| Review Article | ||
Open Vet. J.. 2026; 16(3): 1426-1437 Open Veterinary Journal, (2026), Vol. 16(3): 1426-1437 Review Article Antioxidant chemistry and its role in mitigating oxidative stress in animalsAhmed Fotouh1, Rania M. Elbatawy2, Ali A. Ghania3,4 and Nady Kh. Elbarbary5*1Department of Pathology and Clinical Pathology, Faculty of Veterinary Medicine, New Valley University, Kharga, Egypt 2Department of Pathology, Faculty of Veterinary Medicine, Benha University, Toukh, Egypt 3Department of Food Science and Technology, Faculty of Agriculture, University of Tripoli, Tripoli, Libya 4Food and Drug Control Center, Tripoli, Libya 5Food Hygiene and Control Department, Faculty of Veterinary Medicine, Aswan University, Aswan, Egypt *Corresponding Author: Nady Kh. Elbarbary. Food Hygiene and Control Department, Faculty of Veterinary Medicine, Aswan University, Aswan, Egypt. Email: nadykhairy [at] vet.aswu.edu.eg Submitted: 18/12/2025 Revised: 15/02/2026 Accepted: 26/02/2026 Published: 31/03/2026 © 2025 Open Veterinary Journal
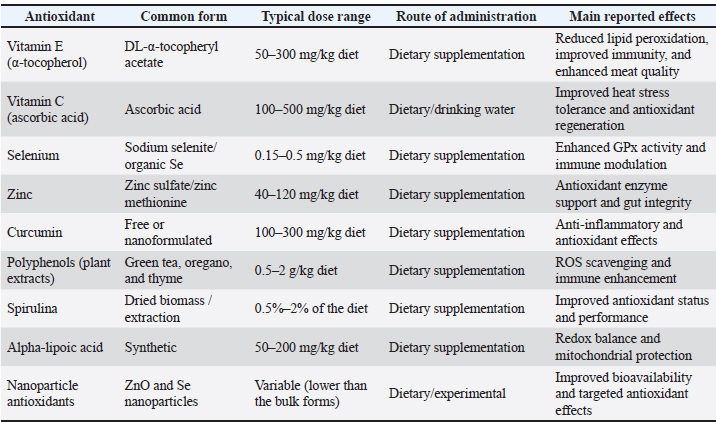
ABSTRACTOxidative stress is a major physiological challenge in poultry and livestock production, arising from an imbalance between reactive oxygen and nitrogen species generation and the antioxidant defense systems of animals. Stressors such as heat, high stocking density, transport, nutritional imbalances, and disease pressure exacerbate oxidative damage under intensive production conditions, leading to impaired immunity, reduced productivity, compromised meat quality, and increased disease susceptibility. Excessive reactive species negatively affect the immune, muscular, reproductive, digestive, and nervous systems. Although animals possess enzymatic and non-enzymatic antioxidant defenses, these mechanisms are often insufficient under high oxidative load, necessitating dietary antioxidant supplementation. Oxidative stress also interacts closely with immune function, inflammatory responses, and thermal stress, particularly heat stress, which intensifies the production of metabolic reactive oxygen species. Dietary antioxidants, including vitamins E and C, selenium, zinc, and plant-derived compounds, have demonstrated protective effects by mitigating oxidative damage, enhancing immune responses, and improving performance and product quality. Advances in oxidative stress biomarkers and analytical techniques have improved assessment accuracy, while emerging strategies, including precision nutrition, nanotechnology-based delivery systems, and regenerative therapies, offer promising avenues for management. Addressing oxidative stress through integrated nutritional, environmental, and therapeutic approaches is essential for sustainable poultry and livestock production. Keywords: Animals, Antioxidants, Chemistry, Oxidative stress. IntroductionHomeostasis, cellular function, and health in the dynamic system of animal physiology depend on the balance between antioxidative and oxidative processes. Oxidative stress is a physiological condition in which an imbalance arises between the production of reactive oxygen species (ROS) and the ability of the biological system to detoxify or neutralize reactive intermediates through its antioxidant defenses. ROS, such as superoxide anion (O₂⁻), hydrogen peroxide (H₂O₂), and hydroxyl radicals (•OH), are typically generated as by-products of normal cell metabolism, particularly from oxidative phosphorylation in the mitochondria (Migdal and Serres, 2011). Although reactive molecules play pivotal roles in cell signaling, immune defense, and homeostasis, excessive accumulation can lead to oxidation and damage to essential cellular components, including lipids, proteins, and Deoxyribonucleic acid (DNA). This disruption can damage cellular functions, induce inflammatory responses, and lead to cell death via apoptosis or necrosis. Oxidative stress is not pathological, but becomes pathological when the redox balance is significantly directed toward oxidants (Chatterjee, 2016). Oxidative stress plays a role in many physiological and pathological processes in animals, including aging, inflammation, reproductive disorders, metabolic disorders, and infections. Therefore, oxidative stress is defined as the overproduction of harmful reactive species and the failure of antioxidant systems to maintain cellular redox balance. Antioxidant chemistry, the study of molecules that can scavenge reactive oxygen species and prevent oxidative damage, has become a core field of animal biology (Schieber and Chandel, 2014). Antioxidants donate electrons to unstable free radicals, stabilizing them to avert chain reactions that lead to cellular damage. The protective agents are universally categorized as enzymatic and non-enzymatic antioxidants. Enzymatic antioxidants, such as Superoxide Dismutase (SOD), Catalase (CAT), and Glutathione Peroxidase (GPx), act synergistically to counteract specific ROS (Ali et al., 2025). Non-enzymatic antioxidants, such as vitamins C and E, carotenoids, flavonoids, and glutathione, are either endogenously biosynthesized or obtained via the diet, and they play complementary roles in free radical scavenging and enzymatic antioxidant recycling (Table 1). In animals, particularly livestock and poultry, the antioxidant system becomes increasingly important under physiological, environmental, or metabolic stress (Mahmoud et al., 2020). Table 1. Antioxidants commonly used in animal and poultry studies, typical dose ranges, and administration routes.
Heat stress, increased metabolic rate, infection, transport, and malnutrition may contribute to ROS generation, leading to an oxidative imbalance. Antioxidant defenses are inadequate in these states, necessitating strategic intervention through dietary supplementation or pharmacologic antioxidant therapy (Aranda-Rivera et al., 2022). Furthermore, oxidative stress has been implicated in reduced reproductive performance, depressed growth rates, impaired immunity, and decreased meat or milk quality; thus, antioxidant management is a significant part of modern animal husbandry and veterinary medicine. The mechanisms by which these molecules act, how their activity is regulated, and how their interactions affect general animal welfare are unearthed in antioxidant chemistry (Ozougwu, 2016). In addition, the development of biomarkers for assessing oxidative stress and antioxidant activity has greatly aided the monitoring and regulation of redox balance in animals. Understanding antioxidant chemistry not only deepens our understanding of animal physiology but also provides valuable tools to enhance animal welfare, productivity, and disease resistance (Amira et al., 2025). Although comprehensive reviews, such as those by Sies et al. (2017) and Celi and Gabai (2015), established the fundamental biochemical and physiological framework of oxidative stress, rapid advances in analytical technologies, biomarker discovery, and antioxidant-based interventions in the last 2 years have necessitated an updated synthesis focused on contemporary animal and poultry production systems. Methodological approachThe literature included in this review was identified through comprehensive searches of major scientific databases, including Scopus, Web of Science, PubMed, and Google Scholar. The search primarily focused on articles published between 2015 and 2025, with particular emphasis on studies from the last 2 years to capture recent advances in oxidative stress biology, antioxidant mechanisms, and emerging therapeutic and technological applications in animal and poultry sciences. Keywords and combinations thereof included oxidative stress, ROS, antioxidants, animal biology, heat stress, immune response, antioxidant supplementation, poultry, and livestock. Additional relevant publications were identified through the manual screening of reference lists from key reviews and original research articles. Studies were selected based on their scientific relevance, methodological quality, and contribution to the understanding of oxidative stress mechanisms and antioxidant strategies in animal systems. Fundamentals of antioxidant chemistry in biological systemsFrom a chemical standpoint, antioxidants are compounds that delay, prevent, or inhibit oxidative damage by participating in redox reactions that neutralize reactive oxygen and nitrogen species. Their activity is governed not only by electron donation but also by redox potential, molecular structure, reaction kinetics, solubility, and interactions within antioxidant networks (Ganesh Yerra et al., 2013). Depending on their molecular configuration and the oxidative environment, antioxidants function through multiple chemical mechanisms, including single-electron transfer (SET), hydrogen atom transfer (HAT), and metal chelation. Lipophilic antioxidants, such as vitamin E, primarily interrupt lipid peroxidation chain reactions by donating hydrogen atoms to lipid radicals, whereas hydrophilic antioxidants, such as vitamin C, mainly operate in aqueous compartments and can regenerate oxidized vitamin E, illustrating the concept of antioxidant network cooperation (Koltover, 2018) Table 2. Table 2. Major classes of antioxidants, chemical mechanisms, and biological roles.
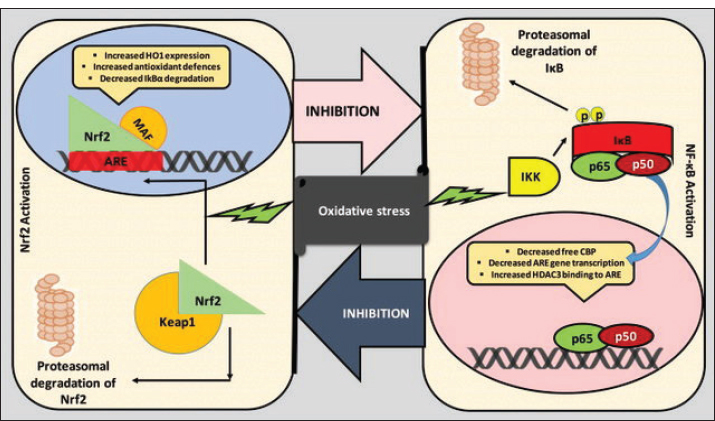
The structure–activity relationship plays a critical role in antioxidant efficacy. For example, phenolic antioxidants derive their activity from hydroxyl group positioning, resonance stabilization of phenoxyl radicals, and steric accessibility. Similarly, carotenoids exhibit singlet oxygen–quenching capacity that depends on the conjugated double-bond length, whereas selenium-containing compounds function through catalytic redox cycling in glutathione peroxidase systems (Suresh et al., 2026). Antioxidant activity is context-dependent, and many antioxidants may exert pro-oxidant effects under specific conditions, such as high concentrations, elevated oxygen tension, or the presence of transition metals (e.g., iron or copper). Under such circumstances, redox cycling can generate rather than neutralize reactive intermediates. This dual behavior underscores the importance of dose, redox environment, and biological compartmentalization when evaluating antioxidant function in vivo (Gulcin, 2025). Reactive species in animal biologyReactive species are unstable molecules that include ROS and reactive nitrogen species (RNS). They possess unpaired electrons, which render them chemically reactive and capable of initiating chain reactions that damage cellular components (Galli et al., 2005). The most abundant ROS are the superoxide anion (O₂⁻), hydrogen peroxide (H₂O₂), hydroxyl radical (•OH), and singlet oxygen (¹O₂). Reactive nitrogen intermediates, such as nitric oxide (NO•) and peroxynitrite (ONOO⁻), are produced in numerous physiological processes and exhibit potent biological activities. Reactive species are considered toxic byproducts of metabolism, but they also have critical functions in cellular signaling, immune defense (e.g., during phagocyte respiratory burst), and homeostatic regulation (Khan et al., 2025). ROS and RNS can modulate gene expression at controlled levels, mediate pathogen responses, and trigger cell growth and apoptosis. However, when their synthesis rates exceed the antioxidant defense system’s capacity, these molecules trigger oxidative or nitrosative stress, leading to cell damage, inflammation, and disease. Therefore, reactive species are double-edged swords within biological systems, acting as needed signaling molecules and potential mediators of cell damage (Sies et al., 2017). Sources of reactive species in animalsReactive species are generated from endogenously (intracellularly) and exogenously (extracellularly) derived sources in animals. Endogenously, ROS are primarily generated in mitochondria, where electron transport chains inadvertently reduce oxygen, generating superoxide radicals during aerobic respiration. Other cellular enzymes, such as nicotinamide adenine dinucleotide phosphate oxidase, xanthine oxidase, and cytochrome P450, generate ROS during normal metabolic reactions. Other intracellular sources include peroxisomes, which function in lipid metabolism, and phagocytic immune cells, which produce reactive oxygen species to combat infectious agents. Nitric oxide synthases produce RNS in the form of nitric oxide during signaling and immune responses (Puppel et al., 2015). Exogenous sources of reactive species include environmental stressors such as ionizing and ultraviolet radiation, exposure to toxic substances and pollutants (e.g., heavy metals, pesticides), and physical stress such as intense exercise or heat/cold exposure. Overcrowding, infection, transportation, or dietary insufficiency in animals can produce ROS and oxidative stress. The relationship between these sources and animal physiology underscores the need for an effective antioxidant defense system to counteract oxidative damage and maintain cellular well-being (Zaki et al., 2025). Balance between oxidative and antioxidant activities in animalsThe balance between antioxidation and oxidation is critical to the physiology of animals and is necessary for cellular homeostasis and health. Animals have evolved an elaborate antioxidant protective system comprising enzymatic antioxidants and non-enzymatic antioxidants (Scialò et al., 2016). These processes detoxify excess ROS, rectify oxidative damage, and restore redox equilibrium. The dynamic equilibrium between antioxidant defense and oxidative activity can be influenced by factors such as age, diet, environmental stress, disease, and genetics. Redox homeostasis is maintained if the antioxidant capacity is sufficient to counteract the ROS burden, and uninjured normal cell processes can ensue (Fotouh et al., 2026). However, a chronic imbalance, whether due to increased ROS production or impaired antioxidant defense, can tip the balance toward oxidative stress, leading to aging, immunosuppression, reduced productivity, and several animal diseases. Therefore, a proper balance between oxidation and antioxidation is essential for animal productivity, health, and resistance and is a significant target for nutrition and management intervention in animal husbandry (Adwas et al., 2019). Oxidative stress and cell signaling pathwaysOxidative stress causes cellular damage and significantly controls cell signaling pathways, affecting all biological processes in animals and birds. ROS, the primary mediators of oxidative stress, are not only harmful metabolic waste products; at low levels, they are essential signaling molecules that control cellular processes such as growth, differentiation, immune function, and apoptosis. The ability of ROS to modulate signaling pathways results from their ability to reversibly oxidize vital molecules, i.e., cysteine residues of proteins, thereby altering protein structure and function (Zhang et al., 2016). One redox-sensitive pathway is the nuclear factor erythroid 2-related factor 1 (Nrf2-Keap1) pathway. Under oxidative stress, ROS activates Keap1, a repressor protein that binds to and promotes Nrf2 degradation under basal conditions (Fig. 1). Keap1 inactivation leads to Nrf2 accumulation, nuclear translocation, and transcriptional activation of antioxidant and cytoprotective genes via antioxidant response elements (AREs) binding. This pathway is essential for initiating an effective defense against oxidative stress by activating detoxifying enzymes such as GPx, SOD, and CAT (Sahu et al., 2015).
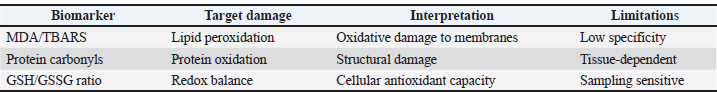
Fig. 1. Crosstalk between the Nrf2 and NF-κB pathways (Ganesh Yerra et al., 2013). Another important pathway that oxidative stress influences is the nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) signaling pathway, which plays a major role in regulating inflammation and immune responses. Low or moderate ROS levels can induce NF-κB activation, leading to the transcription of proinflammatory cytokines, adhesion molecules, and survival genes. Chronic NF-κB activation may result from prolonged oxidative stress, leading to chronic inflammation and tissue damage in animals (Rezatabar et al., 2019). Beyond the classical antioxidant enzyme cascade, the NRF2/KEAP1–HO-1 signaling axis represents a central regulatory node linking oxidative stress to inflammatory modulation in animal systems. Under basal conditions, NRF2 is sequestered in the cytoplasm by Kelch-like ECH-associated protein 1 (KEAP1), which facilitates its ubiquitination and proteasomal degradation (Can et al., 2025). During oxidative stress, modification of cysteine residues within KEAP1 leads to NRF2 stabilization and nuclear translocation, during which NRF2 binds to ARE and promotes transcription of cytoprotective genes, including heme oxygenase-1 (HO-1), glutathione-related enzymes, and phase II detoxifying proteins. HO-1, a downstream effector of NRF2 activation, plays a pivotal role in reducing oxidative injury by degrading heme, generating biliverdin and bilirubin, and modulating redox-sensitive signaling pathways (Sahu et al., 2015). Importantly, increasing evidence demonstrates functional crosstalk between NRF2 and the NF-κB pathway, whereby NRF2 activation suppresses NF-κB–mediated transcription of pro-inflammatory cytokines, while persistent NF-κB activation under chronic oxidative stress may attenuate NRF2 signaling (Puppel et al., 2015). In poultry and livestock, dysregulation of this redox-inflammatory balance contributes to tissue injury, immune dysfunction, and reduced productivity. Furthermore, macrophage activation markers such as CD68 have been associated with oxidative stress–driven inflammatory infiltration in affected tissues, providing histopathological evidence of redox-linked immune activation. Therefore, targeting the NRF2/KEAP1–HO-1 pathway and its interaction with NF-κB represents a promising mechanistic approach for antioxidant-based nutritional and therapeutic interventions in modern animal production systems (Can et al., 2025). Activation of the NRF2–KEAP1–HO-1 signaling pathway constitutes a fundamental adaptive response to oxidative stress in animal cells. Under physiological conditions, NRF2 is retained in the cytoplasm through its interaction with KEAP1, which promotes NRF2 ubiquitination and proteasomal degradation (Mao and Huang, 2017). During oxidative stress, reactive species modify critical cysteine residues on KEAP1, leading to conformational changes that prevent NRF2 degradation. Stabilized NRF2 subsequently translocates to the nucleus, where it binds to AREs in the promoters of cytoprotective genes. HO-1, a key downstream effector, contributes to redox balance by degrading heme and generating antioxidant metabolites, such as biliverdin and bilirubin (Gujarathi et al., 2025). The MAPK signaling pathway is also sensitive to oxidative stress. The pathway includes several kinases, such as Extracellular signal-regulated kinases (ERK), c-Jun amino-terminal kinases, and p38, that transduce extracellular stress signals into cellular responses, including apoptosis, proliferation, and differentiation. ROS may activate MAPKs by altering upstream kinases or phosphatases, thereby driving stress-adaptive responses. Dysregulation of this pathway may result in impaired growth, immune defects, or reproductive disorders in poultry and livestock under chronic oxidative stress (Goyal et al., 2025). Oxidative stress targets the PI3K/Akt pathway, which is involved in cell survival and growth. ROS can activate or inhibit the path, depending on context and oxidative stress levels, thereby affecting insulin signaling, metabolism, and cell survival pathways (Ighodaro, 2018). Antioxidant defense systems in poultry and other animalsThe antioxidant defense system in poultry and other animals is a complex, highly controlled biochemical pathway system that has evolved to counteract ROS to maintain cell redox homeostasis. The system is critical for defending tissues against oxidative stress, which can impair physiological functions, reduce productivity, and increase infection vulnerability (Celi and Gabai, 2015). Antioxidant defenses are broadly categorized into enzymatic and non-enzymatic fractions, which act synergistically. Among the prominent enzymatic antioxidants are SOD, which catalyzes the dismutation of superoxide radicals into hydrogen peroxide; CAT, which further decomposes hydrogen peroxide to water and oxygen; and GPx, which reduces hydrogen peroxide and lipid peroxides with glutathione as a cofactor (Elmeligy et al., 2024). These enzymes are present in numerous tissues, including the liver, kidneys, lungs, and muscles. They are of special importance in highly metabolizing animals, such as poultry, where rapid growth and high feed utilization efficiency activate oxidative metabolism. Non-enzymic antioxidants include endogenous molecules such as glutathione, uric acid, and coenzyme Q, as well as dietary antioxidants such as vitamin E, vitamin C, carotenoids, flavonoids, and trace elements such as selenium and zinc, which supplement antioxidant enzyme activity (Ahmed et al., 2020). Antioxidants in poultry improve growth performance, immunity, and meat and egg quality by preventing lipid peroxidation. Antioxidant supplementation is equally important in ruminants and monogastrics, especially during environmental or metabolic stress such as heat, transport, infection, or excessive production pressure. The antioxidant defense system’s efficiency may be genetically controlled, age-dependent, diet-related, and dependent upon the overall state of health. Hence, promoting antioxidant capacity through dietary means and improved management practices is of utmost concern in animal husbandry today to enhance animal welfare and productivity and prevent oxidative stress-related diseases (Gujarathi et al., 2025). Dietary supplementation of antioxidants in both animals and poultry and therapeutic strategiesDietary supplementation with antioxidants is a key consideration for mitigating the negative effects of oxidative stress by enhancing the health, performance, and disease resistance of poultry and other animals (Salah et al., 2025). Under intensive rearing conditions, animals may be exposed to high stocking density, heat, transport, infection, and poor nutrition, all of which can overwhelm the body’s antioxidant defense mechanisms and increase ROS production. Dietary antioxidants such as vitamin E, vitamin C, selenium, carotenoids, polyphenols, and zinc and manganese are incorporated into animal diets to counteract this. These compounds scavenge free radicals, protect cellular components from oxidative damage, and supplement the activity of endogenous antioxidant enzymes such as SOD, CAT, and GPx (Ugwu et al., 2025). In poultry, antioxidant supplementation increased egg laying, hatchability, immune function, and meat quality by alleviating lipid peroxidation and improving nutrient utilization efficiency (Ponnampalam et al., 2022). Antioxidants also increase fertility, growth rate, immune function, and heat tolerance, and improve resistance to metabolic stress in monogastrics and ruminants. In addition to conventional dietary supplementation, antioxidants are used therapeutically to manage specific health problems. For example, simultaneous administration of vitamins E and C and selenium during heat stress reduces oxidative tissue damage and stabilizes performance (Salami et al., 2016). Antioxidants are administered to improve semen quality, hormonal balance, and embryo viability in both males and females. Antioxidants modulate immune competence and recovery in cases of disease or vaccination by inhibiting inflammation and oxidative burden (Wang et al., 2022). Phytogenic antioxidants derived from plant sources, such as turmeric, oregano, and green tea, possess antimicrobial and antioxidant activity and are used as natural alternatives to antibiotics. The strategic use of dietary antioxidants in relation to physiological requirements, environmental pressures, and target production in animal and poultry nutrition for healthy contributions, improved productivity, and farm system sustainability (Fotouh et al., 2025a,b). Immune system dynamics under oxidative stress in poultry and livestockImmunological function in poultry and other animals is susceptible to oxidative stress, which can profoundly influence the efficacy, effectiveness, and strength of the immune system. Oxidative stress occurs when ROS production outstrips the body’s antioxidant defenses, leading to oxidative damage to immune cells and signaling molecules. Although ROS is a natural part of immune function, notably in the phagocytic killing of microbes during the "respiratory burst," excessive ROS production can be detrimental and compromise immune cell viability, communication, and immunological function (Colitti et al., 2019). Long-term oxidative stress can suppress innate and adaptive immune responses in poultry and livestock. It suppresses the function of macrophages, neutrophils, and natural killer cells, reduces lymphocyte proliferation, and alters cytokine production, thereby undermining the defense mechanisms of animals (Sadek et al., 2024). This leads to increased vulnerability to infections, reduced vaccine efficacy, and susceptibility to diseases such as coccidiosis, respiratory disease, and bacterial or viral epidemics. Oxidative stress consistently induces chronic inflammation, placing additional strain on the immune system and diverting energy away from reproduction and growth (Abu El Hammed et al., 2022). Oxidative damage can be disproportionately harmful in young animals and fast-growing poultry, where the immune system is under development, causing lasting effects on health and performance. Supplementation with antioxidants, such as vitamin E, selenium, vitamin C, and phytochemicals, is a common practice to counteract these effects, increase immune function, reduce inflammation, and enhance disease resistance (Fotouh et al., 2025a,b). These antioxidants maintain immune cell structural integrity, regulate inflammatory processes, and support immune-related enzyme and antibody synthesis. Therefore, the balance of oxidative conditions is crucial for optimal immune function, and antioxidant management is an integral part of health and disease prevention strategies in animal agriculture systems (Li et al., 2024). Physiological and oxidative responses of poultry and animals to thermal stressHeat stress is one of the most stressful environmental conditions affecting poultry and livestock and is closely associated with the development of oxidative stress. When heat-stressed animals are exposed to high ambient temperatures, physiological and behavioral mechanisms compromise their ability to regulate body temperature (Sejian et al., 2018). The resulting thermal imbalance compromises metabolic homeostasis, leading to increased ROS production in tissues, particularly mitochondria. The increased ROS generated under heat stress can overwhelm the antioxidant defense mechanisms of animals, leading to oxidative stress and damage to proteins, lipids, and DNA (Abd-El Hamed et al., 2025). In poultry, thermal stress leads to decreased feed intake, growth performance, egg production, gut integrity, and immunity due to their extremely high metabolic rate and lack of heat loss through sweating (Das et al., 2016). Similarly, heat stress reduces milk yield, reproductive performance, and immunocompetence in ruminants and cattle and increases susceptibility to disease and metabolic disturbances. Heat-induced oxidative stress also damages endocrine function, most notably by disrupting the hypothalamic-pituitary-adrenal axis and leading to hypercortisolism, which further suppresses immunity and productivity (Mahmoud et al., 2025). Furthermore, heat stress compromises intestinal barrier function, potentially leading to endotoxin leakage into the bloodstream and amplifying systemic oxidative and inflammatory reactions. Nutritional therapy with antioxidant supplementation, including vitamin E, vitamin C, selenium, and phytogenic compounds, has proven effective as a countermeasure. These antioxidants scavenge ROS excess, stabilize cell membranes, and support endogenous antioxidant enzyme activities. In addition, management measures, such as augmenting ventilation, using cooling systems, and manipulating feeding times, will help mitigate heat load (Mylostуva et al., 2021). Effect of oxidative stress on meat quality traits in poultry and farm animalsOxidative stress is recognized as a major contributor to variation in meat quality in poultry and farm animals, particularly under conditions of high metabolic demand, environmental stress, and intensive production systems (Cao et al., 2021). In rapidly growing broilers and heavily reared animals, the increased metabolic rate, combined with environmental stressors such as heat, crowding, transport, and nutritional imbalance, leads to increased ROS production. These ROS act on cellular constituents, especially muscle proteins and lipids, leading to protein oxidation, lipid peroxidation, and muscle integrity loss (Tariq et al., 2025). Lipid peroxidation causes rancidity, off-flavor, color change, and off-odor formation, rendering meat less acceptable and reducing shelf life. On the other hand, protein oxidation damages meat tenderness, water-holding capacity, and juiciness by altering muscle fiber composition and enzyme activity. In broilers, oxidative stress has been directly implicated as a causative factor of myopathies, such as white striping, wooden breast, and spaghetti meat, which contribute to loss of meat quality and result in significant economic losses (Ye et al., 2025). Oxidative damage in red meat animals, such as cattle and sheep, may also compromise color stability and tenderness and induce drip loss during storage. Furthermore, postmortem muscle metabolism, including pH decline and enzymatic activity, is also affected by oxidative damage, thereby influencing the development of desirable meat traits (Tumilaar et al., 2024). Antioxidant-supplemented feeding diets, such as vitamin E, selenium, polyphenols, and carotenoids, are widely used to enhance muscle tissue oxidative stability and preserve meat quality. These antioxidants help protect membrane integrity, reduce oxidative damage during muscle development and post-slaughter processing, and improve overall product quality. Antioxidant diets also improve physiological resistance, thereby reducing stress-related muscle breakdown (Morsy et al., 2023). Oxidative stress and its therapeutic targeting in Diabetes mellitusDiabetes mellitus, a chronic metabolic disorder with ongoing hyperglycemia, is typically associated with enhanced oxidative stress, which is believed to be central to the disease’s etiology, pathogenesis, and complications in animal and human models (Newsholme et al., 2016). Hyperglycemia in diabetic conditions leads to ROS overproduction through multiple biochemical mechanisms, including glucose autoxidation, the polyol pathway, protein glycation, and mitochondrial dysfunction. This continuous ROS overproduction overwhelms the endogenous antioxidant defense system, including glutathione (GSH), SOD, and CAT, leading to oxidative damage to vital cellular components, including lipids, proteins, and DNA (Sadek et al., 2024). In animals, diabetes-induced oxidative stress significantly contributes to the dysfunction of pancreatic β-cells and insulin resistance, two hallmarks of the disease. Furthermore, oxidative stress is one of the major driving forces behind the pathogenesis of diabetic complications such as neuropathy, nephropathy, retinopathy, and cardiovascular disease because it creates a state of chronic inflammation, endothelial dysfunction, and target tissue apoptosis (Maiese, 2015). Animal diabetic models have been extensively used to investigate the interaction between oxidative stress and metabolic disturbance, with evidence indicating that antioxidant supplementation, such as vitamins C and E, alpha-lipoic acid, selenium, flavonoids, and polyphenols, reduces oxidative load, improves insulin sensitivity, and protects organs against diabetes-induced damage (Shoker et al., 2025). In veterinary medicine, oxidative stress is also investigated as an etiological factor in insulin resistance and metabolic disease in pet animals (including dogs and cats) and in livestock under stress conditions. Diet, drug, and lifestyle therapy to address oxidative stress is a significant strategy for the overall management of diabetes mellitus, with the promise of slowing disease progression, improving quality of life, and lowering the risk of complications (Yaribeygi et al., 2020). Research tools and biomarkers for studying oxidative stress in poultry and other animalsThe development of specific research tools and biomarkers that enable researchers to assess the extent of oxidative damage and the efficiency of antioxidant defenses has significantly advanced the study of oxidative stress and its impact on animal and poultry health (Demirci-Çekiç et al., 2022). Biomarkers are measurable indicators of biological processes and are used to quantify ROS production, cellular component damage, and the integrity of antioxidant defense mechanisms in the context of oxidative stress (Roba and Umar, 2025) Table 3. Table 3. Common oxidative stress biomarkers and their interpretation.
The commonly used biomarkers include Malondialdehyde and thiobarbituric acid reactive substances (TBARS), which reflect lipid peroxidation, a critical effect of oxidative stress in organ and muscle tissues. Protein carbonyl content and advanced oxidation protein products are used to quantify protein oxidation, while 8-hydroxy-2'-deoxyguanosine is a highly sensitive marker of oxidative DNA damage. The effectiveness of the endogenous antioxidant defense system is quantified using markers such as GSH levels, the GSH/GSSG ratio, and the activities of SOD, CAT, and GPx (Zaki et al., 2025). Modern laboratory tools, such as enzyme-linked immunosorbent assays, spectrophotometric assays, real-time polymerase chain reaction, and western blotting, allow for the precise quantification of such markers (Yang and Lee, 2015). More advanced techniques, such as mass spectrometry and high-performance liquid chromatography, are used to detect oxidative metabolites and profile antioxidant compounds in biological samples. Flow cytometry and fluorescence microscopy are also used to measure ROS levels in specific cell populations (Elbarbary et al., 2025a,b). In addition, the integration of omics science, including genomics, transcriptomics, proteomics, and metabolomics, has enabled a deeper understanding of redox-modulated processes and stress-inducible genes in animals across various environmental and physiological conditions. These biomarkers and research tools can help diagnose oxidative stress, determine the effectiveness of antioxidant supplementation regimens, guide breeding programs aimed at developing stress-resistant animals, and optimize animal welfare and productivity in agricultural systems (Yoshida et al., 2013). Challenges and future perspectives in oxidative stress management in poultry and animalsDespite significant progress in research on oxidative stress and its role in animal and poultry biology, fully controlling its effects and translating basic scientific findings into trusted on-farm benefits remain challenges (Abdelazim and Abomughaid, 2024). One challenge is quantifying oxidative stress in vivo, as oxidative damage varies significantly across tissues, between species, and even during growth or production phases. The availability and affordability of precise biomarkers and advanced diagnostic machinery are also limitations to frequent monitoring in field contexts (Elbarbary et al., 2025a,b). The consistency of antioxidant bioavailability and potency across different environments, diets, and animal species is another issue. For example, some antioxidants are degraded during feed processing or poorly metabolized by the animal, rendering them ineffective. In addition, over supplementation or imbalanced use of synthetic antioxidants may have pro-oxidative effects or disrupt normal redox signaling; thus, dosage and blend calibration is unavoidable (Tumilaar et al., 2024). Furthermore, as animal production systems intensify and animals are subject to increasing climatic and metabolic stressors, the oxidative burden is expected to increase, thereby taking on ever-greater significance in the design of coordinated strategies for redox balance. Future directions include precision nutrition, where antioxidant supplementation is tailored to the specific physiological state, environmental stress exposure, and genotype of the animal (Khairy et al., 2024; Tumilaar et al., 2024). The new science of nutrigenomics, the study of how dietary intake modulates gene expression, holds promise for designing diets that naturally enhance antioxidant defense mechanisms. Dietary antioxidants, naturally occurring in plants, herbs, algae, and plant extracts, are considered environmentally friendly replacements for synthetic additives and meet consumer requirements for clean-label, residue-free animal foods (Singh et al., 2023). In addition, stem cell-derived therapies and nanotechnology-based antioxidant delivery systems show great potential to treat oxidative stress at the cellular level (Denu and Hematti, 2016; Qian et al., 2023). In livestock species, stem cell applications remain largely investigated, with potential relevance to conditions associated with oxidative stress, including impaired immunity, reproductive dysfunction, and tissue degeneration. Recent research has also focused on stem cell–derived extracellular vesicles (exosomes) as a cell-free alternative capable of delivering antioxidant and anti-inflammatory signals with fewer regulatory and safety concerns (Lee et al., 2018). Despite these advances, significant challenges remain, including optimizing delivery methods, addressing long-term safety, managing costs, and addressing species-specific regulatory constraints. Consequently, stem cell–based interventions should be viewed as a future adjunct rather than a replacement for established antioxidant strategies in animal production systems (Fotouh et al., 2025a,b). Integrating omics platforms (genomics, proteomics, and metabolomics) with AI-powered modeling would enable predictive modeling of oxidative stress responses and more effectively guide interventions (Bhatti et al., 2022). Ultimately, a better understanding of oxidative stress and the development of creative, evidence-based solutions will be key to improving animal health, production, and welfare among new agricultural challenges. ConclusionOxidative stress is a critical factor affecting the health, performance, and productivity of poultry and livestock, with wide-ranging impacts on immunity, reproduction, growth, and meat quality. Environmental stressors, nutritional deficiencies, and intensive production systems often intensify this imbalance, leading to cellular and tissue damage. While animals possess natural antioxidant defenses, dietary supplementation with antioxidants such as vitamins E and C, selenium, zinc, and plant-derived compounds has been shown to mitigate oxidative damage and enhance resilience. Advanced research tools and biomarkers have facilitated a deeper understanding of oxidative stress in various biological systems and enabled more effective monitoring. Novel therapeutic strategies, including stem cell therapy and precision antioxidant delivery, offer promising avenues for future intervention. Integrating nutritional, environmental, genetic, and technological strategies will be essential to effectively manage oxidative stress. A comprehensive and sustainable approach will improve the health and welfare of animals and ensure the quality and safety of animal-derived products in modern agriculture. AcknowledgmentsWe acknowledge the valuable help and support from all the staff of the Faculty of Veterinary Medicine, New Valley University, and Aswan University. Conflict of interestNo conflicts of interest were declared. FundingNone. Authors' contributionA. F., R. E., A. G., and N. E. participated in conceiving the investigation idea, preparing the original draft, and preparing the manuscript for publication. The authors have read and approved the revised version of the manuscript. Data availabilityAll data are available from the corresponding author upon reasonable request. ReferencesAbd-El Hamed, A.M.A, Abo-Gamil, Z.H., Elbarbary, N.K., Ghania, A.A., Fotouh, A. and Darweish, M. 2025. A comparative study of performance and profitability measures for broilers raised in open and closed systems: investigating the histopathological effects of heat stress during summer in Egypt. Open Vet. J. 15(5), 2039–2048. Abdelazim, A.M. and Abomughaid, M.M. 2024. Oxidative stress: an overview of past research and future insights. All. Life. 17(1), 2316092. Abu El Hammed, W., Soufy, H., El-Shemy, A., Fotouh, A., Nasr, S. and Dessouky, M. 2022. Prophylactic effect of oregano in chickens experimentally infected with avian pathogenic Escherichia coli O27 with special reference to hematology, serum biochemistry, and histopathology of vital organs. Egypt. J. Chem. 65(6), 269–282. Adwas, A.A., Elsayed, A., Azab, A.E. and Quwaydir, F.A. 2019. Oxidative stress and antioxidant mechanisms in human body. J. Appl. Biotechnol. Bioeng. 6(1), 43–47. Ali, N.M., Hussein, M.K., Elbarbary, N.K., El Amgad, Z. and Noseer, E.A. 2025. Saccharomyces cerevisiae ameliorative impact combined with sulfaclozine on broiler chicken oxidative status. BMC. Vet. Res. 21, 507; doi:10.1186/s12917-025-04955-x Amira, A.M.H., Abo-Gamil, Z.H., Elbarbary, N.K., Ghania, A.A., Fotouh, A. and Darweish, M. 2025. A comparative study of performance and profitability measures for broilers raised in open and closed systems: Investigating the histopathological effects of heat stress during summer in Egypt. Open Vet. J. 15(5), 2039–2048. Aranda-Rivera, A.K., Cruz-Gregorio, A., Arancibia-Hernández, Y.L., Hernández-Cruz, E.Y. and Pedraza-Chaverri, J. 2022. RONS and oxidative stress: an overview of basic concepts. Oxygen 2(4), 437–478. Bhatti, J.S., Sehrawat, A., Mishra, J., Sidhu, I.S., Navik, U., Khullar, N., Kumar, S., Bhatti, G.K. and Reddy, P.H. 2022. Oxidative stress in the pathophysiology of type 2 diabetes and related complications: current therapeutic strategies and future perspectives. Free Radic. Biol. Med. 184, 114–134. Can, I., Guraslan, A., Baser, O.F., Yıldız, G.N., Toplaoglu, I., Aksak Karamese, S. and Karamese, M. 2025. The protective effects of a single dose myricetin application on CLP-induced rat sepsis model by analyzing some immune mechanisms. Immunopharmacol. Immunotoxicol. 47(3), 305–316. Cao, Q.Q., Lin, L.X., Xu, T.T., Lu, Y., Zhang, C.D., Yue, K., Huang, S.C., Dong, H.J. and Jian, F.C. 2021. Aflatoxin B1 alters meat quality associated with oxidative stress, inflammation, and gut microbiota in sheep. Ecotoxicol. Environ. Saf. 225, 112754. Celi, P. and Gabai, G. 2015. Oxidant/antioxidant balance in animal nutrition and health: the role of protein oxidation. Front. Vet. Sci. 2, 48. Chatterjee, S. 2016. Oxidative stress, inflammation, and disease. In: Oxidative stress and biomaterials, Thomas Dziubla, D. and Butterfield, A. Amsterdam, The Netherlands: Elsevier, pp: 35–58; doi: 10.1016/B978-0-12-803269-5.00002-4 Colitti, M., Stefanon, B., Gabai, G., Gelain, M.E. and Bonsembiante, F. 2019. Oxidative stress and nutraceuticals in the modulation of the immune function: current knowledge in animals of veterinary interest. Antioxidants 8(1), 28. Das, R., Sailo, L., Verma, N., Bharti, P., Saikia, J., Imtiwati. and Kumar, R. 2016. Impact of heat stress on health and performance of dairy animals: a review. Vet. World. 9(3), 260. Demirci-Çekiç, S., Özkan, G., Avan, A.N., Uzunboy, S., Çapanoğlu, E. and Apak, R. 2022. Biomarkers of oxidative stress and antioxidant defense. J. Pharm. Biomed. Anal. 209, 114477. Denu, R.A. and Hematti, P. 2016. Effects of oxidative stress on mesenchymal stem cell biology. Oxid. Med. Cell. Longev. 2016, 2989076. Elbarbary, N., Bekhit, M.M., Garh, A., Fotouh, A., Dandrawy, M.K., Darwish, W.S., Abdelmotilib, N.M., Ali, M.A., Salem, M.M. and Abdelhaseib, M. 2025. Meat sarcocystosis: a critical meat-borne parasite impacting carcasses in abattoirs. Ital. J. Food Sci. 37(2), 361–373. Elbarbary, N.K., Gareh, A., Abdelhaseib, M., Fotouh, A., Abdelmotilib, N.M., Ragab, M.F. and Dandrawy, M.K. 2025. Cysticercus bovis in slaughtered cattle in Upper Egypt: implications for food safety. BMC. Vet. Res. 21(1), 1–3. Elmeligy, A., Ghania, A. and Fotouh, A. 2024. Pathological and immunohistochemical studies of lymphoid leukosis in pigeons in Egypt. Open Vet. J. 14(8), 1952. Fotouh, A., Elbarbary, N.K., Momenah, M.A., Khormi, M.A., Mohamed, W.H., Sherkawy, H.S., Ahmed, A.E., Diab, M. and Elshafae, S. 2025a. Hepatoprotective effects of mesenchymal stem cells in CCl₄-induced liver toxicity in rats: restoration of liver enzymes, antioxidants, and histopathological evaluation. Am. J. Vet. Res. 1(aop), 1–10. Fotouh, A., Elbarbary, N.K., Moussa, M.A., Diab, M.S. and Elshafae, S.M. 2025b. Histopathological effects of azithromycin on broilers: immune system alterations and apoptotic changes. Br. Poultry Sci. 17, 1–7; doi:10.1080/00071668.2025.2559391 Fotouh, A., Elsayed, A.K., Zanaty, A.M., Elshafae, S. and Shosha, E.A.E. 2026. Illuminating the pathological, immunohistochemical, and molecular investigations of myelocytomatosis in chicken embryos. Infect. Agents Cancer 21(1), 21. Galli, F., Piroddi, M., Annetti, C., Aisa, C., Floridi, E. and Floridi, A. 2005. Oxidative stress and reactive oxygen species. Contrib. Nephrol. 149, 240–260. Ganesh Yerra, V., Negi, G., Sharma, S.S. and Kumar, A. 2013. Potential therapeutic effects of the simultaneous targeting of the Nrf2 and NF-κB pathways in diabetic neuropathy. Redox. Biol. 1(1), 394–397. Goyal, S., Thirumal, D., Singh, S., Kumar, D., Singh, I., Kumar, G. and Sindhu, R.K., 2025. Basics of antioxidants and their importance. In Antioxidants: Nature's defense against disease. Wiley, pp: 1–20. Gujarathi, N.A., Keservani, R.K., Kesharwani, R.K., Rane, B.R. and Goyal, Y.S. 2025. Antioxidants as nutraceuticals. New York. NY: Apple Academic Press. Taylor & Francis. Gulcin, I. 2025. Antioxidants: a comprehensive review. Arch. Toxicol. 99(5), 1893–1997. Ighodaro, O.M. 2018. Molecular pathways associated with oxidative stress in diabetes mellitus. Biomed. Pharmacother. 108, 656–662. Khairy, N., M. Al-qaaneh, A., M. Bekhit, M., Fotouh, A., S. Madkour, B., M.l. Malak, N., Hadad, G., M. Maher, Z., M. Salem, M. and Abdelhaseib, M. 2024. Advancing meat safety: diverse approaches for bovine tuberculosis detection and control in abattoirs. Ital. J. Food. Sci. 36(4), 240. Khan, I., Hameed, H., Younas, K., Zaman, M., Faheem, S., Majeed, I. and Alvi, M.N. 2025. Unlocking the power of antioxidant nanoparticles: insights into classification, formulation, characterization, and biomedical applications. BioNanoScience 15(1), 131. Koltover, V.K. 2018. Antioxidant biomedicine: from chemistry of free-radicals to reliability of biological systems. Res. Med. Eng. Sci. 3(3), 565. Lee, J., Cho, Y.S., Jung, H. and Choi, I. 2018. Pharmacological regulation of oxidative stress in stem cells. Oxid. Med. Cell. Longev. 2018, 4081890. Li, Y., Liang, J., Jiang, C., Cui, J., Hong, L., Hao, Z., Tang, Y., Liu, Y., Cui, X. and Teng, X. 2024. Selenium alleviated lead-caused neurotoxicity in chickens: sPS2–GPx1–GSH–IL-2/IL-17–NO pathway, selenoprotein suppression, oxidative stress, and inflammatory injury. Antioxidants 13(3), 370. M. Ahmed, O., R. Galaly, S., Raslan, M. and M. A. Mostafa, M.A. 2020. Thyme oil and thymol abrogate doxorubicin-induced nephrotoxicity and cardiotoxicity in Wistar rats via repression of oxidative stress and enhancement of antioxidant defense mechanisms. Biocell 44(1), 41. Mahmoud, M.A.A., Abdel-Kareem, A.H., Abdel-Hafez, M.S., Fotouh, A., Zahran, I.S. and Abdel-Rahman, H.A.R. 2025. Nanoparticles amelioration effects on heat stress in broilers. Assiut Vet. Med. J. 71(185), 314–343. Mahmoud, M.A.M., Megahed, G., Yousef, M.S., Ali, F.A.Z., Zaki, R.S. and Abdelhafeez, H.H. 2020. Salmonella typhimurium–triggered unilateral epididymo-orchitis and splenomegaly in a Holstein bull in Assiut, Egypt: a case report. Pathogens 9(4), 314. Maiese, K. 2015. New insights for oxidative stress and diabetes mellitus. Oxid. Med. Cell. Longev. 2015, 875961. Mao, M. and Huang, M. 2017. Myricetin attenuates lung inflammation and provides protection against lipopolysaccharide-induced acute lung injury by inhibition of NF-κB pathway in rats. Trop. J. Pharm. Res. 16(11), 2585–2593. Migdal, C. and Serres, M. 2011. Reactive oxygen species and oxidative stress. Med. Sci. 27(4), 405–412. Morsy, M.K., Al-Dalain, S.Y., Haddad, M.A., Diab, M., Abd-Elaaty, E.M., Abdeen, A., Ibrahim, S.F., Shukry, M., Banatean-Dunea, I., Fericean, L. and Ghamry, H.I. 2023. Curcumin nanoparticles as a natural antioxidant and antimicrobial preservative against foodborne pathogens in processed chicken fingers. Front. Sustain. Food. Syst. 7, 1267075. Mylostуva, D., Prudnikov, V., Kolisnyk, O., Lykhach, A., Begma, N., Кalinichenko, O., Khmeleva, O., Sanzhara, R., Izhboldina, O. and Mylostyvyi, R. 2021. Biochemical changes during heat stress in productive animals with an emphasis on the antioxidant defense system. J. Anim. Behav. Biometeorol. 10(1), e2209. Newsholme, P., Cruzat, V.F., Keane, K.N., Carlessi, R. and De Bittencourt, P.I.H. 2016. Molecular mechanisms of ROS production and oxidative stress in diabetes. Biochem. J. 473(24), 4527–4550. Ozougwu, J.C. 2016. The role of reactive oxygen species and antioxidants in oxidative stress. Int. J. Res. 1(8), 1–8. Ponnampalam, E.N., Kiani, A., Santhiravel, S., Holman, B.W., Lauridsen, C. and Dunshea, F.R. 2022. The importance of dietary antioxidants on oxidative stress, meat and milk production, and their preservative aspects in farm animals. Animals 12(23), 3279. Puppel, K., Kapusta, A. and Kuczyńska, B. 2015. The etiology of oxidative stress in various animal species: a review. J. Sci. Food. Agric. 95(11), 2179–2184. Qian, H., Shan, Y., Gong, R., Lin, D., Zhang, M., Wang, C. and Wang, L. 2023. Mechanism of action and therapeutic effects of oxidative stress and stem cell-based materials in skin aging. Front. Bioeng. Biotechnol. 10, 1082403. Rezatabar, S., Karimian, A., Rameshknia, V., Parsian, H., Majidinia, M., Kopi, T.A., Bishayee, A., Sadeghinia, A., Yousefi, M., Monirialamdari, M. and Yousefi, B. 2019. RAS/MAPK signaling functions in oxidative stress, DNA damage response and cancer progression. J. Cell. Physiol. 234(9), 14951–14965. Roba, B.B. and Umar, A.B. 2025. Integrated computational analysis of antioxidant compounds: targeting the Keap1–Nrf2 pathway for enhanced therapeutic potential. Prog. Chem. Biochem. Res. 8(3), 278–293. Sadek, A.M., Abdel-Raheem, M.H., AM, A.E., Ibrahim, M.A., Ibrahim, A., Fotouh, A. and Elzoghby, R. 2024. Ameliorative effect of pioglitazone and rosuvastatin on HFD/STZ-induced hepatic injury in rats. Assiut Vet. Med. J. 70(181), 118–132. Sahu, B.D., Kumar, J.M. and Sistla, R. 2015. Baicalein prevents cisplatin-induced acute kidney injury by up-regulating antioxidant defenses and down-regulating MAPKs and NF-κB pathways. PLos One 10(7), 134139. Salah, A.S., El-Tarabany, M.S., Mostafa, M., Zaki, R.S., Azzam, M.M., El Euony, O.I., Alagawany, M., Lestingi, A., Elolimy, A.A., Madkour, M. and Fotouh, A. 2025. Impact of dietary spirulina on performance, antioxidant status, carcass traits and pathological alteration in broilers exposed to ochratoxin A stress. Front. Vet. Sci. 11, 1532353. Salami, S.A., Guinguina, A., Agboola, J.O., Omede, A.A., Agbonlahor, E.M. and Tayyab, U. 2016. In vivo and postmortem effects of feed antioxidants in livestock: implications for authorization of antioxidant feed additives. Animal 10(8), 1375–1390. Schieber, M. and Chandel, N.S. 2014. ROS function in redox signaling and oxidative stress. Curr. Biol. 24(10), R453–R462. Scialò, F., Sriram, A., Fernández-Ayala, D., Gubina, N., Lõhmus, M., Nelson, G., Logan, A., Cooper, H.M., Navas, P., Enríquez, J.A., Murphy, M.P. and Sanz, A. 2016. Mitochondrial ROS produced via reverse electron transport extend animal lifespan. Cell Metab. 23(4), 725–734. Sejian, V., Bhatta, R., Gaughan, J.B., Dunshea, F.R. and Lacetera, N. 2018. Adaptation of animals to heat stress. Animal 12(S2), S431–S444. Shoker, F.E., Elsayed, G.R., Abdelmagid, A.D., Huseiny, H.B. and Fotouh, A. 2025. Amelioration of diabetic nephropathy using ellagic acid-encapsulated zinc nanoparticles through modulation of oxidative stress and inflammatory signaling in rats. Egypt. J. Vet. Sci. 57(6), 269–284. Sies, H., Berndt, C. and Jones, D.P. 2017. Oxidative stress. Annu. Rev. Biochem. 86, 715–748. Singh, N., Sherin, G.R. and Mugesh, G. 2023. Antioxidant and prooxidant nanozymes: from cellular redox regulation to next-generation therapeutics. Angew. Chem. 135(33), e202301232. Suresh, R., Mal, S., Panchal, S. and Karuppasamy, R. 2026. Mechanisms of antioxidant and prooxidant interactions: redox dynamics in plants. In Recent advances in oxidative stress associated chronic diseases. Eds., Kasinathan, K., Muthukumaran, A. Singapore, Springer, 1, pp: 177–98. Tariq, S., Mia, N., Jan, M. and Hashem, M. 2025. Effects of cold storage on meat quality, oxidative stability, and organic antioxidants: a comprehensive overview. Meat Res. 5(4), 1–7. Tumilaar, S.G., Hardianto, A., Dohi, H. and Kurnia, D. 2024. A comprehensive review of free radicals, oxidative stress, and antioxidants. J. Chem. 2024, 5594386. Ugwu, J.C., Ubah, C.B., Lawrence, P., Edim, M.M., N, M.E., Enyike, J.O. and Edet, H.O. 2025. Investigating the antioxidant potential and mechanism of a hydrazide bioactive component of garlic: insights from density functional theory calculations, drug-likeness and molecular docking studies. Appl. Biochem. Biotechnol. 197(2), 847–872. Wang, J., Si, W., Du, Z., Zhang, J. and Xue, M. 2022. Antioxidants in animal feed. Antioxidants 11(9), 1760. Yang, H.Y. and Lee, T.H. 2015. Antioxidant enzymes as redox-based biomarkers: a brief review. BMB. Rep. 48(4), 200. Yaribeygi, H., Sathyapalan, T., Atkin, S.L. and Sahebkar, A. 2020. Molecular mechanisms linking oxidative stress and diabetes mellitus. Oxid. Med. Cell. Longev. 2020, 8609213. Ye, H., Wang, Y., Zhu, H., Huang, C., Wang, W., Jia, Y., Hu, Z., Zhou, H., Liang, S., Ling, C., Zhang, C., Dong, Z. and Zuo, J. 2025. Effects of short-term feeding of resveratrol on growth performance, meat quality, antioxidant capacity, serum biochemical parameters and intestinal health in yellow-feathered broilers under dexamethasone-induced oxidative stress. Antioxidants 14(12), 1459. Yoshida, Y., Umeno, A. and Shichiri, M. 2013. Lipid peroxidation biomarkers for evaluating oxidative stress and assessing antioxidant capacity in vivo. J. clinical bioch. Nutr. 52(1), 9–16. Zaki, R.S., Elbarbary, N.K., Mahmoud, M.A., Bekhit, M.M., Salem, M.M., Darweish, M. and Fotouh, A. 2025. Avian pathogenic Escherichia coli and ostriches: pathological and microbiological investigation. Am. J. Vet. Res. 86(2), 1–10. Zhang, J., Wang, X., Vikash, V., Ye, Q., Wu, D., Liu, Y. and Dong, W. 2016. ROS and ROS-mediated cellular signaling. Oxidative. Med. Cellular. Longevity. 2016, 4350965. | ||
| How to Cite this Article |
| Pubmed Style Fotouh A, Elbatawy RM, Ghania AA, Elbarbary NK. Antioxidant chemistry and its role in mitigating oxidative stress in animals. Open Vet. J.. 2026; 16(3): 1426-1437. doi:10.5455/OVJ.2026.v16.i3.2 Web Style Fotouh A, Elbatawy RM, Ghania AA, Elbarbary NK. Antioxidant chemistry and its role in mitigating oxidative stress in animals. https://www.openveterinaryjournal.com/?mno=303705 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.2 AMA (American Medical Association) Style Fotouh A, Elbatawy RM, Ghania AA, Elbarbary NK. Antioxidant chemistry and its role in mitigating oxidative stress in animals. Open Vet. J.. 2026; 16(3): 1426-1437. doi:10.5455/OVJ.2026.v16.i3.2 Vancouver/ICMJE Style Fotouh A, Elbatawy RM, Ghania AA, Elbarbary NK. Antioxidant chemistry and its role in mitigating oxidative stress in animals. Open Vet. J.. (2026), [cited March 31, 2026]; 16(3): 1426-1437. doi:10.5455/OVJ.2026.v16.i3.2 Harvard Style Fotouh, A., Elbatawy, . R. M., Ghania, . A. A. & Elbarbary, . N. K. (2026) Antioxidant chemistry and its role in mitigating oxidative stress in animals. Open Vet. J., 16 (3), 1426-1437. doi:10.5455/OVJ.2026.v16.i3.2 Turabian Style Fotouh, Ahmed, Rania M. Elbatawy, Ali A. Ghania, and Nady Kh. Elbarbary. 2026. Antioxidant chemistry and its role in mitigating oxidative stress in animals. Open Veterinary Journal, 16 (3), 1426-1437. doi:10.5455/OVJ.2026.v16.i3.2 Chicago Style Fotouh, Ahmed, Rania M. Elbatawy, Ali A. Ghania, and Nady Kh. Elbarbary. "Antioxidant chemistry and its role in mitigating oxidative stress in animals." Open Veterinary Journal 16 (2026), 1426-1437. doi:10.5455/OVJ.2026.v16.i3.2 MLA (The Modern Language Association) Style Fotouh, Ahmed, Rania M. Elbatawy, Ali A. Ghania, and Nady Kh. Elbarbary. "Antioxidant chemistry and its role in mitigating oxidative stress in animals." Open Veterinary Journal 16.3 (2026), 1426-1437. Print. doi:10.5455/OVJ.2026.v16.i3.2 APA (American Psychological Association) Style Fotouh, A., Elbatawy, . R. M., Ghania, . A. A. & Elbarbary, . N. K. (2026) Antioxidant chemistry and its role in mitigating oxidative stress in animals. Open Veterinary Journal, 16 (3), 1426-1437. doi:10.5455/OVJ.2026.v16.i3.2 |