| Research Article | ||
Open Vet. J.. 2026; 16(2): 1351-1360
Open Veterinary Journal, (2026), Vol. 16(2): 1351-1360 Research Article Influence of photoperiod on testicular activity in rams: Comparative reproductive, hormonal, and histological insights across seasonsMaha M. Elgebaly1*, Nehal Ibrahim2, Nehal I. A. Goda3, Said Amin1 and Asmaa A. Abdallah11Department of Theriogenology, Faculty of Veterinary Medicine, Zagazig University, Zagazig, Egypt 2Department of Surgery, Anesthesiology and Radiology, Faculty of Veterinary Medicine, Zagazig University, Zagazig, Egypt 3Department of Histology and Cytology, Faculty of Veterinary Medicine, Zagazig University, Zagazig, Egypt *Corresponding Author: Maha M. Elgebaly. Department of Theriogenology, Faculty of Veterinary Medicine, Zagazig University, Zagazig, Egypt. Email: mahaelgebaly46 [at] gmail.com Submitted: 02/12/2025 Revised: 14/01/2026 Accepted: 19/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
ABSTRACTBackground: The photo period has a great effect on the testicular efficiency of Rams, as it differs between breeding in comparison to, Non- Breeding periods. The male hormone also shows great variation according to the photo period. Aim: This study aimed to examine the seasonal variation in ram fertility (ultrasonographic, hormonal, histopathological). Methods: Throughout the breeding and non-breeding seasons, blood samples were collected weekly from the animals’ jugular veins. On the same day as the measurement, 10 ml of blood from each animal was collected in Vacutainer tubes, and testosterone hormone, follicle stimulating hormone (FSH) , and leutinizing hormone (LH) concentrations were evaluated. Then, scrotal and testicular dimensions were recorded weekly. An ultrasound machine was used to image the testicles and epididymis of the ram in the standing position. For each animal, six representative fields were selected for morphometric analysis, and quantitative measurements were carried out. Results: The result dedicated to that season has a greater significant impact on testosterone levels and FSH and LH levels. A higher level of significant variation in testicular measurement was observed. The testicular parenchyma of both ram groups was uniformly echogenic (B-mode), but during the breeding season, the mediastinum testis was large in size and more clear compared to out of breeding. Ultrasonographic image of the tail of the epididymis. Appears to be enlarged and heterogeneous in the echotexture and less echogenic than the testis. Seminiferous tubules were lined with stratified epithelium, which comprised distinct phases of one row of spermatoic cells, huge primary and secondary smaller spermatocytes, and round spermatids clustered at the lumen of the tubules. Immunohistochemical studies showed that the androgen receptor antibody was expressed at the seminiferous tubules, particularly in Sertoli cells and myoid cells which appeared obviously in the breeding season. The immunoreaction for S-100 protein revealed strong S-100 expression in Sertoli cells, especially in their nuclei, which appeared darker in the breeding season. The strength of S100 protein immunoreactivity in Sertoli cells is somewhat reliant on the cyclical fluctuations of the seminiferous epithelium. Conclusion: The present study concluded that differences were evident in the hormonal profile, ultrasonographic findings, and histopathological characteristics of the tests, and this was associated with increased testicular activity and more active seminiferous tubules histologically in breeding season comparing to non-breeding seasons. These results confirm the strong seasonal influence on reproductive performance in rams. Keywords: Ram, Hormones, Breeding season, Ultrasonography, Seminiferous tubules. IntroductionInfertile rams should be detected at the beginning of the breeding season. This can be achieved by evaluating the breeding efficiency of rams and detecting how successfully they reproduce (Gouletsou et al., 2003b ; Fthenakis et al., 2010). Multiple environmental influences, including elevated ambient temperature, relative humidity, and variations in photoperiod, shape Ram reproductive activity (Hulet et al., 1986). In the subtropical region, estrus activity and male reproduction vary according to the length of the day (Aboul-Ela et al., 1987). In Egypt, the midsummer climate is characterized by unfavorable temperatures, which affect the fertility of male rams. Male reproductive efficiency is generally constrained by three major elements: sexual activity, semen quality, and semen volume (Karagiannidis et al., 2000; Gouletsou et al., 2003b; Zarei et al., 2009). Moreover, previous records indicate that sheep in Egypt exhibit their highest conception rates during the autumn season, when ambient temperatures are moderate (Aboul-Naga et al., 1992; Ahmed, 2008). Furthermore, (Pérez et al., 1997) documented that the reproductive performance of rams fluctuates throughout the year, with testicular activity increasing during autumn and winter and showing a marked decline in the summer months. This change in temperature and humidity may be the main cause of thermal load that harms spermatogenesis and affects the semen feature (Kunavongkrit et al., 2005). Several hidden abnormalities cannot be identified by routine physical examination. Ultrasonographic scanning is employed to evaluate the genital organs of rams and detect any abnormalities (Gouletsou et al., 2003b). Ultrasonographic scanning is widely used to examine the testicles in small ruminants (Andrade et al., 2014; Gouletsou, 2017; Ribeiro et al., 2017). At a frequency of 5–7.5MHz, ultrasonography can be used to see the ovine male genitalia (Aboul-Ela et al., 1987; Sargison et al., 2018). Additionally, the internal reproductive organs can be seen with B-mode, grayscale ultrasonography (Chandolia et al., 1997). Testicular ultrasonography is mainly used to assess an animal’s reproductive status (Ahmadi et al., 2012). Additionally, testicular ultrasonographic measures can give an indication of testicular sperm production. Ribeiro et al. (2017) reported that evaluating testicular echogenicity and assessing the size of the vesicular and bulbourethral glands may assist in identifying rams with superior sexual performance. In addition, Gouletsou and Fthenakis (2010) highlighted that in most animal species, male accessory glands are often overlooked in standard breeding soundness examinations. Using transrectal ultrasonography for studying the rams sex glands still needs more work (Camela et al., 2017). In addition, sexual hormones are affected by climatic fluctuations, which can significantly influence the physiological events of the reproductive cycle (Todini et al., 2007; Abecia et al., 2012). Testosterone is a crucial hormone for spermatogenesis and sexual activity; therefore, following seasonal changes in testosterone production will establish periods that are beneficial for male fertility (Swelum et al., 2017; Todini et al., 2017). Higher testosterone concentration is also positively associated with improved sperm motility (Swelum et al., 2017). Tests are enclosed by Tunica albuginea, that is made up of dense, irregular type connective tissue that includes fibroblasts, sparse reticular collagen fibers, smooth muscle cells, and elastic fibers. The tunica vaginalis comprises a mesothelial layer supported by connective tissue that merges with the tunica albuginea. Mediastinum tests revealed that thin fibrous septa extend inward, partially or completely dividing the testicular parenchyma into lobules that house the seminiferous tubules (Al-Mahmodi, 2018). The seminiferous tubules are lined with stratified epithelium created from several phases of spermatogenic cells, such as primary and secondary spermatocytes, as well as spermatids clustered within the lumen of the tubules. These tubules also contain Sertoli cells, which are very sparse in number but function as reinforcement cells positioned along the basement membrane (Sanad et al., 2021). The interstitial tissue found between the seminiferous tubules is composed of loose connective tissue cells, Leydig cells, and a vast network of blood arteries (Al-Mahmodi, 2018). Androgen plays an essential role in several physiological processes, particularly those required for male sexual maturation. They are crucial for maintaining spermatogenesis and regulating the secretion of gonadotropin The action of androgens occurs by binding to the androgen receptor (AR). In human tests, AR immunoexpression has been identified in Sertoli, myoid, and Leydig cells, but not in germ cells. Androgen receptors demonstrate variable immunohistochemical reactivity depending on the age and stage of the spermatogenic cycle (Rodríguez et al., 2008). The term S-100 refers to the characteristic solubility of these proteins in a 100% ammonium sulfate–saturated solution under neutral pH conditions (Moore, 1965). A group of small, acidic, water-soluble, Ca²+-binding proteins(Zimmer et al., 1995). These proteins play diverse cellular roles, ranging from calcium homeostasis to the modulation of intracellular processes such as enzymatic activity, energy metabolism, cell motility, and secretion. In addition, they contribute to nuclear functions, such as transcription and programed cell death, which are associated with extracellular regulatory mechanisms. Materials and MethodsAnimalsThe Veterinary Medicine Faculty Hospital at Zagazig University, Egypt, provided ten mature rams weighing between 40 and 50 kg and aged between 18 and 24 months. The pasture of the college’s veterinary hospital is home to these rams, and similar feeding and management are administered to keep animals in captivity. Experimental designCollection of blood samplesThroughout the breeding and non-breeding seasons, blood samples were collected weekly from the jugular veins of the animals. On the same day as the measurement, 10 ml of blood from each animal was collected in Vacutainer tubes. Serum samples were collected, centrifuged at 850 × g for 10 minutes at room temperature, then placed at −20°C until further analysis. The enzyme immunoassay reagent kit was used to measure testosterone, follicle stimulating hormone (FSH), and leutinizing hormone (LH) concentrations (varying from 0.1 to 20 ng/ml). Testicular measurementFor each male patient, scrotal and testicular dimensions were recorded weekly during the designated months, ensuring that measurements were performed at a consistent time. The scrotal circumference was measured using a plastic meter (Ahmed and Noakes, 1995). The testicular dimensions were determined using a metallic caliper, measuring the length from the proximal to distal pole and the width from the point of attachment to the free edge of each testis (Islam and Land, 1977). Ultrasonographic examination of scrotum and its contentsAn ultrasound machine (software 2020) was used to image the testicles and epididymis of the ram in the standing position. Rams were examined ultrasonographical using the 7–10 MHz linear probe of the scanner. To lift the tail, the animal was briefly confined by a helper. The scrotal hair was clipped to evaluate the echotexture and echogenicity of the testicular parenchyma (Gouletsou et al, 2003a; Camella et al., 2017). Ultrasonographic evaluation of the accessory sex glandsThe ultrasonic transducer was placed in the rectum and advanced cranially up to the urinary bladder-pelvic urethral junction after using a coupling gel lubricant, which acted as a guide after fecal clearing. As accessory sex glands, the ampulla and seminal glands are located next to the urine bladder. Upon imaging, the disseminated section of the prostate gland was obvious at the pelvic urethra level, whereas the bulbourethral glands were readily discernible after withdrawing the rectal probe. The size of the accessory sex glands was subsequently assessed using ultrasonography. HistopathologySamples were removed from the animals immediately after slaughter. Preparation of tissue samplesThe research samples were embedded in 10% neutrally buffered formalin, dried, and finally purified in xylene. All specimens were first saturated with soft melted paraffin and then imbedded in hard paraffin. The paraffin-embedded tissue sections were routinely stained using Harris’ hematoxylin and eosin staining (Suvarna et al., 2018). Preparation of tissue samples for IHC analysisSections were placed on charged slides, deparaffinized using xylene, and rehydrated. Subsequently, they were immersed in 0.3% hydrogen peroxide solution, rinsed with 10% normal rabbit serum, and incubated with antisera containing the primary antibodies.
After washing, the sections were stained with Mayer’s hematoxylin and then dehydrated, refined, and fixed in Canada balsam for additional microscopic analysis (Suvarna et al., 2018). All stained sections were examined under a standard light microscope. An Olympus BX21 microscope was used, and images were captured with a high-resolution Canon® camera at the Department of Histology and Cytology, Zagazig University. Histomorphometric analysisFor each animal, six representative fields were selected for morphometric analysis, and quantitative measurements were performed using ImageJ software (Fiji ImageJ; 1.51n, NIH) to detect the following:
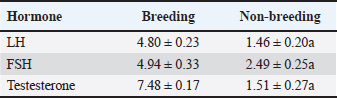
Statistical analysisData were statistically analyzed using analysis of variance (paired independent t-test) using a statistical software program (SPSS for Windows, version 16.0, SPSS Inc., Chicago, IL), with some data analyzed using GraphPad Prism 8.0.2 (GraphPad Software, Inc). Results are given as Mean ± SE. The independent samples t-test was performed to evaluate all the morphometric measurements. A p-value < 0.05 was considered significant. Ethical approvalThe study was approved by the Institutional Animal Ethics Committee, and all operations were conducted in accordance with the animal care and use guidelines and ethical regulations of Zagazig University in Egypt. ResultsHormonal analysisSignificant seasonal variation was observed in testosterone, FSH, and LH levels, with marked differences between breeding and nonbreeding periods. The results indicated that season exerts a significant influence on testosterone levels, as well as on FSH and LH concentrations (Table 1). Table 1. Hormone levels during the breeding and nonbreeding periods are represented by the mean ± SE, p < 0.05 considered significant.
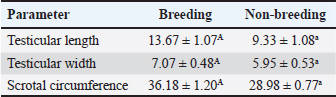
Testicular measurementThe scrotal measurements are presented in (Table 2). Measurable characteristics, including scrotal circumference, testicular lengths, and testicular widths, were all statistically significant (p ≤ 0.05), indicating a higher level of significant variation in testicular measurement. Table 2. Testicular measurements are represented by the mean ± SE, p < 0.05 considered significant the mean value (±SD) clearly indicates higher measurement in the breeding season groups compared with the non-breeding season groups.
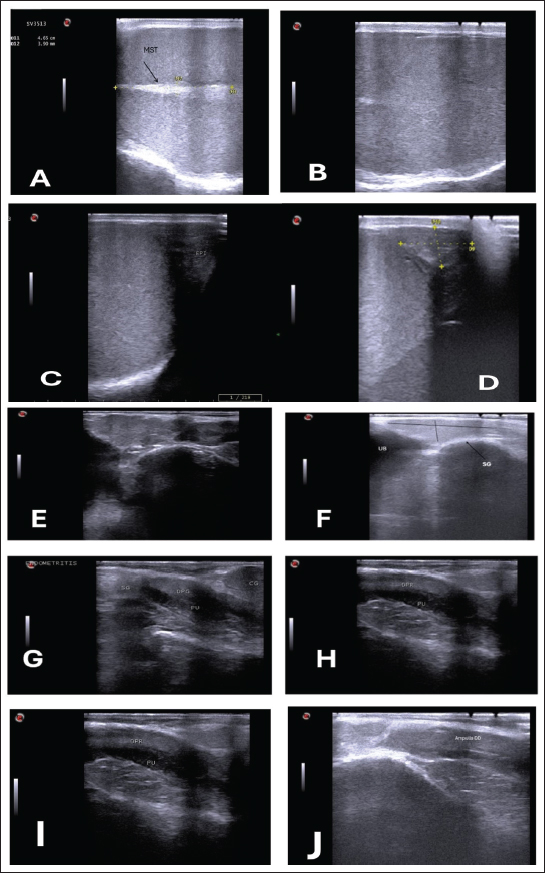
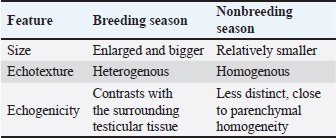
Ultrasonographic examinationIn the rams’ genitalia throughout the clinical evaluation had a uniform consistency, and this investigation did not have any pain reactions. The testicular parenchyma of both ram groups was uniformly echogenic (B-mode) (Fig. 1A), but during the breeding season, the mediastinum testis was large and also more clear compared to out of breeding (Fig. 1B).
Fig. 1. (A) Parenchyma showing uniform echogenicity with large mediastinum tests (length and thickness) and homogenous appearance during the non-breeding season (B), (C) Show large size of epididymis compared to small size during nonbreeding season in(D). (E–H) Show different ecogenicity and variation in the size of male sex gland during breeding and non breeding season. (E) show enlargement and lobulation of seminal gland comparing to loss of lobulation and decrease in its size in (F). (I) also show more clear dorsal part of prostate gland during breeding season. The tail of the epididymis caught on from a slanting plane near the distal pole of the testis. Ultrasonographic scan of the testis showing the tail of the epididymis. The tail appears to be larger, heterogeneous in echotexture, and less echogenic than the testis, which may be related to distension with semen content during the mating season (Fig. 1C). During the non-breeding season, the tail of the epididymis is significantly smaller and more homogenous, indicating decreased sperm storage activity (Fig. 1D; Table 3). Table 3. Comparison of the epididymal tails of rams during and outside the breeding season.
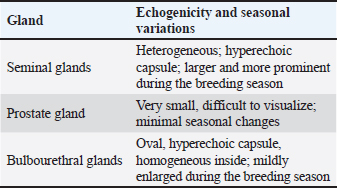
The sex glands of the rats were evaluated ultrasonographically, and the results declared seasonal fluctuations. Higher secretory activity was reflected in the seminal gland by enhanced echogenicity, more lobulated appearance, and clearly defined margins (heterogenous appearance) throughout the breeding season (Fig. 1E; Table 4). Additionally, compared with the nonbreeding phase, the prostate and bulbourethral glands had a denser echotexture and were marginally more noticeable (Fig. 1G–J). The glands seemed smaller, less distinct, and had a more uniform and diminished echogenic pattern during the non-breeding season as a result of lower functional activity (Fig. 1F). Table 4. Seasonal variation in the echogenicity of the glands.
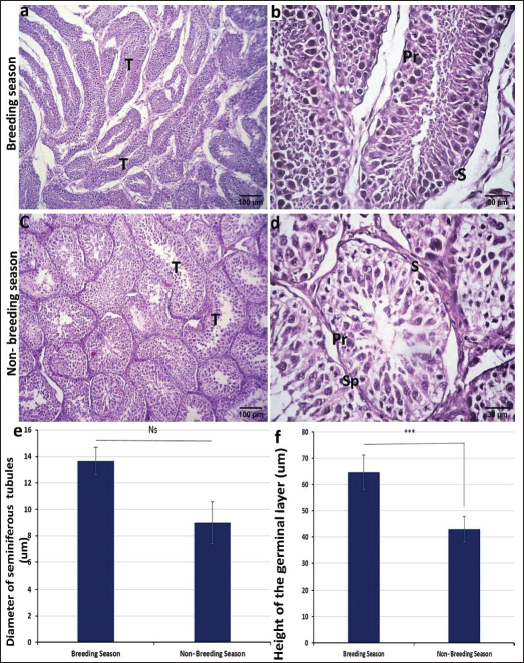
Histological examinationTo study the differences in histological structure during the fluctuations from the breeding season to the non-breeding season, testes were collected at different time points from December to March (breeding season) and from early June to late July (non-breeding season). The testis was divided by dense connective tissue septa into lobules containing seminiferous tubules where the sperms are produced. These tubules were more elongated in the breeding season (Fig. 2a) and round or oval in shape during the non-breeding season (Fig. 2c). The mean diameter of these tubules was larger in the breeding season than in the nonbreeding season, which is statistically insignificant (Fig. 2e). These seminiferous tubules were lined by stratified germinal epithelium composed of several phases, such as round spermatogonia with round dark nuclei, huge primary spermatocytes with enormous spherical nuclei, secondary spermatocytes, and spermatids. Sertoli cells can also be found in seminiferous tubules, which are oval or pyramidal in shape and positioned in the basal portion of the cell with ovoid nuclei (Fig. 2b and d).
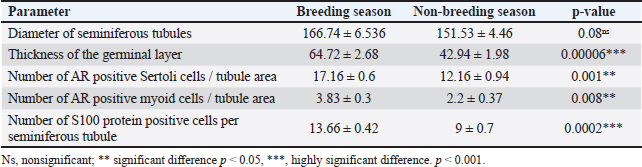
Fig. 2. Photomicrograph of H and E stained section of ram testis during breeding season (a and b) and ram testis during non-breeding season (c and d); showing seminiferous tubules “T”, spermatogonium “SP”, primary spermatocytes “Pr”, and sertoli cells “S”. Morphometrical analysis of seminiferous tubule diameters and germinal layer thickness (e and f). Data are presented as mean ± SE. Ns, non-significant; *** highly significant p < 0.001. Histomorphometry revealed a highly significant difference (p < 0.001) in the germinal epithelium thickness between the breeding and non-breeding seasons. The interstitial tissue among the seminiferous tubules contains loose connective tissue and interstitial Leydig cells (Table 5). Table 5. Histomorphometry measurements representing the mean ± SE of seminiferous tubule diameter, germinal layer thickness, number of AR-positive Sertoli cells/tubule area, number of AR-positive myoid cells/tubule area, and number of S100 protein-positive cells per seminiferous tubule.
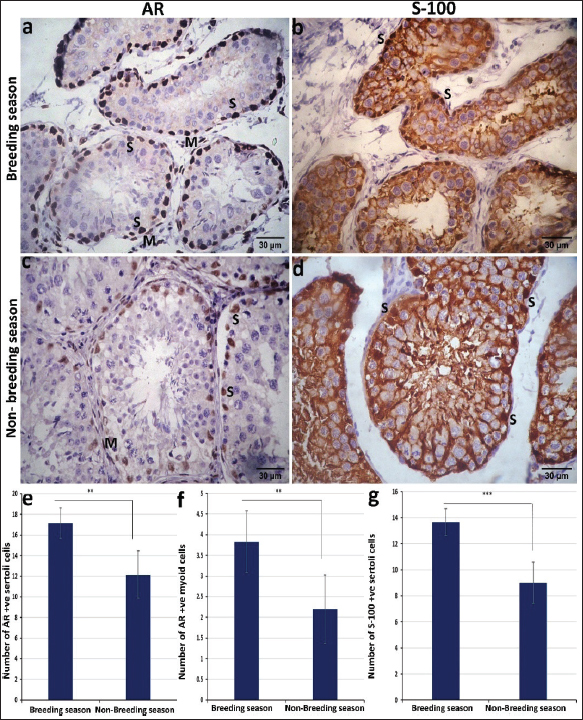
Immunohistochemical studies showed that the androgen receptor antibody was expressed at the seminiferous tubules, particularly in the sertoli and myoid cells. The reaction was obviously strong in sertoli and myoid cells in the breeding season, in contrast to the non-breeding season, where the intensity of the reaction was intermediate in sertoli cells and very mild to negative in myoid cells (Fig. 3a and c). Statistically, the number of AR-positive Sertoli cells and myoid cells was higher in the breeding season than in the non-breeding season p < 0.05 (Fig. 3e and f). The immunoreaction for S-100 protein revealed strong S-100 expression in Sertoli cells, especially in their nuclei, which appeared darker in the breeding season than in the non-breeding season (Fig. 3b and d). Histomorphometry revealed a highly significant difference (p < 0.001) in the number of S-100-positive Sertoli cells between the breeding and non-breeding seasons (Fig. 3g).
Fig. 3. Photomicrograph of the immunohistochemical expression of ram testis during the breeding season (a and b) and the non-breeding season (c and d). The intensity of the reaction for the AR of the sertoli and myoid cells was strong in the breeding season (a), intermediate in the sertoli cells, and very mild to negative in the myoid cells in the non-breeding season (c). Immunoreactivity for S-100 protein revealed stronger expression in the breeding season (b) than in the non-breeding season (d). Morphometric analysis for the number of AR-positive sertoli cells/tubule area (e), number of AR-positive myoid cells/tubule area (f), and number of S100 protein-positive cells per seminiferous tubule (g). Data are presented as mean ± SE. ** significant different p < 0.05, *** highly significant p < 0.001. DiscussionSignificant seasonal differences were observed between the breeding and nonbreeding periods. This difference was considerably higher. Rosa and Bryant (2003) confirmed that sheep breeds found in mid- and high-altitude areas generally reveled seasonality in their reproduction ability, which is proven by a few studies that effect on reproductive traits of rams (Zamiri et al., 2010). The testicular measurements that were taken in our study manifested a clear seasonal variation. Measurements of the tests, including length, width, and circumference, showed a marked increase during the breeding season relative to the non-breeding periods. This variance reverberates that the tests show physiological activation under the effect of high testosterone concentrations and increased gonadotropic release during the reproductive season. There was a higher significant difference (p > 0.05) for all measures (length, width, and circumference), which agrees with the result reported by Belkhiri et al. (2017). This conclusion may be related to the increase in testicular volume, which is directly related to the increase in the diameter of the seminiferous tubules and the improvement of spermatogenesis activity. Several previous studies in rams and bucks have reported the same findings, indicating that testicular dimensions are true indicators of reproductive capacity (Zaher et al., 2020). According to our findings, hormonal levels significantly varied by season. There was a higher significant difference (p > 0.05) in testosterone, LH, and FSH levels. These findings concur with those of Abecia et al. (2012), who found that season reproductivity is a phenomenon primarily impacted by changes during the year and the photoperiod, leading to clearly defined reproductive variation. In agreement with Elbaz and Abdel Razek (2019), who observed variation in the hormonal profile in various sheep breeds over the different seasons of the year. The present study investigated the ultrasonographic images and seasonal variations of the accessory sex glands, epididymis, and testes in rams, with a focus on the mediastinum testis, seminal vesicles, and disseminating part of the prostate during breeding and non-breeding seasons. Our findings revealed a significant increase in the size of the mediastinum testis and higher echogenicity of the parenchyma during the breeding season compared with the nonbreeding season. This matched the findings of Elbaz and Abdel Razek (2019). These changes are dedicated to increasing testicular activity and improving spermatogenesis, which are strongly influenced by seasonal fluctuations in testosterone secretion. Previous studies in rams (Kaulfuss et al., 2006) and other small ruminants have also reported enlargement of the testes and increased echogenicity of the mediastinum testis during the peak of reproductive activity, supporting the present result. The seminal vesicles had a clear lobulated appearance in addition to hyperechoic septa that separated the glandular tissue, which became more prominent during the breeding season. This finding is attributed to the oversecretion of seminal plasma under the effect of androgens. Comparable features have been reported in bulls and stallions, where the glandular tissue becomes more hyperechogenic and highly lobulated during periods of higher sexual activity (Edwards, 2008; Gnemmi and Lefebvre, 2009). Therefore, the marked lobulation observed in rams may act as an ultrasonographic indicator of reproductive status. Taken together, these seasonal changes highlight the strong relationship between ultrasonographic findings and ram endocrine activity. The observed increase in testicular size and increased echogenicity of the testes and accessory sex glands during the breeding season are consistent with elevated testosterone levels and improved reproductive performance. The distinct lobulation of the seminal gland and the seasonal enhancement of the accessory gland and mediastinum are important indicators of reproductive activity and may serve as practical markers for breeding soundness evaluation. The primary function of the testes is sperm production, with the seminiferous tubules, Sertoli cells, and Leydig cells being key components. In addition, a network of ducts and other testicular tissues plays a supportive role in the production and transport of sperm (Elzoghby et al., 2014, Al-Mahmodi, 2018). Our work revealed that the seminiferous tubules were tubular, round, or oval in shape with a mean diameter from 166.74 ± 6.536 (breeding season) to 151.53 ± 4.46 (nonbreeding season). This is in contrast to the findings of Al-Mahmodi (2018) who reported that the mean diameters of the seminiferous tubules were 264 ± 2.9 µm in rams and 245 ± 1.9 µm in bucks. Seminiferous tubules were lined with stratified epithelium consisting of different stages of one row spermatogenic cells, large primary and smaller secondary spermatocytes, and round spermatids clustered at the lumen of the tubules (Tala’a and Hamdi, 2023). This result is in line with our findings. Within the seminiferous tubules, Sertoli cells are positioned at the basal layer and display oval to pyramidal morphology, containing ovoid-shaped nuclei. These cells are relatively few and serve as supporting cells at the basement membrane of the seminiferous tubules. Although they do not directly produce sperm or testosterone, Sertoli cells perform several critical functions that facilitate spermatogenesis and spermatozoa transport (Al-Mahmodi, 2018). The testes’ interstitial connective tissue comprises blood vessels, fibroblasts, loose connective tissue cells, and interstitial Leydig cells. Leydig cells are nearly absent before the onset of puberty, when testosterone production begins. Their primary role is the synthesis and secretion of androgens, particularly testosterone, the principal male sex hormone (Sikarwar et al., 2022). Our work revealed that immunohistochemical studies showed that the androgen receptor antibody was expressed at the seminiferous tubules, especially in Sertoli cells and myoid cells, which appeared obviously in the breeding season compared to the non-breeding season. (Rodríguez et al. (2008) reported that Sertoli cells and contractile peritubular cells express a high percentage of the androgen receptor, whereas the germinal line is negative in reaction. Our study showed that the immunoreaction for S-100 protein revealed strong S-100 expression in Sertoli cells, especially in their nuclei, which appeared darker in the breeding season. The intensity of S-100 protein immunoreactivity in Sertoli cells is influenced by cyclic changes within the seminiferous epithelium. Relatively strong immunoreactivity is observed during spermatid elongation stages. Notably, this study highlights the particularly intense staining observed in the intratesticular excurrent duct system (Amselgruber et al., 1994) . S-100 immunoreactivity was pronounced in Sertoli cells, with the protein localized within both the cytoplasmic and nuclear compartments (Abd-Elmaksoud et al., 2014). Moreover, S-100 expression was absent in germ cells, leydig, and myofibroblast cells of the studied species at different stages. Spermatogonia, spermatocytes in different phases of meiosis, spermatids, and spermatozoa were consistently negative for S-100 protein. A strong immunoreaction for S-100 in leydig cells was characteristic for the testes of rats and cats, and a weak immunoreaction of this cell type was also observed in horses and pigs. Staining of particular cells has been found in rodents and carnivores (Amselgruber et al., 1994). ConclusionThe abovementioned study concluded that differences were evident in the hormonal profile, ultrasonographic findings, and histopathological characteristics of the tests, and this was associated with increased testicular activity and more active seminiferous tubules histologically in the breeding season compared to the non-breeding seasons. These results confirm the strong seasonal influence on reproductive performance in rams. AcknowledgmentsNone. Conflict of interestThe authors have no conflicts of interest to declare. FundingNot applicable. Authors’ contributionsAll authors contributed equally to this study. Data availabilityAll data were included in this manuscript. ReferencesTala'a, A.A. and Hamdi, Y. 2023. Histomorphometric study of seminiferous tubules and epididymis in adult ram. Univ. Thi-Qar J. Agri. Res. 12, 206–217. Abd-Elmaksoud, A., Shoeib, M.B. and Marei, H.E.S. 2014. Localization of S-100 proteins in the testis and epididymis of poultry and rabbits. Anatomy Cell. Biol. 47, 180–187. Abecia, J.A., Forcada, F. and González-Bulnes, A. 2012. Hormonal control of reproduction in small ruminants. Anim. Reprod. Sci. 130, 173–179. Aboul-Ela, M.B., Aboul-Naga, A.M., Shalaby, T.H. and Maijala, K. 1987. Physiological response to climatic changes in Finnish Landrace ewes imported to Egypt and their half-sibs raised in Finland. Livestock. Prod. Sci. 17, 179–185. Aboul-Naga, A.M., Aboul-Ela, M.B. and Hassan, F. 1992. Manipulation of reproductive activity in subtropical sheep. Small. Ruminant. Res. 7, 151–160. Ahmadi, B., Lau, C.P.S., Giffin, J., Santos, N., Hahnel, A., Raeside, J., Christie, H. and Bartlewski, P. 2012. Suitability of epididymal and testicular ultrasonography and computerized image analysis for assessment of current and future semen quality in the ram. Exp. Biol. Med. 237, 186–193. Ahmed, N. and Noakes, D. 1995. Seasonal variation in testes size, libido and plasma testosterone concentration in British goat. Anim. Sci. 61, 553–559. Ahmed, A. 2008. Biological evaluation of Barki sheep under two different breeding seasons. Egypt. J. Anim. Prod. 45, 15–24. Al-Mahmodi, A. 2018. Microscopical and biometrical investigations of the testes in adult ram and adult buck in Al-Najaf Al-Ashraf Province. Amselgruber, W.M., Sinowatz, F. and Erhard, M. 1994. Differential distribution of immunoreactive S-100 protein in mammalian testis. Histochemistry 102, 241–245. Andrade, A.K.G., Soares, A.T., Freitas, F.F., Silva, S., Peña-Alfaro, C.E., Batista, A. and Guerra, M. 2014. Testicular and epididymal ultrasonography in Santa Inês lambs raised in Brazil. Anim. Reprod. 11, 110–118. Belkhiri, Y., Bouzebda-Afri, F., Bouzebda, Z. and Djaout, A. 2017. Testicular morphometric measurements of Ouled Djellal lambs from birth to puberty. Global Veterinaria 18, 85–91. Camela, E.S.C., Nociti, R.P., Santos, V.J.C., Macente, B.I., Maciel, G.S., Feliciano, M.A.R., Vicente, W.R.R., Gill, I., Bartlewski, P.M. and Oliveira, M.E.F. 2017. Ultrasonographic characteristics of accessory sex glands and spectral Doppler indices of the internal iliac arteries in peri- and post-pubertal Dorper rams raised in a subtropical climate. Anim. Reprod. Sci. 184, 29–35. Chandolia, R.K., Bartlewski, P.M., Omeke, B.C., Beard, A.P., Rawlings, N.C. and Pierson, R.A. 1997. Ultrasonography of the developing reproductive tract in ram lambs: effects of a GnRH agonist. Theriogenology 48, 99–117. Edwards, J.F. 2008. Pathologic conditions of the stallion reproductive tract. Anim. Reprod. Sci. 107, 197–207. Elbaz, H.T. and Abdel Razek, E.M. 2019. Ultrasonographic measurements of reproductive organs of male goat during non-breeding season. PSM. Vet. Res. 4, 13–23. Elzoghby, E., Sosa, G. and Hussein, M. 2014. Postnatal development of the sheep testis. Benha. Vet. Med. J. 26, 186–190. Fthenakis, G., Gouletsou, P., Mavrogianni, V. and Fragkou, I. 2010. Sheep diagnostic medicine. Small Ruminant Res. 92(1), 1. Gnemmi, G. and Lefebvre, R.C. 2009. Ultrasound imaging of the bull reproductive tract: an important field of expertise for veterinarians. Vet. Clin. North Am. Food Anim. Pract. 25, 767–779. Gouletsou, P.G. 2017. Ultrasonographic examination of the scrotal contents in rams. Small. Ruminant. Res. 152, 100–106. Gouletsou, P.G. and Fthenakis, G.C. 2010. Clinical evaluation of reproductive ability of rams. Small. Ruminant. Res. 92, 45–51. Gouletsou, P.G., Amiridis, G.S., Cripps, P.J., Lainas, T., Deligiannis, K., Saratsis, P. and Fthenakis, G.C. 2003. Ultrasonographic appearance of clinically healthy testicles and epididymides of rams. Theriogenology 59, 1959–1972. Hulet, C.V., Shupe, W.L., Ross, T. and Richards, W. 1986. Effects of nutritional environment and ram effect on breeding season in range sheep. Theriogenology 25, 317–323. Islam, A.B.M.M. and Land, R.B. 1977. Seasonal variation in testis diameter and sperm output of rams of breeds of different prolificacy. Anim. Sci. 25, 311–317. Karagiannidis, A., Varsakeli, S. and Karatzas, G. 2000. Characteristics and seasonal variations in semen of Alpine, Saanen and Damascus goat bucks born and raised in Greece. Theriogenology 53, 1285–1293. Kaulfuss, K.H., Giucci, E., Süss, R. and Wójtowski, J. 2006. An ultrasonographic method to study reproductive seasonality in ewes isolated from rams. Reprod. Domest. Anim. 41, 416–422. Kunavongkrit, A., Suriyasomboon, A., Lundeheim, N., Heard, T.W. and Einarsson, S. 2005. Management and sperm production of boars under differing environmental conditions. Theriogenology 63, 657–667. Moore, B.W. 1965. A soluble protein characteristic of the nervous system. Biochem. Biophysical Res. Commun. 19, 739–744. Perez, C.R., Lopez, A., Castrillejo, A., Bielli, A., Laborde, D., Gastei, T., Tagle, R., Queirolo, D., Franco, J., Forsberg, M. and Rodriguez-Martinez, H. 1997. Reproductive seasonality of Corriedale rams under extensive rearing conditions. Acta. Vet. Scand. 38, 109–117. Ribeiro , M.D.S., Quirino, C.R., Bartholazzi Junior, A. and Pacheco, A. 2017. Biometry and ultrasound evaluation of testicles and accessory glands in Santa Ines rams. Revista Brasileira De Zootecnia 46, 317–323. Rodríguez, H., Espinoza, O., Sarabia, L., Tamayo, C., Sepúlveda, M., Inostroza Escobar, J., Araya, J. and Moriguchi, K. 2008. Histological and functional organization in human testicle: expression of receptors c-kit and androgens. Rosa, H.J.D. and Bryant, M.J. 2003. Seasonality of reproduction in sheep. Small. Ruminant. Res. 48, 155–171. Sanad, N.H., Abbas, H.R., Yaseen, A.A., Habeeb, I.A. and Alsalim, H.A. 2021. Hormonal, histological and comparative study of the effect of pure ginseng on testicular function in the breeding/non-breeding season of rams in Basrah. Arch. Razi. Inst. 76, 1519–1535. Sargison , N., Crilly, J. and Hopker, A. 2018. Practical Lambing and Lamb Care: a Veterinary GuideOX4 2DQ, UK: Oxford. Sikarwar, V.S., Mašláni, A., Hl., na, M., Fathi, J., Mates, T., Pohořelý, M., Meers, E., Šyc, M. and Jeremiáš, M. 2022. Thermal plasma-assisted pyrolysis and gasification of RDF by utilizing sequestered CO2 as gasifying agent. J. CO2. Utilization. 66, 102275. Suvarna, K.S., Layton, C. and Bancroft, J.D. 2018. Bancroft’s Theory and Practice of Histological Techniques. Amesterdam, Holand: Elsevier. Swelum, A.A., Ayadi, M., Alhidary, I., Alowaimer, A. and Abouheif, M. 2017. Relationships between body fatness, leptin, testosterone and reproductive performance in ram lambs as affected by level and frequency of feeding. Theriogenology 89, 79–85. Todini, L., Malfatti, A., Terzano, G.M., Borghese, A., Pizzillo, M. and Debenedetti, A. 2007. Seasonality of plasma testosterone in males of four Mediterranean goat breeds and in three different climatic conditions. Theriogenology 67, 627–631. Zaher, H., Alawaash, S. and Swelum, A. 2020. Effects of season and breed on reproductive performance of sheep. J. Anim. Reprod. Biotechnol. 35, 149–154. Zamiri, M.J., Khalili, B., Jafaroghli, M. and Farshad, A. 2010. Seasonal variation in seminal parameters, testicular size and plasma testosterone concentration in Iranian Moghani rams. Small Ruminant Res. 94, 132–136. Zarei, M.A., Farshad, A. and Akhondzade, S. 2009. Variations in thyroidal activity during estrous cycle and natural breeding season in Markhoz goat breeds. Pak. J. Biol. Sci. 12, 1420–1424. Zimmer, D.B., Cornwall, E.H., Landar, A. and Song, W. 1995. The S100 protein family: history, function and expression. Brain Res. Bull. 37, 417–429. | ||
| How to Cite this Article |
| Pubmed Style Elgebaly MM, Ibrahim N, Goda NIA, Amin S, Abdallah AA. Influence of photoperiod on testicular activity in rams: Comparative reproductive, hormonal, and histological insights across seasons. Open Vet. J.. 2026; 16(2): 1351-1360. doi:10.5455/OVJ.2026.v16.i2.51 Web Style Elgebaly MM, Ibrahim N, Goda NIA, Amin S, Abdallah AA. Influence of photoperiod on testicular activity in rams: Comparative reproductive, hormonal, and histological insights across seasons. https://www.openveterinaryjournal.com/?mno=301026 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.51 AMA (American Medical Association) Style Elgebaly MM, Ibrahim N, Goda NIA, Amin S, Abdallah AA. Influence of photoperiod on testicular activity in rams: Comparative reproductive, hormonal, and histological insights across seasons. Open Vet. J.. 2026; 16(2): 1351-1360. doi:10.5455/OVJ.2026.v16.i2.51 Vancouver/ICMJE Style Elgebaly MM, Ibrahim N, Goda NIA, Amin S, Abdallah AA. Influence of photoperiod on testicular activity in rams: Comparative reproductive, hormonal, and histological insights across seasons. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 1351-1360. doi:10.5455/OVJ.2026.v16.i2.51 Harvard Style Elgebaly, M. M., Ibrahim, . N., Goda, . N. I. A., Amin, . S. & Abdallah, . A. A. (2026) Influence of photoperiod on testicular activity in rams: Comparative reproductive, hormonal, and histological insights across seasons. Open Vet. J., 16 (2), 1351-1360. doi:10.5455/OVJ.2026.v16.i2.51 Turabian Style Elgebaly, Maha M., Nehal Ibrahim, Nehal I. A. Goda, Said Amin, and Asmaa A. Abdallah. 2026. Influence of photoperiod on testicular activity in rams: Comparative reproductive, hormonal, and histological insights across seasons. Open Veterinary Journal, 16 (2), 1351-1360. doi:10.5455/OVJ.2026.v16.i2.51 Chicago Style Elgebaly, Maha M., Nehal Ibrahim, Nehal I. A. Goda, Said Amin, and Asmaa A. Abdallah. "Influence of photoperiod on testicular activity in rams: Comparative reproductive, hormonal, and histological insights across seasons." Open Veterinary Journal 16 (2026), 1351-1360. doi:10.5455/OVJ.2026.v16.i2.51 MLA (The Modern Language Association) Style Elgebaly, Maha M., Nehal Ibrahim, Nehal I. A. Goda, Said Amin, and Asmaa A. Abdallah. "Influence of photoperiod on testicular activity in rams: Comparative reproductive, hormonal, and histological insights across seasons." Open Veterinary Journal 16.2 (2026), 1351-1360. Print. doi:10.5455/OVJ.2026.v16.i2.51 APA (American Psychological Association) Style Elgebaly, M. M., Ibrahim, . N., Goda, . N. I. A., Amin, . S. & Abdallah, . A. A. (2026) Influence of photoperiod on testicular activity in rams: Comparative reproductive, hormonal, and histological insights across seasons. Open Veterinary Journal, 16 (2), 1351-1360. doi:10.5455/OVJ.2026.v16.i2.51 |