirus can persist in immunoprivileged sites such as the testes, eyeballs, and central nervous system for several months after clinical recovery (Berry et al., 2019). This makes sexual transmission from male survivors possible, as viral RNA can be detected in semen for up to 9 months or more after infection (Meek et al., 2025). Molecularly confirmed cases of sexual transmission emphasize the importance of long-term monitoring of individuals who have recovered from EVD (Hewson, 2024). Furthermore, research indicates the possibility of reactivation of latent or persistent infections that could trigger small outbreaks, as occurred in Guinea in 2021, which originated in survivors of the 2014–2016 outbreak (Keita et al., 2021). These findings confirm that individuals who have recovered from EVD can serve as secondary reservoirs of the virus within the human population (Delgado and Simón, 2018). The rate of EVD transmission is influenced by social behavior, community structure, and health system capacity (Chowell and Nishiura, 2014). The basic reproduction number (R0) of EVD is estimated to range from 1.5 to 2.5, meaning that each infected individual can transmit the disease to one to two others if no intervention is undertaken (Muzembo et al., 2024). Factors that increase the risk of transmission include delayed diagnosis, limited isolation facilities, poor adherence to biosafety protocols, and public perception of the health system (Ryan et al., 2022). Furthermore, cross-border population mobility and economic activity between countries in West and Central Africa also play a role in expanding the virus’s geographic distribution (Onyekuru et al., 2023). VaccinationVaccination is a crucial element in EVD prevention and control strategies, given the high mortality rate and the virus’s potential to trigger large, transregional outbreaks (Kuehn et al., 2024). Vaccine development focuses on generating long-term protective immunity by stimulating humoral and cellular immune responses to key viral antigens, particularly surface GPs, which play a role in viral adhesion and entry into host cells (Wang et al., 2017). The GP of Ebolavirus acts as the primary immunogenic antigen and is a key target for vaccine development (Ilinykh et al., 2024). This protein facilitates viral attachment to host cell receptors and triggers an adaptive immune response, including the activation of cytotoxic CD8+ T cells and the production of neutralizing antibodies (Lee and Saphire, 2009). By expressing GP through various vector platforms, vaccines can stimulate an immune response that mimics natural infection without causing disease (Romanyuk et al., 2022). Multiple vaccine platforms have been developed and thoroughly tested through preclinical and clinical trials, with two leading candidates having received regulatory approval. rVSV-ZEBOV vaccine (Ervebo®)This vaccine utilizes recombinant vesicular stomatitis virus (rVSV) modified to express the GP gene from EBOV (Regules et al., 2017). As a live, attenuated vaccine, rVSV-ZEBOV is capable of inducing a rapid and robust immune response with just one dose (Pinski and Messaoudi, 2020). Field trials during the 2015 outbreak in Guinea demonstrated a protective efficacy of 97.5%, making it the first vaccine approved by the WHO and FDA for use in humans, particularly in high-risk groups such as healthcare workers and close contacts of EVD patients (Henao-Restrepo et al., 2015). Ad26.ZEBOV/MVA-BN-Filo (Zabdeno®/Mvabea®) vaccineThis two-dose heterologous vaccine regimen uses adenovirus type 26 (Ad26) as a primary dose and modified vaccinia Ankara as a booster dose (Larivière et al., 2024). This strategy provides prolonged immunity with a good safety profile. In addition to targeting Ebolavirus, the vaccine also expresses antigens from other filoviruses, potentially providing cross-protection against multiple Ebolavirus species (Zhou et al., 2025). In addition to these two vaccines, several other platforms—such as DNA vaccines, mRNA vaccines, and recombinant protein-based nanoparticles—are being developed to improve stability, simplify production processes, and enhance immune effectiveness against various virus variants (Malik et al., 2023). EVD vaccination is implemented based on risk assessment and epidemiological conditions (Kuehn et al., 2024). The most common approach is “ring vaccination,” which involves administering the vaccine to direct contacts of confirmed cases and their contacts, thus establishing an immune barrier around the case (Garcia-Fogeda et al., 2025). This strategy has proven effective in interrupting the chain of transmission during outbreaks in West Africa and the Democratic Republic of the Congo (Henao-Restrepo et al., 2015). In addition, preventive vaccinations are provided to high-risk groups, including medical personnel, laboratory workers, and residents living in endemic areas (Kallay et al., 2024). During acute outbreaks, a reactive vaccination strategy is implemented to limit the spread of the virus in areas with emerging cases (Guttieres et al., 2024). The success of a vaccination program depends largely on the availability of a cold supply chain, the readiness of healthcare workers, and public acceptance (AL-Eitan et al., 2025). Significant challenges in Central and West Africa include low public trust in vaccines, political instability, and limited health infrastructure (Omoleke et al., 2016). ControlEVD control is a complex challenge that requires an integrated approach based on One Health principles, as the disease involves interactions between humans, animals, and the environment (Meseko et al., 2015). Control strategies focus on breaking the chain of transmission, improving early detection, and reducing morbidity and mortality through a combination of medical, social, and behavioral interventions (Mulenga-Cilundika et al., 2022). Key pillars of Ebola outbreak control-early case detection and isolation, protection of health workers, ring vaccination, safe and dignified burial, and community engagement summarized in Figure 3.
Fig. 3. Summary of Ebola outbreak control measures. Control efforts begin with the rapid detection of confirmed cases and strict isolation of infected patients (Onyekuru et al., 2023). Rapid diagnosis using RT-PCR and antigen detection allows for immediate quarantine to prevent further transmission (Broadhurst et al., 2016). All patients with suspected or confirmed EVD must be treated in high-biosafety facilities (minimum BSL-3) that implement strict infection control protocols (Zhang et al., 2017). Medical personnel are required to wear full PPE, including respirators, face shields, double gloves, and fluid-resistant gowns (Weber et al., 2015). Cross-contamination is prevented through proper donning and doffing procedures and routine disinfection with a chlorine-based disinfectant, such as a 0.5% sodium hypochlorite solution (Smither et al., 2016). Community-level control efforts emphasize education about transmission mechanisms, early signs of disease, and preventive measures, including avoiding direct contact with patient fluids or corpses (Jalloh et al., 2017). Risk communication and community empowerment programs have proven effective in increasing compliance with prevention protocols, particularly in areas with strong traditional practices and beliefs (Kusena et al., 2024). Safe and dignified burial practices are crucial, as many previous EVD outbreaks have been exacerbated by traditional burial rituals that involve direct contact with the body (Sikakulya et al., 2021). These procedures include wrapping the body in a fluid-tight bag and ensuring no direct contact with mourners, while respecting local cultural values (Warsame et al., 2023). Because EVD is a zoonotic disease, control of animal reservoirs is crucial (Mirazimi, 2015). Fruit bats have been identified as the primary reservoir, so direct interaction between humans and wildlife should be minimized (Leendertz et al., 2016). Governments in endemic areas are encouraged to prohibit the trade and consumption of bushmeat, particularly from primates and bats (Pooley et al., 2015). Furthermore, ecological surveillance and virus monitoring in wildlife populations are crucial to detect potential spillovers before infections spread to humans (Fauziah et al., 2024). Furthermore, medical and biological waste from EVD patients must be managed according to biosafety and biosecurity protocols (Hewlett et al., 2015). All contaminated materials, including disposable medical devices and protective clothing, must be autoclaved or incinerated at high temperatures to completely inactivate the virus (Elveborg et al., 2022). The EVD outbreak was handled by establishing a multidisciplinary emergency response team involving health authorities, international organizations (such as the WHO and CDC), and humanitarian agencies (Reece et al., 2017). Key strategies in the outbreak response included: active surveillance and contact tracing to identify at-risk individuals and break the chain of transmission; decontamination of exposed areas and management of infectious waste; 21-day quarantine and monitoring of individuals who had contact with patients; and provision of medical logistics and temporary isolation facilities in affected areas (Gupta et al., 2021). An approach that directly involves communities, including the role of traditional and religious leaders, is crucial for building public trust, increasing transparency, and encouraging compliance with quarantine policies and vaccination programs (Dada et al., 2019). Future challenges and prospectsDespite significant progress in the detection, control, and treatment of EVD, numerous scientific and operational challenges remain that hamper efforts to eliminate the disease globally (Singh et al., 2017). One major obstacle is the virus’s long-term persistence in survivors in immunoprivileged tissues, such as the testes and eyes, potentially triggering relapses or re-emergence of outbreaks after disease-free periods (Jacob et al., 2020). This situation emphasizes the importance of developing long-term surveillance strategies for survivors to prevent undetected secondary transmission (Den Boon et al., 2019). Furthermore, limited health infrastructure in endemic areas, particularly in Sub-Saharan Africa, presents significant obstacles to rapid diagnosis, intensive care, and vaccine distribution (Kebenei and Okoth, 2021). Dependence on international assistance emphasizes the importance of strengthening local laboratory capacity, training medical personnel, and engaging communities in epidemiological preparedness (Gunnlaugsson et al., 2019). Additional challenges include social and cultural resistance to public health interventions, such as traditional burial practices and mistrust of vaccines, which can exacerbate disease spread during outbreaks (Manguvo and Mafuvadze, 2015). From a scientific perspective, continued research into the genetic evolution and molecular dynamics of the Ebola virus is crucial for understanding how the virus adapts to human and animal hosts (Ndayambaje et al., 2024). Moving forward, the development of multivalent vaccines and combination therapies capable of providing long-term protection against various Ebolavirus strains will be a key focus. Implementing a One Health approach, strengthening global surveillance systems, and cross-sector collaboration are key factors in building a resilient response to high-risk infectious disease threats like EVD (Choi and Croyle, 2013). ConclusionEVD is a highly virulent zoonotic disease with a significant impact on global public health. Its characteristics, including rapid transmission, high mortality, and potential for re-emergence, emphasize the need for an integrated control strategy based on the One Health principle. Efforts such as early detection, strict isolation, implementation of biosafety protocols, and vaccination have proven effective in limiting the spread of outbreaks. Furthermore, strengthening laboratory capacity, active surveillance, and ongoing research into the pathogenesis and immunology of the virus are crucial elements in preventing future outbreaks. AcknowledgmentsThe author expresses his gratitude for the support and contributions that helped in the preparation of this review article. All writing, literature analysis, and manuscript preparation were carried out independently by the author without external funding. Author’s contributionsMM, LH, and ARK drafted the manuscript. FF, BPP, and AHF revise and edits the manuscripts. LFD, DS, and YAF took part in preparing and critical checking this manuscript. SD, NRP, and EYD edits the references. All authors are major contributors to this work and have approved the final version of the manuscript. Conflict of interestThe authors declare that there is no conflict of interest. FundingAll writing and manuscript preparation activities were independently funded by the author. Data availabilityAll references are open access, so data can be obtained from the online web. ReferencesAl-Eitan, L.N., Almahdawi, D.L., Abu Khiarah, R.A. and Alghamdi, M.A. 2025. Bridging gaps in vaccine access and equity: a middle eastern perspective. Vaccines 13(8), 806. Alexander, K.A., Sanderson, C.E., Marathe, M., Lewis, B.L., Rivers, C.M., Shaman, J., Drake, J.M., Lofgren, E., Dato, V.M., Eisenberg, M.C. and Eubank, S. 2015. What factors might have led to the emergence of Ebola in West Africa? PLos Negl. Trop. Dis. 9(6), 3652. Almeida-Pinto, F., Pinto, R. and Rocha, J. 2024. Navigating the complex landscape of Ebola infection treatment: a review of emerging pharmacological approaches. Infect. Dis. Ther. 13(1), 21–55. Baller, A., Padoveze, M.C., Mirindi, P., Hazim, C.E., Lotemo, J., Pfaffmann, J., Ndiaye, A., Carter, S., Chabrat, M.D., Mangala, S., Banzua, B., Umutoni, C., Niang, N.R., Kabego, L., Ouedraogo, A., Houdjo, B., Mwesha, D., Ousman, K.B., Kolwaite, A., Blaney, D.D., Choi, M.J., Pallawo, R., Legand, A., Park, B., Formenty, P., Montgomery, J.M., Gueye, A.S., Allegranzi, B., Yao, N.K.M. and Fall, I.S. 2022. Ebola virus disease nosocomial infections in the Democratic Republic of the Congo: a descriptive study of cases during the 2018-2020 outbreak. Int. J. Infect. Dis. 115(1), 126–133. Baluma Didier, L., Jude, L.L., Franck, E.I., Wang, H. and Wang, X.L. 2024. Spatial modeling and ecological suitability of Ebola virus disease in Africa. PLos One. 19(10), 311936. Banik, A. and Basu, S. 2025. Drivers and consequences of viral zoonoses: public health and economic perspectives. Zoonotic Dis. 5(4), 32. Beeching, N.J., Fenech, M. and Houlihan, C.F. 2014. Ebola virus disease. BMJ 349(1), 7348. Berry, D.E., Li, A.L., Yeh, S. and Shantha, J.G. 2019. Ocular complications in Ebola virus disease survivors: the importance of continuing care in West Africa. Expert Rev. Ophthalmology 14(3), 179–185. Bettini, A., Lapa, D. and Garbuglia, A.R. 2023. Diagnostics of Ebola virus. Front. Public Health 11(1), 1123024. Bhatia, B., Sonar, S., Khan, S. and Bhattacharya, J. 2024. Pandemic-proofing: intercepting zoonotic spillover events. Pathogens 13(12), 1067. Branda, F., Mahal, A., Maruotti, A., Pierini, M. and Mazzoli, S. 2023. The challenges of open data for future epidemic preparedness: the experience of the 2022 Ebolavirus outbreak in Uganda. Front. Pharmacol. 14(1), 1101894. Breman, J.G., Heymann, D.L., Lloyd, G., Mccormick, J.B., Miatudila, M., Murphy, F.A., Muyembé-Tamfun, J.J., Piot, P., Ruppol, J.F., Sureau, P., Van Der Groen, G. and Johnson, K.M. 2016. Discovery and description of Ebola zaire virus in 1976 and relevance to the West African epidemic during 2013-2016. J. Infect. Dis. 214(suppl 3), S93–S101. Broadhurst, M.J., Brooks, T.J.G. and Pollock, N.R. 2016. Diagnosis of Ebola virus disease: past, present, and future. Clin. Microbiol. Rev. 29(4), 773–793. Cárdenas, W.B., Loo, Y.M., Gale M Jr., Hartman, A.L., Kimberlin, C.R., Martínez-Sobrido, L., Saphire, E.O. and Basler, C.F. 2006. Ebola virus VP35 protein binds double-stranded RNA and inhibits alpha/beta interferon production induced by RIG-I signaling. J. Virol. 80(11), 5168–5178. Caron, A., Bourgarel, M., Cappelle, J., Liégeois, F., De Nys, H.M. and Roger, F. 2018. Ebola virus maintenance: if not (only) bats, what else? Viruses 10(10), 549. Chiappelli, F., Bakhordarian, A., Thames, A.D., Du, A.M., Jan, A.L., Nahcivan, M., Nguyen, M.T., Sama, N., Manfrini, E., Piva, F., Rocha, R.M. and Maida, C.A. 2015. Ebola: translational science considerations. J. Transl. Med. 13(1), 11. Choi, J.H. and Croyle, M.A. 2013. Emerging targets and novel approaches to Ebola virus prophylaxis and treatment. BioDrugs 27(6), 565–583. Chowell, G. and Nishiura, H. 2014. Transmission dynamics and control of Ebola virus disease (EVD): a review. BMC. Med. 12(1), 196. Coarsey, C.T., Esiobu, N., Narayanan, R., Pavlovic, M., Shafiee, H. and Asghar, W. 2017. Strategies in Ebola virus disease (EVD) diagnostics at the point of care. Crit. Rev. Microbiol. 43(6), 779–798. Collier, K.M., Klein, E.K., Sevalie, S., Molleh, B., Kabba, Y., Kargbo, A., Bangura, J., Gbettu, H., Simms, S., O’Leary, C., Drury, S., Schieffelin, J.S., Betancourt, T.S. and Crea, T.M. 2024. Ebola virus disease sensitization: community-driven efforts in Sierra Leone. J. Community Health 49(1), 108–116. Dada, S., Mckay, G., Mateus, A. and Lees, S. 2019. Lessons learned from engaging communities for Ebola vaccine trials in Sierra Leone: reciprocity, relatability, relationships and respect (the four R’s). BMC. Public. Health. 19(1), 1665. De Nys, H.M., Kingebeni, P.M., Keita, A.K., Butel, C., Thaurignac, G., Villabona-Arenas, C.J., Lemarcis, T., Geraerts, M., Vidal, N., Esteban, A., Bourgarel, M., Roger, F., Leendertz, F., Diallo, R., Ndimbo-Kumugo, S.P., Nsio-Mbeta, J., Tagg, N., Koivogui, L., Toure, A., Delaporte, E., Ahuka-Mundeke, S., Tamfum, J.J.M., Mpoudi-Ngole, E., Ayouba, A. and Peeters, M. 2018. Survey of Ebola viruses in frugivorous and insectivorous bats in guinea, cameroon, and the Democratic Republic of the Congo, 2015-2017. Emerg. Infect. Dis. 24(12), 2228–2240. Delgado, R. and Simón, F. 2018. Transmission, human population, and pathogenicity: the Ebola case in point. Microbiol. Spectr. 6(2), doi: 10.1128/microbiolspec.mtbp-0003-2016 Dembek, Z., Hadeed, S., Tigabu, B., Schwartz-Watjen, K., Glass, M., Dressner, M., Frankel, D., Blaney, D., Iii, T.G.E., Chekol, T., Owens, A. and Wu, A. 2024. Ebola virus disease outbreaks: lessons learned from past and facing future challenges. Mil. Med. 189(7–8), e1470–e1478. Den Boon, S., Marston, B.J., Nyenswah, T.G., Jambai, A., Barry, M., Keita, S., Durski, K., Senesie, S.S., Perkins, D., Shah, A., Green, H.H., Hamblion, E.L., Lamunu, M., Gasasira, A., Mahmoud, N.O., Djingarey, M.H., Morgan, O., Crozier, I. and Dye, C. 2019. Ebola virus infection associated with transmission from survivors. Emerg. Infect. Dis. 25(2), 249–255. Dewan, S.M.R. and Khan, S.A. 2025. The silent risk of sudan virus: a review addressing global health vulnerabilities. Health Sci. Rep. 8(11), e71522. Di Paola, N., Sanchez-Lockhart, M., Zeng, X., Kuhn, J.H. and Palacios, G. 2020. Viral genomics in Ebola virus research. Nat. Rev. Microbiol. 18(7), 365–378. Do, T.S. and Lee, Y.S. 2016. Modeling the spread of Ebola. Osong. Public. Health. Res. Perspect. 7(1), 43–48. Donahue, C.J., Kesari, A.S., Thakur, N., Wang, L., Stubbs, S.H., Williams, C.G., Sharma, S.B., Kirby, C.D., Leung, D.W., Aryal, U.K., Basler, C.F., Lacount, D.J. and Davey, R.A. 2025. A protein-proximity screen reveals Ebola virus co-opts the mRNA decapping complex through the scaffold protein EDC4. Nat. Commun. 16(1), 8485. Dyall, J., Hart, B.J., Postnikova, E., Cong, Y., Zhou, H., Gerhardt, D.M., Freeburger, D., Michelotti, J., Honko, A.N., DeWald, L.E., Bennett, R.S., Olinger GG Jr., Jahrling, P.B. and Hensley, L.E. 2017. Interferon-β and interferon-γ are weak inhibitors of Ebola virus in cell-based assays. J. Infect. Dis. 215(9), 1416–1420. Elveborg, S., Monteil, V. and Mirazimi, A. 2022. Methods of inactivation of highly pathogenic viruses for molecular, serology or vaccine development purposes. Pathogens 11(2), 271. Emanuel, J., Marzi, A. and Feldmann, H. 2018. Filoviruses: ecology, molecular biology, and evolution. Adv. Virus Res. 100(1), 189–221. Escudero-Pérez, B., Volchkova, V.A., Dolnik, O., Lawrence, P. and Volchkov, V.E. 2014. Shed GP of Ebola virus triggers immune activation and increased vascular permeability. PLos Pathog. 10(11), e1004509. Esposito, M.M., Turku, S., Lehrfield, L. and Shoman, A. 2023. The impact of human activities on zoonotic infection transmissions. Animals. (Basel). 13(10), 1646. Falasca, L., Agrati, C., Petrosillo, N., Di Caro, A., Capobianchi, M.R., Ippolito, G. and Piacentini, M. 2015. Molecular mechanisms of Ebola virus pathogenesis: focus on cell death. Cell Death Differentiation 22(8), 1250–1259. Fall, I.S. 2019. Ebola virus disease outbreak in Liberia: application of lessons learnt to disease surveillance and control. Pan. Afr. Med. J. 33(Suppl 2), 1. Fang, J., Pietzsch, C., Tsaprailis, G., Crynen, G., Cho, K.F., Ting, A.Y., Bukreyev, A., De La Torre, J.C. and Saphire, E.O. 2022. Functional interactomes of the Ebola virus polymerase identified by proximity proteomics in the context of viral replication. Cell. Rep. 38(12), 110544. Fauziah, I., Nugroho, H.A., Yanthi, N.D., Tiffarent, R. and Saputra, S. 2024. Potential zoonotic spillover at the human-animal interface: a mini-review. Vet. World. 17(2), 289–302. Fitzgerald, F., Awonuga, W., Shah, T. and Youkee, D. 2016. Ebola response in Sierra Leone: the impact on children. J. Infect. 72, S6–S12. Garcia-Fogeda, I., Abrams, S., Vanhee, S., Salloum, M., Ogunjimi, B. and Hens, N. 2025. Within-host mathematical models to study antibody kinetics after the prophylactic Ebola vaccine in the Democratic Republic of the Congo. Vaccine 64(1), 127707. Garry, R.F. 2019. Ebola mysteries and conundrums. J. Infect. Dis. 219(4), 511–513. Ghosh, S., Saha, A., Samanta, S. and Saha, R.P. 2021. Genome structure and genetic diversity in the Ebola virus. Curr. Opin. Pharmacol. 60(1), 83–90. Guetiya Wadoum, R.E., Sevalie, S., Minutolo, A., Clarke, A., Russo, G., Colizzi, V., Mattei, M. and Montesano, C. 2021. The 2018-2020 Ebola outbreak in the democratic republic of congo: a better response had been achieved through inter-state coordination in Africa. Risk Manag. Healthc. Policy 14(1), 4923–4930. Gunnlaugsson, G., Hauksdóttir, I.E., Bygbjerg, I.C. and Pinkowski Tersbøl, B. 2019. ‘Tiny Iceland’ preparing for Ebola in a globalized world. Glob. Health. Action. 12(1), 1597451. Gupta, M., Mahanty, S., Greer, P., Towner, J.S., Shieh, W.J., Zaki, S.R., Ahmed, R. and Rollin, P.E. 2004. Persistent infection with ebola virus under conditions of partial immunity. J. Virol. 78(2), 958–967. Gupta, S., Gupta, N., Yadav, P. and Patil, D. 2021. Ebola virus outbreak preparedness plan for developing Nations: lessons learnt from affected countries. J. Infect. Public. Health. 14(3), 293–305. Guttieres, D., Diepvens, C., Decouttere, C. and Vandaele, N. 2024. Modeling supply and demand dynamics of vaccines against epidemic-prone pathogens: case study of Ebola virus disease. Vaccines 12(1), 24. Hangi, S., Balagizi, F., Biamba, C., Banga, S., Elembwe, H., Cakwira, H., Bisimwa, J., Sironge, L., Bhattacharjee, P. and Akilimali, A. 2024. Crisis of Ebola virus disease in Democratic Republic of the Congo, a call to action. New Microbes New Infect. 60–61(1), 101426. Hasan, S., Ahmad, S.A., Masood, R. and Saeed, S. 2019. Ebola virus: a global public health menace: a narrative review. J. Fam. Med. Prim. Care. 8(7), 2189–2201. Hayman, D.T.S., John, R.S. and Rohani, P. 2022. Transmission models indicate Ebola virus persistence in non-human primate populations is unlikely. J. R. Soc. Interface. 19(187), 20210638. Henao-Restrepo, A.M., Longini, I.M., Egger, M., Dean, N.E., Edmunds, W.J., Camacho, A., Carroll, M.W., Doumbia, M., Draguez, B., Duraffour, S., Enwere, G., Grais, R., Günther, S., Hossmann, S., Kondé, M.K., Kone, S., Kuisma, E., Levine, M.M., Mandal, S., Norheim, G., Riveros, X., Soumah, A., Trelle, S., Vicari, A.S., Watson, C.H., Kéïta, S., Kieny, M.P. and Røttingen, J.A. 2015. Efficacy and effectiveness of an rVSV-vectored vaccine expressing Ebola surface glycoprotein: interim results from the Guinea ring vaccination cluster-randomised trial. Lancet 386(9996), 857–866. Hewlett, A.L., Varkey, J.B., Smith, P.W. and Ribner, B.S. 2015. Ebola virus disease: preparedness and infection control lessons learned from two biocontainment units. Curr. Opin. Infect. Dis. 28(4), 343–348. Hewson, R. 2024. Understanding viral haemorrhagic fevers: virus diversity, vector ecology, and public health strategies. Pathogens 13(10), 909. Hume, A.J. and Mühlberger, E. 2019. Distinct genome replication and transcription strategies within the growing filovirus family. J. Mol. Biol. 431(21), 4290–4320. Husby, M.L., Amiar, S., Prugar, L.I., David, E.A., Plescia, C.B., Huie, K.E., Brannan, J.M., Dye, J.M., Pienaar, E. and Stahelin, R.V. 2022. Phosphatidylserine clustering by the Ebola virus matrix protein is a critical step in viral budding. EMBO. Rep. 23(11), e51709. Hussein, H.A. 2023. Brief review on ebola virus disease and one health approach. Heliyon 9(8), e19036. Ilinykh, P.A., Huang, K., Gunn, B.M., Kuzmina, N.A., Kedarinath, K., Jurado-Cobena, E., Zhou, F., Subramani, C., Hyde, M.A., Velazquez, J.V., Williamson, L.E., Gilchuk, P., Carnahan, R.H., Alter, G., Crowe, J.E. and Bukreyev, A. 2024. Antibodies targeting the glycan cap of Ebola virus glycoprotein are potent inducers of the complement system. Commun. Biol. 7(1), 871. Izudi, J. and Bajunirwe, F. 2024. Case fatality rate for Ebola disease, 1976-2022: a meta-analysis of global data. J. Infect. Public Health 17(1), 25–34. Jacob, S.T., Crozier, I., Fischer, W.A., Hewlett, A., Kraft, C.S., Vega, M.A., Soka, M.J., Wahl, V., Griffiths, A., Bollinger, L. and Kuhn, J.H. 2020. Ebola virus disease. Nat. Rev. Dis. Prim. 6(1), 13. Jain, S., Martynova, E., Rizvanov, A., Khaiboullina, S. and Baranwal, M. 2021. Structural and functional aspects of Ebola virus proteins. Pathogens 10(10), 1330. Jalloh, M.F., Sengeh, P., Monasch, R., Jalloh, M.B., Deluca, N., Dyson, M., Golfa, S., Sakurai, Y., Conteh, L., Sesay, S., Brown, V., Li, W., Mermin, J. and Bunnell, R. 2017. National survey of Ebola-related knowledge, attitudes and practices before the outbreak peak in Sierra Leone: august 2014. BMJ. Glob. Health. 2(4), 285. Jarrett, A. 2015. Ebola: a practice summary for nurse practitioners. J. Nurse. Pract. 11(1), 16–26. Ji, Y.J., Duan, X.Z., Gao, X.D., Li, L., Li, C., Ji, D., Li, W.G., Wang, L.F., Meng, Y.H., Yang, X., Ling, B.F., Song, X.A., Gu, M.L., Jiang, T., Koroma, S.K.M., Bangalie, J. and Duan, H.J. 2016. Clinical presentations and outcomes of patients with Ebola virus disease in Freetown, Sierra Leone. Infect. Dis. Poverty 5(1), 101. Judson, S., Prescott, J. and Munster, V. 2015. Understanding ebola virus transmission. Viruses 7(2), 511–521. Judson, S.D. and Munster, V.J. 2023. The multiple origins of ebola disease outbreaks. J. Infect. Dis. 228(Suppl 7), S465–S473. Judson, S.D., Fischer, R., Judson, A. and Munster, V.J. 2016. Ecological contexts of index cases and spillover events of different ebolaviruses. PLos Pathog. 12(8), e1005780. Jun, S.R., Leuze, M.R., Nookaew, I., Uberbacher, E.C., Land, M., Zhang, Q., Wanchai, V., Chai, J., Nielsen, M., Trolle, T., Lund, O., Buzard, G.S., Pedersen, T.D., Wassenaar, T.M. and Ussery, D.W. 2015. Ebolavirus comparative genomics. FEMS Microbiol. Rev. 39(5), 764–778. Kabami, Z., Ario, A.R., Harris, J.R., Ninsiima, M., Ahirirwe, S.R., Ocero, J.R.A., Atwine, D., Mwebesa, H.G., Kyabayinze, D.J., Muruta, A.N., Kagirita, A., Tegegn, Y., Nanyunja, M., Kizito, S.N., Kadobera, D., Kwesiga, B., Gidudu, S., Migisha, R., Makumbi, I., Eurien, D., Elyanu, P.J., Ndyabakira, A., Naiga, H.N., Zalwango, J.F., Agaba, B., Kawungezi, P.C., Zalwango, M.G., King, P., Simbwa, B.N., Akunzirwe, R., Wanyana, M.W., Zavuga, R. and Kiggundu, T. 2024. Ebola disease outbreak caused by the Sudan virus in Uganda, 2022: a descriptive epidemiological study. Lancet Global Health 12(10), e1684–e1692. Kadanali, A. and Karagoz, G. 2015. An overview of Ebola virus disease. North. Clin. Istanb. 2(1), 81–86. Kallay, R., Doshi, R.H., Muhoza, P., Choi, M.J., Legand, A., Aberle-Grasse, E., Bagayoko, A., Hyde, T.B., Formenty, P. and Costa, A. 2024. Use of Ebola vaccines - worldwide, 2021-2023. MMWR. Morb. Mortal. Wkly. Rep. 73(16), 360–364. Kebenei, C.K. and Okoth, P. 2021. Ebola virus disease, diagnostics and therapeutics: where is the consensus in over three decades of clinical research? Sci. Afr. 13(1), 862. Keita, A.K., Koundouno, F.R., Faye, M., Düx, A., Hinzmann, J., Diallo, H., Ayouba, A., Le Marcis, F., Soropogui, B., Ifono, K., Diagne, M.M., Sow, M.S., Bore, J.A., Calvignac-Spencer, S., Vidal, N., Camara, J., Keita, M.B., Renevey, A., Diallo, A., Soumah, A.K., Millimono, S.L., Mari-Saez, A., Diop, M., Doré, A., Soumah, F.Y., Kourouma, K., Vielle, N.J., Loucoubar, C., Camara, I., Kourouma, K., Annibaldis, G., Bah, A., Thielebein, A., Pahlmann, M., Pullan, S.T., Carroll, M.W., Quick, J., Formenty, P., Legand, A., Pietro, K., Wiley, M.R., Tordo, N., Peyrefitte, C., Mccrone, J.T., Rambaut, A., Sidibé, Y., Barry, M.D., Kourouma, M., Saouromou, C.D., Condé, M., Baldé, M., Povogui, M., Keita, S., Diakite, M., Bah, M.S., Sidibe, A., Diakite, D., Sako, F.B., Traore, F.A., Ki-Zerbo, G.A., Lemey, P., Günther, S., Kafetzopoulou, L.E., Sall, A.A., Delaporte, E., Duraffour, S., Faye, O., Leendertz, F.H., Peeters, M., Toure, A. and Magassouba, N.F. 2021. Resurgence of Ebola virus in 2021 in Guinea suggests a new paradigm for outbreaks. Nature 597(7877), 539–543. Kjaldgaard, L., Claude, K.M., Mukadi-Bamuleka, D., Kitenge-Omasumbu, R., Dixit, D., Edidi-Atani, F., Kuamfumu, M.M., Bulabula-Penge, J., Mambu-Mbika, F., Tshiani-Mbaya, O., Diaz, J., Mulangu, S., Legand, A., Mbala-Kingebeni, P., Formenty, P., Ahuka-Mundeke, S., Muyembe-Tamfum, J.J. and Hawkes, M.T. 2022. Virus kinetics and biochemical derangements among children with Ebolavirus disease. EClinicalMedicine 53(1), 101638. Koenig, K., Majestic, C. and Burns, M. 2014. Ebola virus disease: essential public health principles for clinicians. West. J. Emerg. Med. 15(7), 728–731. Kuehn, R., Ryan, H., Okwaraeke, K.C., Gould, S., Chaplin, M., Riley, M., Turtle, L., Jacob, S.T. and Fletcher, T. 2024. Vaccines for preventing Ebola virus disease. Cochrane Database Syst. Rev. 11(11), CD015828. Kühl, A. and Pöhlmann, S. 2012. How Ebola virus counters the interferon system. Zoonoses. Public. Health. 59(Suppl 2), 116–131. Kuhn, J.H., Becker, S., Ebihara, H., Geisbert, T.W., Johnson, K.M., Kawaoka, Y., Lipkin, W.I., Negredo, A.I., Netesov, S.V., Nichol, S.T., Palacios, G., Peters, C.J., Tenorio, A., Volchkov, V.E. and Jahrling, P.B. 2010. Proposal for a revised taxonomy of the family Filoviridae: classification, names of taxa and viruses, and virus abbreviations. Archiv. Für. Virologie. 155(12), 2083–2103. Kumar, A. 2016. Ebola virus altered innate and adaptive immune response signalling pathways: implications for novel therapeutic approaches. Infect. Disord. Drug Targets 16(2), 79–94. Kuroda, M., Fujikura, D., Nanbo, A., Marzi, A., Noyori, O., Kajihara, M., Maruyama, J., Matsuno, K., Miyamoto, H., Yoshida, R., Feldmann, H. and Takada, A. 2015. Interaction between TIM-1 and NPC1 is important for cellular entry of Ebola virus. J. Virol. 89(12), 6481–6493. Kusena, P., Metta, E., Mohamed, H., Kakoko, D., Nyamhanga, T., Bahuguna, S., Sirili, N., Kinanda, J., Araya, A., Mwiru, A., Magesa, S., Makene, L., Rwechungura, A., Kirakoya, F.B., Rangi, J., Kileo, N., Mlembwa, J., Kazaura, M., Mpelembe, C. and Frumence, G. 2024. “If I die for touching him, let me die”: a rapid ethnographic assessment of cultural practices and Ebola transmission in high-risk border regions of Tanzania. BMC Public Health 24(1), 1838. Lacroix, A., Mbala Kingebeni, P., Ndimbo Kumugo, S.P., Lempu, G., Butel, C., Serrano, L., Vidal, N., Thaurignac, G., Esteban, A., Mukadi Bamuleka, D., Likofata, J., Delaporte, E., Muyembe Tamfum, J.J., Ayouba, A., Peeters, M. and Ahuka Mundeke, S. 2021. Investigating the circulation of Ebola viruses in bats during the Ebola virus disease outbreaks in the Equateur and North Kivu Provinces of the Democratic Republic of Congo from 2018. Pathogens 10(5), 557. Lai, K.Y., Ng, W.Y.G. and Cheng, F.F. 2014. Human Ebola virus infection in West Africa: a review of available therapeutic agents that target different steps of the life cycle of Ebola virus. Infect. Dis. Poverty 3(1), 43. Larivière, Y., Garcia-Fogeda, I., Zola Matuvanga, T., Isekah Osang’ir, B., Milolo, S., Meta, R., Kimbulu, P., Robinson, C., Katwere, M., McLean, C., Hens, N., Matangila, J., Maketa, V., Mitashi, P., Muhindo-Mavoko, H., Van Geertruyden, J.P. and Van Damme, P. 2024. Safety and immunogenicity of the heterologous 2-dose Ad26.ZEBOV, MVA-BN-Filo vaccine regimen in health care providers and frontliners of the Democratic Republic of the Congo. J. Infect. Dis. 229(4), 1068–1076. LaVergne, S.M., Sakabe, S., Kanneh, L., Momoh, M., Al-Hassan, F., Yilah, M., Goba, A., Sandi, J.D., Gbakie, M., Cubitt, B., Boisen, M., Mayeux, J.M., Smira, A., Shore, K., Bica, I., Pollard, K.M., De La Torre, J.C., Branco, L.M., Garry, R.F., Grant, D.S., Schieffelin, J.S., Oldstone, M.B.A. and Sullivan, B.M. 2020. Ebola-specific CD8+ and CD4+ T-Cell responses in Sierra Leonean Ebola virus survivors with or without post-Ebola sequelae. J. Infect. Dis. 222(9), 1488–1497. Lee, J.E. and Saphire, E.O. 2009. Ebolavirus glycoprotein structure and mechanism of entry. Future. Virol. 4(6), 621–635. Lee-Cruz, L., Lenormand, M., Cappelle, J., Caron, A., De Nys, H., Peeters, M., Bourgarel, M., Roger, F. and Tran, A. 2021. Mapping of Ebola virus spillover: suitability and seasonal variability at the landscape scale. PLos Negl. Trop. Dis. 15(8), 9683. Leendertz, S.A.J., Gogarten, J.F., Düx, A., Calvignac-Spencer, S. and Leendertz, F.H. 2016. Assessing the evidence supporting fruit bats as the primary reservoirs for Ebola viruses. Ecohealth 13(1), 18–25. Leung, L.W., Park, M.S., Martinez, O., Valmas, C., López, C.B. and Basler, C.F. 2011. Ebolavirus VP35 suppresses IFN production from conventional but not plasmacytoid dendritic cells. Immunol. Cell Biol. 89(7), 792–802. Licata, J.M., Johnson, R.F., Han, Z. and Harty, R.N. 2004. Contribution of ebola virus glycoprotein, nucleoprotein, and VP24 to budding of VP40 virus-like particles. J. Virol. 78(14), 7344–7351. Locke, M., Lythe, G., López-García, M., Muñoz-Fontela, C., Carroll, M. and Molina-París, C. 2021. Quantification of type I interferon inhibition by viral proteins: Ebola virus as a case study. Viruses 13(12), 2441. Logue, J., Licona, W.V., Cooper, T.K., Reeder, B., Byrum, R., Qin, J., Murphy, N.D., Cong, Y., Bonilla, A., Sword, J., Weaver, W., Kocher, G., Olinger, G.G., Jahrling, P.B., Hensley, L.E. and Bennett, R.S. 2019. Ebola virus isolation using Huh-7 cells has methodological advantages and similar sensitivity to isolation using other cell types and sucking BALB/c Laboratory Mice. Viruses 11(2), 161. Longet, S., Mellors, J., Carroll, M.W. and Tipton, T. 2021. Ebolavirus: comparison of survivor immunology and animal models in the search for a correlate of protection. Front. Immunol. 11(1), 599568. Lubaki, N.M., Younan, P., Santos, R.I., Meyer, M., Iampietro, M., Koup, R.A. and Bukreyev, A. 2016. The Ebola interferon inhibiting domains attenuate and dysregulate cell-mediated immune responses. PLos Pathog. 12(12), e1006031. Lyon, G.M., Mehta, A.K. and Ribner, B.S. 2017. Clinical management of patients with ebola virus disease in high-resource settings. Curr. Top. Microbiol. Immunol. 411(1), 115–137. Mahanty, S., Gupta, M., Paragas, J., Bray, M., Ahmed, R. and Rollin, P.E. 2003. Protection from lethal infection is determined by innate immune responses in a mouse model of Ebola virus infection. Virology 312(2), 415–424. Malik, S., Kishore, S., Nag, S., Dhasmana, A., Preetam, S., Mitra, O., León-Figueroa, D.A., Mohanty, A., Chattu, V.K., Assefi, M., Padhi, B.K. and Sah, R. 2023. Ebola virus disease vaccines: development, current perspectives & challenges. Vaccines. (Basel). 11(2), 268. Mandizadza, O.O., Phebeni, R.T. and Ji, C. 2024. Prevalence of somatic symptoms among Ebola virus disease (EVD) survivors in Africa: a systematic review and meta-analysis. BMC. Public. Health. 24(1), 1511. Manguvo, A. and Mafuvadze, B. 2015. The impact of traditional and religious practices on the spread of Ebola in West Africa: time for a strategic shift. Pan Afr. Med. J. 22(Suppl 1), 9. Mariappan, V., Pratheesh, P., Shanmugam, L., Rao, S.R. and Pillai, A.B. 2021. Viral hemorrhagic fever: molecular pathogenesis and current trends of disease management-an update. Curr. Res. Virol. Sci. 2(1), 100009. Martínez, M.J., Salim, A.M., Hurtado, J.C. and Kilgore, P.E. 2015. Ebola virus infection: overview and update on prevention and treatment. Infect. Dis. Ther. 4(4), 365–390. Marzi, A. and Feldmann, H. 2014. Ebola virus vaccines: an overview of current approaches. Expert Rev. Vaccines 13(4), 521–531. Matson, M.J., Chertow, D.S. and Munster, V.J. 2025. Ebola virus disease: uniquely challenging among the viral hemorrhagic fevers. Open Forum Infect. Dis. 12(8), 464. Matson, M.J., Ricotta, E., Feldmann, F., Massaquoi, M., Sprecher, A., Giuliani, R., Edwards, J.K., Rosenke, K., De Wit, E., Feldmann, H., Chertow, D.S. and Munster, V.J. 2022. Evaluation of viral load in patients with Ebola virus disease in Liberia: a retrospective observational study. Lancet. Microbe. 3(7), e533–e542. Mclean, C., Dijkman, K., Gaddah, A., Keshinro, B., Katwere, M., Douoguih, M., Robinson, C., Solforosi, L., Czapska-Casey, D., Dekking, L., Wollmann, Y., Volkmann, A., Pau, M.G., Callendret, B., Sadoff, J., Schuitemaker, H., Zahn, R., Luhn, K., Hendriks, J. and Roozendaal, R. 2023. Persistence of immunological memory as a potential correlate of long-term, vaccine-induced protection against Ebola virus disease in humans. Front. Immunol. 14(1), 1215302. Meek, O., Fornace, K. and Carroll, M.W. 2025. Ebola virus persistence: implications for human-to-human transmission and new outbreaks. Explor. Med. 6(1), 1001333. Mendez-Rodriguez, L.C. 2015. Transmission of the Ebola virus in the wild. Therya 6(3), 515–518. Meseko, C.A., Egbetade, A.O. and Fagbo, S. 2015. Ebola virus disease control in West Africa: an ecological, one health approach. Pan Afr. Med. J. 21(1), 6. Meurer, I. 2025. The importance of medical knowledge about Q fever in the context of timely diagnosis and treatment and the use of the one health approach in combating this and other neglected zoonotic diseases letter. Infect. Drug. Resist. 18, 5007–5008. Milambo, J.P.M. and Businge, C.B. 2025. Clinical and occupational predictors of mortality in Ebola virus disease: a commentary from the Democratic Republic of Congo (2018–2020). Infect. Dis. Rep. 17(3), 71. Mirazimi, A. 2015. Ebola virus disease: societal challenges and new treatments. J. Intern. Med. 278(3), 227–237. Misasi, J. and Sullivan, N.J. 2014. Camouflage and misdirection: the full-on assault of ebola virus disease. Cell 159(3), 477–486. Mulenga-Cilundika, P., Ekofo, J., Kabanga, C., Criel, B., Van Damme, W. and Chenge, F. 2022. Indirect effects of Ebola virus disease epidemics on health systems in the Democratic Republic of the Congo, Guinea, Sierra Leone and Liberia: a scoping review supplemented with expert interviews. Int. J. Environ. Res. Public Health 19(20), 13113. Muñoz-Fontela, C. and McElroy, A.K. 2017. Ebola virus disease in humans: pathophysiology and immunity. Curr. Top. Microbiol. Immunol. 411(1), 141–169. Muzembo, B.A., Kitahara, K., Mitra, D., Ntontolo, N.P., Ngatu, N.R., Ohno, A., Khatiwada, J., Dutta, S. and Miyoshi, S.I. 2024. The basic reproduction number (R0) of ebola virus disease: a systematic review and meta-analysis. Travel Med. Infect. Dis. 57(1), 102685. Muzembo, B.A., Kitahara, K., Ohno, A., Ntontolo, N., Ngatu, N., Okamoto, K. and Miyoshi, S.I. 2022. Rapid diagnostic tests versus RT-PCR for Ebola virus infections: a systematic review and meta-analysis. Bull. World. Health. Organ. 100(7), 447–458. Ndayambaje, M., Wahnou, H., Naya, A. and Oudghiri, M. 2025. Therapeutic management of Ebola virus: targeting oxidative stress and inflammatory pathways. BioChem 5(1), 3. Ndayambaje, M., Yadufashije, C., Habyarimana, T., Niyonsaba, T., Wahnou, H., Iradukunda, P.G., Izere, C., Uwishema, O., Ndishimye, P. and Oudghiri, M. 2024. Molecular characterization of Ebola virus, immune response, and therapeutic challenges: a narrative review. Egypt. J. Med. Hum. Genet. 25(1), 133. Nikiforuk, A.M., Cutts, T.A., Theriault, S.S. and Cook, B.W.M. 2017. Challenge of liquid stressed protective materials and environmental persistence of Ebola virus. Sci. Rep. 7(1), 4388. Obeagu, E.I. and Ezeala, C.C. 2025. Guardians of immunity: neutrophils and their role in Ebola virus infection. Ann. Med. Surg. (Lond). 87(11), 7369–7380. Olivero, J., Fa, J.E., Farfán, M.A., Márquez, A.L., Real, R., Juste, F.J., Leendertz, S.A. and Nasi, R. 2020. Human activities link fruit bat presence to Ebola virus disease outbreaks. Mammal. Rev. 50(1), 1–10. Olukitibi, T.A., Ao, Z., Mahmoudi, M., Kobinger, G.A. and Yao, X. 2019. Dendritic cells/macrophages-targeting feature of Ebola glycoprotein and its potential as immunological facilitator for antiviral vaccine approach. Microorganisms 7(10), 402. Omoleke, S.A., Mohammed, I. and Saidu, Y. 2016. Ebola viral disease in West Africa: a threat to global health, economy and political stability. J. Public. Health. Afr. 7(1), 534. Onyekuru, N.A., Ihemezie, E.J., Ezea, C.P., Apeh, C.C. and Onyekuru, B.O. 2023. Impacts of Ebola disease outbreak in West Africa: implications for government and public health preparedness and lessons from COVID-19. Sci. Afr. 19(1), e01513. Osterholm, M.T., Moore, K.A., Kelley, N.S., Brosseau, L.M., Wong, G., Murphy, F.A., Peters, C.J., LeDuc, J.W., Russell, P.K., Van Herp, M., Kapetshi, J., Muyembe, J.J., Ilunga, B.K., Strong, J.E., Grolla, A., Wolz, A., Kargbo, B., Kargbo, D.K., Sanders, D.A. and Kobinger, G.P. 2015. Transmission of Ebola viruses: what we know and what we do not know. mBio 6(2), 137. Oumarou Hama, H., Aboudharam, G., Barbieri, R., Lepidi, H. and Drancourt, M. 2022. Immunohistochemical diagnosis of human infectious diseases: a review. Diagn. Pathol. 17(1), 17. Pan, Y., Zhang, W., Cui, L., Hua, X., Wang, M. and Zeng, Q. 2014. Reston virus in domestic pigs in China. Arch. Virol. 159(5), 1129–1132. Pare, B.C., Camara, A.M., Camara, A., Kourouma, M., Enogo, K., Camara, M.S., Akilimali, L., Sani, S., De Sainte Fare, E.B., Lame, P., Mouly, N., Castro-Rial, M.L., Sivahera, B., Cherif, M.S., Beavogui, A.H., Muamba, D., Tamba, J.B., Moumié, B., Kojan, R. and Lang, H.J. 2023. Ebola outbreak in Guinea, 2021: clinical care of patients with Ebola virus disease. S. Afr. J. Infect. Dis. 38(1), 454. Park, C. 2020. Traditional funeral and burial rituals and Ebola outbreaks in West Africa: a narrative review of causes and strategy interventions. J. Health. Soc. Sci. 5(1), 73–90. Paterson, A., Kabajaasi, O., Adlard, F., Dimitrios, K.H., Cheyne, A., Ssewankambo, Y., Kaggwa, D., Olliaro, P., Kenya-Mugisha, N. and Rojek, A. 2025. Ebola stigma and its impact on outbreak control: lessons from key informant interviews in Central Uganda. Trop. Med. Int. Health 30(9), 1006–1017. Paweska, J.T., Moolla, N., Storm, N., Msimang, V., Conteh, O., Weyer, J. and Vuren, P.J.V. 2019. Evaluation of diagnostic performance of three indirect enzyme-linked immunosorbent assays for the detection of IgG Antibodies to Ebola virus in human sera. Viruses 11(8), 678. Peñas, J.A., Miranda, M.E., De Los Reyes, V.C., Magpantay, R. and Sucaldito, M.N. 2019. Risk assessment of Ebola Reston virus in humans in the Philippines. Western Pac. Surveill. Response J. 10(3), 1–8. Perez-Valencia, L.J., Vannella, K.M., Ramos-Benitez, M.J., Sun, J., Abu-Asab, M., Dorward, D.W., Awad, K.S., Platt, A., Jacobson, E., Kindrachuk, J. and Chertow, D.S. 2023. Ebola virus shed glycoprotein is toxic to human T, B, and natural killer lymphocytes. iScience 26(8), 107323. Perry, D.L., Bollinger, L. and L.White, G. 2012. The baboon (Papio spp.) as a model of human Ebola virus infection. Viruses 4(10), 2400–2416. Pigott, D.M., Golding, N., Mylne, A., Huang, Z., Henry, A.J., Weiss, D.J., Brady, O.J., Kraemer, M.U., Smith, D.L., Moyes, C.L., Bhatt, S., Gething, P.W., Horby, P.W., Bogoch, I.I., Brownstein, J.S., Mekaru, S.R., Tatem, A.J., Khan, K. and Hay, S.I. 2014. Mapping the zoonotic niche of Ebola virus disease in Africa. eLife 3(1), e04395. Pinski, A.N. and Messaoudi, I. 2020. To B or Not to B: mechanisms of protection conferred by rVSV-EBOV-GP and the roles of innate and adaptive immunity. Microorganisms 8(10), 1473. Pooley, S., Fa, J.E. and Nasi, R. 2015. No conservation silver lining to Ebola. Conserv. Biol. 29(3), 965–967. Raab, M., Roth, E., Nguyen, V.K. and Froeschl, G. 2021. The 2021 Ebola virus outbreak in Guinea: mistrust and the shortcomings of outbreak surveillance. PLos Negl. Trop. Dis. 15(6), 9487. Rai, A., Hamiidah, N., Abbass, M., Masunga, D.S., Shoumar, H., Kantawala, B.I., Wellington, J. and Uwishema, O. 2022. Ebola Virus Disease in Uganda: a global emergency call. Ann. Med. Surg. (Lond). 84(1), 104825. Ramirez-Plascencia, H.H.F., Colima-Fausto, A.G., Licona-Lasteros, K.C., Díaz-Zaragoza, M., Cazarez-Navarro, G., Macias-Barragan, J.G. and Rodriguez-Preciado, S.Y. 2025. Presence of microorganisms in the environment: one health approach. Microorganisms 13(11), 2435. Ravichandran, S. and Khurana, S. 2022. Ebola-detect: a differential serodiagnostic assay for Ebola virus infections and surveillance in the presence of vaccine-induced antibodies. EBioMedicine 82(1), 104186. Redding, D.W., Atkinson, P.M., Cunningham, A.A., Lo Iacono, G., Moses, L.M., Wood, J.L.N. and Jones, K.E. 2019. Impacts of environmental and socio-economic factors on emergence and epidemic potential of Ebola in Africa. Nat. Commun. 10(1), 4531. Reece, S., Brown, C.S., Dunning, J., Chand, M.A., Zambon, M.C. and Jacobs, M. 2017. The UK’s multidisciplinary response to an Ebola epidemic. Clin. Med. (Lond). 17(4), 332–337. Regules, J.A., Beigel, J.H., Paolino, K.M., Voell, J., Castellano, A.R., Hu, Z., Muñoz, P., Moon, J.E., Ruck, R.C., Bennett, J.W., Twomey, P.S., Gutiérrez, R.L., Remich, S.A., Hack, H.R., Wisniewski, M.L., Josleyn, M.D., Kwilas, S.A., Van Deusen, N., Mbaya, O.T., Zhou, Y., Stanley, D.A., Jing, W., Smith, K.S., Shi, M., Ledgerwood, J.E., Graham, B.S., Sullivan, N.J., Jagodzinski, L.L., Peel, S.A., Alimonti, J.B., Hooper, J.W., Silvera, P.M., Martin, B.K., Monath, T.P., Ramsey, W.J., Link, C.J., Lane, H.C., Michael, N.L., Davey RT Jr., Thomas, S.J. and Group, R.Z.G.S. 2017. A recombinant vesicular stomatitis virus Ebola vaccine. N. Engl. J. Med. 376(4), 330–341. Rewar, S. and Mirdha, D. 2014. Transmission of ebola virus disease: an overview. Ann. Glob. Health 80(6), 444–451. Richards, P.T., Fleck, A.M., Patel, R., Fakhimi, M., Bohan, D., Geoghegan-Barek, K., Honko, A.N., Stolte, A.E., Plescia, C.B., Messingham, C.O., Connell, S.J., Crowe, T.P., Gourronc, F.A., Carrion R Jr., Griffiths, A., Meyerholz, D.K., Klingelhutz, A.J., Davey, R.A., Messingham, K.N. and Maury, W. 2025. Ebola virus’s hidden target: virus transmission to and infection of skin. J. Virol. 99(10), 130025. Rivera, A. and Messaoudi, I. 2016. Molecular mechanisms of Ebola pathogenesis. J. Leukoc. Biol. 100(5), 889–904. Rodriguez, J. 2024. One health ethics and the ethics of zoonoses: a silent call for global action. Vet. Sci. 11(9), 394. Rojas, M., Monsalve, D.M., Pacheco, Y., Acosta-Ampudia, Y., Ramírez-Santana, C., Ansari, A.A., Gershwin, M.E. and Anaya, J.M. 2020. Ebola virus disease: an emerging and re-emerging viral threat. J. Autoimmun. 106(1), 102375. Rojek, A., Horby, P. and Dunning, J. 2017. Insights from clinical research completed during the west Africa Ebola virus disease epidemic. Lancet Infect. Dis. 17(9), e280–e292. Romanyuk, A., Wang, R., Marin, A., Janus, B.M., Felner, E.I., Xia, D., Goez-Gazi, Y., Alfson, K.J., Yunus, A.S., Toth, E.A., Ofek, G., Carrion, R., Prausnitz, M.R., Fuerst, T.R. and Andrianov, A.K. 2022. Skin vaccination with Ebola virus glycoprotein using a polyphosphazene-based microneedle patch protects mice against lethal challenge. J. Funct. Biomater. 14(1), 16. Ryan, C.S., Belizaire, M.R.D., Nanyunja, M., Olu, O.O., Ahmed, Y.A., Latt, A., Kol, M.T., Bamuleke, B., Tusiime, J., Nsabimbona, N., Conteh, I., Nyashanu, S., Ramadan, P.O., Woldetsadik, S.F., Nkata, J.P.M., Ntwari, J.T., Nzeyimana, S.D., Ouedraogo, L., Batona, G., Ndahindwa, V., Mgamb, E.A., Armah, M., Wamala, J.F., Guyo, A.G., Freeman, A.Y.S., Chimbaru, A., Komakech, I., Kuku, M., Firmino, W.M., Saguti, G.E., Msemwa, F., O-Tipo, S., Kalubula, P.C., Nsenga, N. and Talisuna, A.O. 2022. Sustainable strategies for Ebola virus disease outbreak preparedness in Africa: a case study on lessons learnt in countries neighbouring the Democratic Republic of the Congo. Infect. Dis. Poverty. 11(1), 118. Schindell, B.G., Webb, A.L. and Kindrachuk, J. 2018. Persistence and sexual transmission of filoviruses. Viruses 10(12), 683. Schmidt, J.P., Maher, S., Drake, J.M., Huang, T., Farrell, M.J. and Han, B.A. 2019. Ecological indicators of mammal exposure to Ebolavirus. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 374(1782), 20180337. Shen, L., Song, J., Zhou, Y., Yuan, X., Seery, S., Fu, T., Liu, X., Liu, Y., Shao, Z., Li, R. and Liu, K. 2025. Assessing the ecological resilience of Ebola virus in Africa and potential influencing factors based on a synthesized model. PLos Negl. Trop. Dis. 19(2), 12843. Shu, T., Gan, T., Bai, P., Wang, X., Qian, Q., Zhou, H., Cheng, Q., Qiu, Y., Yin, L., Zhong, J. and Zhou, X. 2019. Ebola virus VP35 has novel NTPase and helicase-like activities. Nucleic Acids Res. 47(11), 5837–5851. Sikakulya, F.K., Ilumbulumbu, M.K., Djuma, S.F., Bunduki, G.K., Sivulyamwenge, A.K. and Jones, M.K. 2021. Safe and dignified burial of a deceased from a highly contagious infectious disease ebolavirus: socio-cultural and anthropological implications in the Eastern DR Congo. One Health 13(1), 100309. Singh, K., Marasini, B., Chen, X. and Spearman, P. 2018. A novel Ebola virus antibody-dependent cell-mediated cytotoxicity (Ebola ADCC) assay. J. Immunol. Methods. 460(1), 10–16. Singh, R.K., Dhama, K., Malik, Y.S., Ramakrishnan, M.A., Karthik, K., Khandia, R., Tiwari, R., Munjal, A., Saminathan, M., Sachan, S., Desingu, P.A., Kattoor, J.J., Iqbal, H.M.N. and Joshi, S.K. 2017. Ebola virus - epidemiology, diagnosis, and control: threat to humans, lessons learnt, and preparedness plans - an update on its 40 year’s journey. Vet. Q. 37(1), 98–135. Sivanandy, P., Jun, P.H., Man, L.W., Wei, N.S., Mun, N.F.K., Yii, C.A.J. and Ying, C.C.X. 2022. A systematic review of Ebola virus disease outbreaks and an analysis of the efficacy and safety of newer drugs approved for the treatment of Ebola virus disease by the US Food and Drug Administration from 2016 to 2020. J. Infect. Public. Health. 15(3), 285–292. Smither, S., Phelps, A., Eastaugh, L., Ngugi, S., O’Brien, L., Dutch, A. and Lever, M. 2016. Effectiveness of four disinfectants against ebola virus on different materials. Viruses 8(7), 185. Stein, R.A. 2015. What is Ebola?. Int. J. Clin. Pract. 69(1), 49–58. Stephens, M.T., Juniastuti, Sulistiawati. and Dossen, P.C. 2024. The potential risk components and prevention measures of the Ebola virus disease outbreak in Liberia: an in-depth interview with the health workers and stakeholders. Belitung Nurs. J. 10(1), 67–77. Thompson, M.R., Kaminski, J.J., Kurt-Jones, E.A. and Fitzgerald, K.A. 2011. Pattern recognition receptors and the innate immune response to viral infection. Viruses 3(6), 920–940. Vetter, P., Fischer, W.A., Schibler, M., Jacobs, M., Bausch, D.G. and Kaiser, L. 2016. Ebola virus shedding and transmission: review of current evidence. J. Infect. Dis. 214(suppl 3), S177–S184. Vine, V., Scott, D.P. and Feldmann, H. 2017. Ebolavirus: an overview of molecular and clinical pathogenesis. Methods. Mol. Biol. 1628(1), 39–50. Wahl-Jensen, V.M., Afanasieva, T.A., Seebach, J., Ströher, U., Feldmann, H. and Schnittler, H.J. 2006. Effects of Ebola virus glycoproteins on endothelial cell activation and barrier function. J. Virol. 79(16), 10442–11050. Wang, H., Shi, Y., Song, J., Qi, J., Lu, G., Yan, J. and Gao, G.F. 2016. Ebola viral glycoprotein bound to its endosomal receptor niemann-pick C1. Cell 164(1–2), 258–268. Wang, S., Li, W., Wang, Z., Yang, W., Li, E., Xia, X., Yan, F. and Chiu, S. 2024. Emerging and reemerging infectious diseases: global trends and new strategies for their prevention and control. Signal Transduct. Target. Ther. 9(1), 223. Wang, Y., Li, J., Hu, Y., Liang, Q., Wei, M. and Zhu, F. 2017. Ebola vaccines in clinical trial: the promising candidates. Hum. Vaccin. Immunother. 13(1), 153–168. Wang, Z., Bennett, R.S., Roehler, M., Guillon, G., Fischl, M.J., Donadi, M.C., Makovetz, J., Holmes, N., Zaveri, T., Toolan, E., Gontz, H.L., Yearwood, G.D., Logue, J., Bohannon, J.K., Mistretta, L., Byrum, R., Ragland, D., St. Claire, M., Kurtz, L.A., Miller, T., Reed, M.R., Young, J., Lee, J., Hensley, L.E., Kardos, K. and Berry, J.D. 2023. Development and clinical evaluation of a rapid point of care test for Ebola virus infection in humans. Viruses 15(2), 336. Ward, M.D., Kenny, T., Bruggeman, E., Kane, C.D., Morrell, C.L., Kane, M.M., Bixler, S., Grady, S.L., Quizon, R.S., Astatke, M. and Cazares, L.H. 2020. Early detection of Ebola virus proteins in peripheral blood mononuclear cells from infected mice. Clin. Proteomics 17(1), 11. Warren, T., Zumbrun, E., Weidner, J.M., Gomba, L., Rossi, F., Bannister, R., Tarrant, J., Reed, M., Lee, E., Raymond, J.L., Wells, J., Shamblin, J., Wetzel, K., Donnelly, G., Van Tongeren, S., Lackemeyer, N., Steffens, J., Kimmel, A., Garvey, C., Bloomfield, H., Blair, C., Singh, B., Bavari, S., Cihlar, T. and Porter, D. 2020. Characterization of Ebola virus disease (EVD) in rhesus monkeys for development of EVD therapeutics. Viruses 12(1), 92. Warsame, A., Eamer, G., Kai, A., Dios, L.R., Rohan, H., Keating, P., Katshishi, J. and Checchi, F. 2023. Performance of a safe and dignified burial intervention during an Ebola epidemic in the eastern Democratic Republic of the Congo, 2018-2019. BMC Med. 21(1), 484. Wauquier, N., Becquart, P., Gasquet, C. and Leroy, E.M. 2009. Immunoglobulin G in Ebola outbreak survivors, Gabon. Emerg. Infect. Dis. 15(7), 1136–1137. Weber, D.J., Fischer, W.A., Wohl, D.A. and Rutala, W.A. 2015. Protecting healthcare personnel from acquiring Ebola virus disease. Infect. Control. Hosp. Epidemiol. 36(10), 1229–1232. Wegner, G.I., Murray, K.A., Springmann, M., Muller, A., Sokolow, S.H., Saylors, K. and Morens, D.M. 2022. Averting wildlife-borne infectious disease epidemics requires a focus on socio-ecological drivers and a redesign of the global food system. EClinicalMedicine 47(1), 101386. Weinberg, M. and Yovel, Y. 2022. Revising the paradigm: are bats really pathogen reservoirs or do they possess an efficient immune system?. iScience 25(8), 104782. Winter, S.L., Golani, G., Lolicato, F., Vallbracht, M., Thiyagarajah, K., Ahmed, S.S., Lüchtenborg, C., Fackler, O.T., Brügger, B., Hoenen, T., Nickel, W., Schwarz, U.S. and Chlanda, P. 2023. The Ebola virus VP40 matrix layer undergoes endosomal disassembly essential for membrane fusion. EMBO. J. 42(11), e113578. Wong, S.S. and Wong, S.C. 2015. Ebola virus disease in nonendemic countries. J. Formos. Med. Assoc. 114(5), 384–398. Woolhouse, M.E.J., Rambaut, A. and Kellam, P. 2015. Lessons from Ebola: improving infectious disease surveillance to inform outbreak management. Sci. Transl. Med. 7(307), 307. Yeabah, T.O., Kaba, I., Ramaswamy, G., Dahal, P., Delamou, A., Vonhm, B.T., Jetoh, R.W., Merson, L., Levine, A.C., Relan, P., Harries, A.D. and Kumar, A.M.V. 2025. Factors associated with death in patients admitted with Ebola virus disease to Ebola Treatment Units in Guinea, Sierra Leone, and Liberia - December 2013 to March 2016. F1000Research 13(1), 672. Zampieri, C.A., Sullivan, N.J. and Nabel, G.J. 2007. Immunopathology of highly virulent pathogens: insights from Ebola virus. Nat. Immunol. 8(11), 1159–1164. Zhang, H.Q., Zhang, Q.Y., Yuan, Z.M. and Zhang, B. 2023. The potential epidemic threat of Ebola virus and the development of a preventive vaccine. J. Biosaf. Biosecur. 5(2), 67–78. Zhang, Y., Gong, Y., Wang, C., Liu, W., Wang, Z., Xia, Z., Bu, Z., Lu, H., Sun, Y., Zhang, X., Cao, Y., Yang, F., Su, H., Hu, Y., Deng, Y., Zhou, B., Zhao, Z., Fu, Y., Kargbo, D., Dafae, F., Kargbo, B., Kanu, A., Liu, L., Qian, J. and Guo, Z. 2017. Rapid deployment of a mobile biosafety level-3 laboratory in Sierra Leone during the 2014 Ebola virus epidemic. PLos Negl. Trop. Dis. 11(5), e0005622. Zhou, J., Zhang, X., Yao, Y., Xie, Y., Li, F., Wang, W., Yan, F., Peng, C., Zhang, B., Wu, Y., Cao, Z., Peng, Y., Gao, G., Zhang, H., Gong, P. and Chiu, S. 2025. Recombinant live attenuated measles virus-based vaccines inducing comprehensive protection against Ebola and Marburg viruses. Mol. Ther. 33(11), 5470–5488. | ||
| How to Cite this Article |
| Pubmed Style Muniroh M, Hermawati L, Fathurrohim F, Darifah S, Dewi LF, Putri NR, Fauziah YA, Darmadi EY, Setianingtyas D, Khairullah AR, Furqoni AH, Pratama BP. Ebola virus disease: A persistent challenge in global health security. Open Vet. J.. 2026; 16(2): 768-787. doi:10.5455/OVJ.2026.v16.i2.1 Web Style Muniroh M, Hermawati L, Fathurrohim F, Darifah S, Dewi LF, Putri NR, Fauziah YA, Darmadi EY, Setianingtyas D, Khairullah AR, Furqoni AH, Pratama BP. Ebola virus disease: A persistent challenge in global health security. https://www.openveterinaryjournal.com/?mno=298574 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.1 AMA (American Medical Association) Style Muniroh M, Hermawati L, Fathurrohim F, Darifah S, Dewi LF, Putri NR, Fauziah YA, Darmadi EY, Setianingtyas D, Khairullah AR, Furqoni AH, Pratama BP. Ebola virus disease: A persistent challenge in global health security. Open Vet. J.. 2026; 16(2): 768-787. doi:10.5455/OVJ.2026.v16.i2.1 Vancouver/ICMJE Style Muniroh M, Hermawati L, Fathurrohim F, Darifah S, Dewi LF, Putri NR, Fauziah YA, Darmadi EY, Setianingtyas D, Khairullah AR, Furqoni AH, Pratama BP. Ebola virus disease: A persistent challenge in global health security. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 768-787. doi:10.5455/OVJ.2026.v16.i2.1 Harvard Style Muniroh, M., Hermawati, . L., Fathurrohim, . F., Darifah, . S., Dewi, . L. F., Putri, . N. R., Fauziah, . Y. A., Darmadi, . E. Y., Setianingtyas, . D., Khairullah, . A. R., Furqoni, . A. H. & Pratama, . B. P. (2026) Ebola virus disease: A persistent challenge in global health security. Open Vet. J., 16 (2), 768-787. doi:10.5455/OVJ.2026.v16.i2.1 Turabian Style Muniroh, Muniroh, Luluk Hermawati, Fathurrohim Fathurrohim, Siti Darifah, Lola Febriana Dewi, Neshya Ruriana Putri, Yessy Andriani Fauziah, Eveline Yulia Darmadi, Dwi Setianingtyas, Aswin Rafif Khairullah, Abdul Hadi Furqoni, and Bima Putra Pratama. 2026. Ebola virus disease: A persistent challenge in global health security. Open Veterinary Journal, 16 (2), 768-787. doi:10.5455/OVJ.2026.v16.i2.1 Chicago Style Muniroh, Muniroh, Luluk Hermawati, Fathurrohim Fathurrohim, Siti Darifah, Lola Febriana Dewi, Neshya Ruriana Putri, Yessy Andriani Fauziah, Eveline Yulia Darmadi, Dwi Setianingtyas, Aswin Rafif Khairullah, Abdul Hadi Furqoni, and Bima Putra Pratama. "Ebola virus disease: A persistent challenge in global health security." Open Veterinary Journal 16 (2026), 768-787. doi:10.5455/OVJ.2026.v16.i2.1 MLA (The Modern Language Association) Style Muniroh, Muniroh, Luluk Hermawati, Fathurrohim Fathurrohim, Siti Darifah, Lola Febriana Dewi, Neshya Ruriana Putri, Yessy Andriani Fauziah, Eveline Yulia Darmadi, Dwi Setianingtyas, Aswin Rafif Khairullah, Abdul Hadi Furqoni, and Bima Putra Pratama. "Ebola virus disease: A persistent challenge in global health security." Open Veterinary Journal 16.2 (2026), 768-787. Print. doi:10.5455/OVJ.2026.v16.i2.1 APA (American Psychological Association) Style Muniroh, M., Hermawati, . L., Fathurrohim, . F., Darifah, . S., Dewi, . L. F., Putri, . N. R., Fauziah, . Y. A., Darmadi, . E. Y., Setianingtyas, . D., Khairullah, . A. R., Furqoni, . A. H. & Pratama, . B. P. (2026) Ebola virus disease: A persistent challenge in global health security. Open Veterinary Journal, 16 (2), 768-787. doi:10.5455/OVJ.2026.v16.i2.1 |