| Original Article | ||
Open Vet J. 2022; 12(5): 754-761 Open Veterinary Journal, (2022), Vol. 12(5): 754–761 Original Research Seroprevalence of Besnoitia besnoiti in Assiut Governorate, EgyptHuda Mohammed Kuraa1*, Zainab M. A. Youssef2, Fatma S. Mahmoud2, and Safaa S. Malek21Parasitology Department, Animal Health Research Institute, Agriculture Research Center, Ministry of Agriculture, Assiut, Egypt 2Department of Animal Medicine, Faculty of Veterinary Medicine, Assiut University, Assiut, Egypt *Corresponding Author: Huda Mohammed Kuraa. Parasitology Department, Animal Health Research Institute, Agriculture Research Center, Ministry of Agriculture, Assiut, Egypt. Email: Huda5380 [at] yahoo.com Submitted: 08/05/2022 Accepted: 18/09/2022 Published: 08/10/2022 © 2022 Open Veterinary Journal
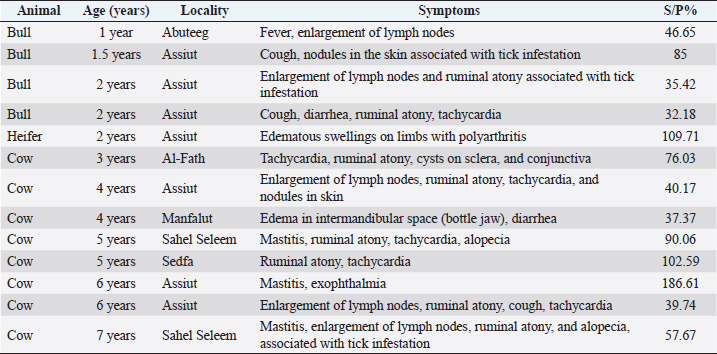
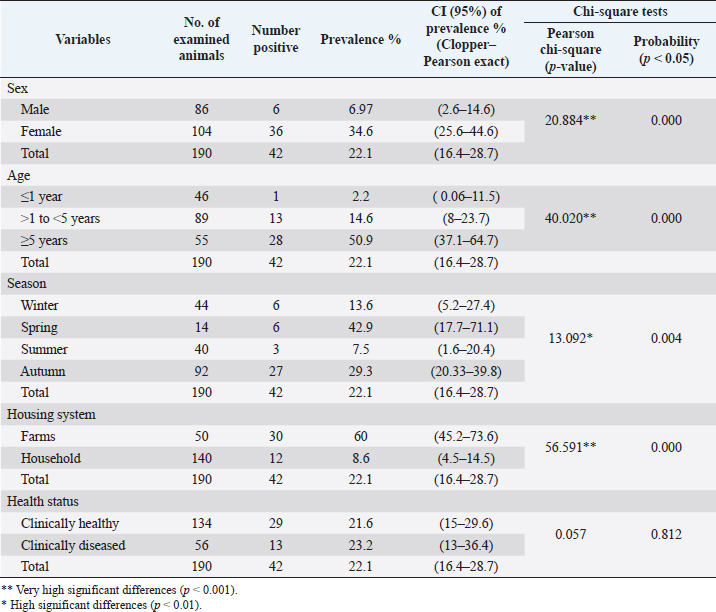
AbstractBackground: Bovine besnoitiosis is a widespread disease caused by Besnoitia besnoiti with significant economic losses in cattle production. There is a lack of knowledge about it in Egypt. Aim: This study was conducted to detect the seroprevalence of B. besnoiti in cattle and to find out the presence of the disease and the most important symptoms of besnoitiosis in cattle in Assiut Governorate, Egypt. Methods: A total of 190 cattle from Assiut city and its different rural centers were examined clinically and serologically for the presence of B. besnoiti. The serological examination was carried out by using the indirect enzyme-linked immunosorbent assay (ELISA) kit in serum (ID.Vet Innovative Diagnostics Louis Pasteur. Grabeis, France). The results were analyzed statistically using the chi-square test to assess the association between seroprevalence and different parameters (age, sex, season, housing, and health status). Result: Thirteen cattle were seropositive for B. besnoiti by ELISA and showed symptoms of besnoitiosis. Acute symptoms included fever, tachycardia, edematous swellings of intermandibular space and limbs with polyarthritis, diarrhea, ruminal atony, and enlarged lymph nodes. The chronic symptoms included cough, mastitis, exophthalmia, cysts on the sclera and conjunctiva, nodules in the skin, and alopecia associated with tick infestation. The overall seroprevalence of B. besnoiti was 22.1%. Regarding sex, the seroprevalence was higher for females 34.6% than for males 6.97%. While, according to age susceptibility, the seroprevalence was highest (50.9%) with age ≥5 years, followed by age >1 to <5 years (14.6%), and only one animal of age ≤1 year was recorded at 2.2%. Concerning seasonal variations, the seroprevalence was highest in spring 42.9%, followed by autumn 29.3%, winter 13.6%, and summer 7.5%. Whereas, according to the housing system, it was 60% and 8.6% in farm and household rearing, respectively. Depending on the health status, the seroprevalence was 21.6% of clinically healthy and 23.2% of clinically diseased cattle. Conclusion: The existence of B. besnoiti antibodies has been demonstrated in clinical and subclinical infected cattle in Assiut Governorate, Egypt. The ELISA test is considered to be a good diagnostic method for detecting infection. Furthermore, additional studies are essential to minimize and prevent the spread of infection. Keywords: Assiut, Besnoitia besnoiti, Cattle, Egypt, ELISA. IntroductionBovine besnoitiosis is a parasitic disease in cattle caused by Besnoitia besnoiti “cyst-forming apicomplexan protozoan” (Besnoit and Robin, 1912). This parasite is closely related to Toxoplasma gondii, Sarcocystis species, and Neospora caninum (Gonzalez-Barrio et al., 2020). It is held to be an emerging disease in European countries and described previously in Africa, the Middle East, and Europe and caused outbreaks in Germany, Switzerland, Italy, and Spain (Mehlhorn et al., 2009; Álvarez-García et al., 2013; Rinaldi et al., 2013; Nieto-Rodríguez et al., 2016). It was endemic in France and Spain (Alzieu et al., 2007a; Fernández-García et al., 2010) and recently reported in Ireland (Ryan et al., 2016). The whole lifecycle of B. besnoiti is not completely known until now with unidentified final host species “carnivores” (Basso et al., 2011). On the other hand, the parasite transmission between cattle was suggested by direct contact between infected and uninfected cattle (nasopharyngeal and natural mating) and mechanically via blood-sucking insects (Stomoxys calcitrans and Tabanus spp.) (Gollnick et al., 2015; Gutiérrez-Expósito et al., 2017a; Tainchum et al., 2018). In addition, Bigalke (1968) discovered that insects, biting flies, and iatrogenically reusing hypodermic needles on numerous animals were transmission methods during the experimental studies. Jacobs et al. (2016) stated that B. besnoiti has an alternating carnivore-herbivore “two host life cycle,” the intestinal phase includes both schizogony and gametogony in the final host. Oocysts are released shortly after their formation and sporulated externally in the environment. During this time, the asexual stages of this parasite progress in the intermediate host. Two infective stages have been defined up to now, one including the fast-replicating tachyzoites and the other including the slower-dividing bradyzoites which unite into macroscopic cysts located within the subcutaneous connective tissue (Bigalke, 1981; Diesing et al., 1988; Njagi et al., 1998). Bovine besnoitiosis is a debilitating disease affecting all cattle ages and breeds. In severe acute cases, it causes anorexia, weakness, pyrexia, nasal and ocular discharge, conjunctivitis, intensive respiratory disorders, increased heart rates, generalized edema, peripheral lymphadenopathy, photophobia, swollen joints, lameness, reduced milk yield, and orchitis associated with bulls’ permanent infertility (Bigalke, 1968, 1981; Álvarez-García et al., 2013; Cortes et al., 2014; Gollnick et al., 2018). Severe chronic disease is characterized by noticeable skin changes with hyperkeratosis, skin wrinkling, alopecia, and non-healing ulcers. In addition, visible tissue cysts were detected in the scleral conjunctiva and genital mucosa (Álvarez-Garcia et al., 2013; Cortes et al., 2014). Chronically infected cattle that do not exhibit severe clinical signs are a reservoir “source of infection.” When these animals are inadvertently integrated into other flocks, besnoitiosis can propagate from one flock to another and can even cross-national boundaries (Álvarez-Garcia et al., 2013). Additionally, the progress of the disease can happen rapidly, and severely impacted animals may die due to respiratory impairment, nephrotic syndrome, or cardio-respiratory failure, which takes place 2 weeks after infection before the development of specific “IgG” antibodies (Dubey et al., 2013; Gonzalez-Barrio et al., 2020). Economic losses take place due to decreased milk and meat production, damage to hides, infertility of bulls, and death (Jacobs et al., 2016). In infected herds, the prevalence rate increased rapidly after the first appearance of the disease, leading to increased epidemicity with a mortality rate of 7%–10% (Fernández-García et al., 2010; Jacquiet et al., 2010; Alzieu and Jacquiet, 2015). In addition, several authors from varying regions of the world recorded that the seroprevalence of bovine besnoitiosis in cattle increased over time (Liénard et al., 2011; Nieto-Rodríguez et al., 2016; Gazzonis et al., 2017; Gutiérrez-Expósito et al., 2017a). Early stages of besnoitiosis are hard to be diagnosed because of non-specific signs and antibodies that are not easily detectable in the first 2 weeks (Jacobs et al., 2016). At the chronic stage, the clinical signs are specific and diagnosis can be proven with an enzyme-linked immunosorbent assay (ELISA) test which is deemed a useful test to detect asymptomatic cattle and aid in the prevention of more propagation of disease via reservoirs and its extirpation from affected flocks for the disease control (Álvarez-Garcia et al., 2013; Jacobs et al., 2016). There is no available published data about the prevalence of besnoitiosis in Egypt except for one study by (Ashmawy and Abu-Akkada, 2014) who examined the seroprevalence of B. besnoiti in bovine. Referring to the high economic importance of besnoitiosis with the current lack of knowledge about it in Egypt, the main purpose of this work was to find out the presence of the disease and the most important symptoms of besnoitiosis in cattle in Assiut Governorate, Egypt. Materials and MethodsAnimalsDuring a period of investigation, from June 2021 to April 2022, a total of 190 cattle of different ages, sex, and localities were examined clinically and serologically for the presence of B. besnoiti. These animals were collected from Assiut city and its different rural centers (Assiut, Manfalut, Al-Qusiya, Abnoub, Sahel Seleem, Al-Fath, Abuteeg, and Sedfa) including different farms and individual cases that were admitted to the Veterinary Teaching Hospital, Faculty of Veterinary Medicine at Assiut University. Clinical examination was carried out according to (Jackson and Cockcroft, 2002). SamplingBlood samples were obtained from 156 clinically healthy and 34 clinically diseased cattle containing 104 females and 86 males. The cattle were selected by age ranging from 4 months to 7 years and divided into three age groups: ≤1 year; >1 to <5 years; and ≥5 years and from farms and household rearing (individual cases that reared in a farmer’s house in the villages). About 5 ml of blood was taken from the jugular vein of cattle into clean, dry glass tubes without anticoagulant and transported to the laboratory in cold conditions. Sera were separated by centrifugation at 500× g for 15 minutes, then labeled and preserved at −20°C until used (Gonzalez-Barrio et al., 2020). Serological examinationThe serum samples were examined for the presence of specific antibodies for B. besnoiti using a commercially available indirect ELISA kit in serum (ID Screen® Besnoitia Indirect 2.0). Indirect biwell ELISA was done for the detection of antibodies against B. besnoiti in bovine serum or plasma (ID.Vet Innovative Diagnostics Louis Pasteur. Grabeis, France; BSNTB ver 0614 GB, LOT: 137) according to the manufacturer’s instructions. The optical density values were read with an ELISA reader (Sunrise, TECAN) at a wavelength of 450 nm within 15 minutes in Molecular Biology Research Center, Assiut University. Antibody analysisFor each sample, calculate the S/P percentage (S/P%) obtained by an equation provided by the manufacturer: Serum samples with an S/P% ≥ 30% were considered positive; 25% > S/P% < 30% were considered doubtful; and those with S/P% ≤ 25% were considered negative. Statistical analysisClopper–Pearson exact confidence intervals (CI) for the prevalence rates were calculated at the level of 95%. The obtained results were analyzed by the chi-square test to assess the association between seroprevalence and different parameters (age, sex, season, housing, and health status) by using Statistical Package for the Social Sciences 22.0 statistical software (IBM Corp., USA). Significance was defined as p < 0.05 (Özdal et al., 2019). Ethical approvalDuring sample taking from animals, adequate measures were taken to minimize the pain or discomfort of animals with concern for ethical standards. ResultsA total of 190 cattle were clinically and serologically examined for the presence of B. besnoiti in some localities of Assiut Governorate, Egypt. Thirteen cattle (4 bulls, 1 heifer, and 8 cows) were seropositive by ELISA and showed symptoms of besnoitiosis. These symptoms involved acute symptoms such as fever, tachycardia, edematous swellings of intermandibular space (bottle jaw), and limbs with polyarthritis, diarrhea, ruminal atony, and enlarged lymph nodes, while the chronic symptoms included cough, mastitis, exophthalmia, cysts on sclera and conjunctiva, nodules in skin, and alopecia associated with tick infestation on some seropositive animals (Table 1 and Fig. 1). The overall seroprevalence of B. besnoiti was 22.1% (42/190) with the ELISA. Concerning sex, females show a higher ratio of 34.6% (36/104) than males, who show a low ratio of 6.97% (6/86). Regarding age, the susceptibility was highest at 50.9% (28/55) with group ≥5 years, followed by group >1 to <5 years at 14.6% (13/89), and only one animal of group ≤1 year was recorded at 2.2% (1/46) (Table 2). Regarding seasonal variations, B. besnoiti was serologically highest in spring 42.9% (6/14), followed by autumn 29.3% (27/92), winter at 13.6% (6/44), and summer at 7.5% (3/40). While the seroprevalence according to the housing system was 60% (30/50) of the farm and 8.6% (12/140) of household rearing. Depending on the health status, it was 21.6% (29/134) of clinically healthy and 23.2% (13/56) of clinically diseased cattle (Table 2). There were highly significant differences in the seroprevalence between different season groups (p < 0.01). Also, very high significant differences in the seroprevalence of infected cattle with B. besnoiti among the sex, age, and housing system were recorded (p < 0.001). However, there were no significant differences in seroprevalence depending on the health status (Table 2). DiscussionBovine besnoitiosis is regarded as responsible for significant economic losses in the cattle industry due to the extensive reduction of productivity, male sterility, abortion, and mortality (Jacobs et al., 2016). The severity of the disease varies greatly between infected cattle (Jacobs et al., 2016). It has been described in numerous countries with varying prevalence rates (Álvarez-Garcia et al., 2013; APHA, 2020; Zhou et al., 2020). Serologically identifying infected cattle is important to preclude the introduction of infected animals into native herds (Papadopoulos et al., 2014). Table 1. Seropositive cattle showing clinical signs of bovine besnoitiosis.
Fig. 1. Clinical signs of acute and chronic B. besnoiti infection in cattle in Assiut. Acute (A and B) as follows: (A): Edematous swelling of the neck. (B): Hyperemia of limbs due to tick infestation. Chronic signs (C–G) as follows: (C): Cysts on the sclera and conjunctiva; (D and E): nodules on skin; and (F and G): alopecia. Clinical symptoms of bovine besnoitiosis in our cases matched typical signs previously summarized by Fernández-García et al. (2010), Álvarez-García et al. (2013), Cortes et al. (2014), Nieto-Rodríguez et al. (2016), Gazzonis et al. (2017), APHA (2020), Gonzalez-Barrio et al. (2020), Zhou et al. (2020), and Neve et al. (2022). Also, Njagi et al. (1998) added that the cattle showing signs of besnoitiosis were heavily infested with ticks in Kenya. The total seroprevalence of B. besnoiti in our study was 22.1%, which was nearly similar to Ashmawy and Abu-Akkada (2014) who found that the seroprevalence of B. besnoiti was 17.13% among cattle in Egypt. In comparison to our result, the same result was recorded in Greece (22%) by Papadopoulos et al. (2014), in the Alentejo region from Southern Portugal (25.8%) by Waap et al. (2014) and in the Kirikkale province of Turkey (26.6%) by Öcal et al. (2016). Whereas, our result was higher than those previously reported in Turkey by Özdal et al. (2019) and Kula and Gokpınar (2021) who found that 2.7% and 5% of cattle were seropositive against B. besnoiti, respectively. On the other hand, the present result was lower than those recorded in France (89%) (Alzieu et al., 2007a), in Italy (44.1%) (Rinaldi et al., 2013), in Navarra, North Spain (90.5% and from 35.6%–86.5%) by Fernández-Garcia et al. (2010) and Gutiérrez-Expósito et al. (2017b), respectively and in Sicily (61.6%) (Neve et al., 2022). Such variations could be due to differences in the study locality, livestock management systems, different levels of contact between infected and uninfected cattle, and exposure to reservoirs and insects (Ashmawy and Abu-Akkada, 2014; Talafha et al., 2015). The current results revealed that the seroprevalence of B. besnoiti was higher in females 34.6% than in males 6.97%. These results are matched with those of Fernández-García et al. (2010) who documented that 90.8% of females and 71.4% of males were seropositive in an outbreak in Spain. In addition, Ashmawy and Abu-Akkada (2014) observed that the prevalence of the disease was higher in females (17.13%) than in males (8.33%), and Özdal et al. (2019) found that the females were infected (3%) without parasite detection in males. On the other hand, Álvarez-Garcia et al. (2013) and Gutiérrez-Expósito et al. (2014) discovered that infection of B. besnoiti in males was more prevalent than in females, and Gazzonis et al. (2017) recorded that males had a higher risk of infection with an incidence of 60% versus 38.8% among females in Italy. Table 2. Seroprevalence of B. besnoiti infection in cattle according to the sex, age susceptibility, seasonal variation, housing system, and health status by using ELISA.
In this study, a higher prevalence of infection in females than in males might be explained by that females may be have increased exposure to infective stages or more susceptible to the parasite. Increased susceptibility in females may occur from the suppressed immune functions related to reproductive stress (Schuurs and Verheul, 1990; Freudiger, 2008; Jacquiet et al., 2010; Ashmawy and Abu-Akkada, 2014; Gazzonis et al., 2017). Besides, the risk of disease spreading to females through mating with an infected bull (Esteban-Gil et al., 2014). Our study revealed that the highest prevalence of B. besnoiti was 50.9% in the age group ≥5 years, followed by the age group >1 to <5 years 14.6%, and only one animal of age group ≤1 year was recorded at 2.2%. This was consistent with results pointed out by Ashmawy and Abu-Akkada (2014) who recorded the highest infection rate of B. besnoiti was 17.13% in the age “5–10 years” followed by the age “1–5 years” at 15.38%, and only one case 1.58% in the age “<1 year.” As well, Özdal et al. (2019) detected that cattle aged “6–8 years” had a higher infection rate of 3.3%, followed by the age group “3–5 years” at 2.8% with no detection of the parasite in the age group “1–2 years.” In addition, previous researchers have found that the risk of infection increases with age increasing (Gutiérrez-Expósito et al., 2014, Talafha et al., 2015; Gazzonis et al., 2017). These results can be attributed to rapid horizontal disease transmission through repeated exposure to parasitic infection either by continuous parasitic replicate within the infected host or by cattle exposure to the insects (Bourdeau et al., 2004; Fernández-García et al., 2010; Ashmawy and Abu-Akkada, 2014). Concerning seasonal variations, the seroprevalence of B. besnoiti was highest in spring 42.9% (6/14), followed by autumn 29.3% (27/92), winter 13.6% (6/44), and summer 7.5% (3/40). Similarly, Alzieu et al. (2007b) and Freudiger (2008) suggested that bovine besnoitiosis was a seasonal infection and were clinically observed from spring to autumn. In view of this, Bigalke (1968) suggested that bloodsucking arthropods such as horseflies act as mechanical vectors of Besnoitia. Spring and autumn might be accompanied by increased insect activity and parasite contact (Gollnick et al., 2018). Our climate can encourage the development and prolong the activity period of vectors “biting insects,” which facilitate the rapid propagation of the parasite through vector-borne horizontal transmission (Bigalke, 1968), in agreement with Jacquiet et al. (2010) who found that climatic changes may have helped the spread of the parasite. In contrast with Fernández-García et al. (2010) who discovered the appearance of clinical signs coincides with the summer season, when herds shared pastures and there are vectors. This study revealed that the seroprevalence of B. besnoiti according to the housing system was 60% of farm and 8.6% of household rearing. The high infection in farms can be explained by continuous exposure of animals to infection. This accords with Gazzonis et al. (2017) who stated that natural mating could be considered a possible method of B. besnoiti diffusion through the herd and insect-bite-promoted infection passage. Likewise, direct contact between infected and non-infected animals may aid nasopharyngeal transmission. Therefore, a minimum distance of 20 m between infected and uninfected animals was recently proved sufficient to avoid seroconversion of negative cows (Esteban-Gil et al., 2014; Gollnick et al., 2015). It has been reported that animals without clinical signs in endemic regions have been reported to be an important factor in the dissemination of the disease (Frey et al., 2013). Even in endemic situations, only some animals develop the characteristic clinical symptom of besnoitiosis in an infected herd, while the majority are seropositive but still subclinically infected. The recognition of subclinical cases through the ELISA test can be useful in carrying out control programs (Fernández-Garcia et al., 2010; García-Lunar et al., 2013). Culling would have been the best way to control cattle besnoitiosis on farms (Zhou et al., 2020). In our study, the seroprevalence of B. besnoiti according to the health status was 21.6% of clinically healthy cattle, while it was 23.2% of diseased cattle. This is in agreement with Fernández-García et al. (2010) who registered that only 43% developed clinical signs in 90.5% of seropositive cattle in Spain. This is contrary to Neve et al. (2022) who found that clinically diseased cattle had a low prevalence (1.5%) and confirmed that the clinical appearance of bovine besnoitiosis remains sporadic. These differences in the prevalence of clinically diseased cattle may be due to various factors such as stress, the resistance of the animal, and the amount of parasites transmitted to animals by blood-sucking insects (Alzieu et al., 2007b). ConclusionBased on our findings, the existence of B. besnoiti antibodies has been demonstrated in clinical and subclinical infected cattle in Assiut Governorate, Egypt. The ELISA test is considered to be a good diagnostic method for detecting B. besnoiti infection. Furthermore, it is a major need to raise veterinarians’ awareness of the clinical cases and methods of disease spread. Moreover, additional studies are essential to developing specific programs to combat this disease and minimize the spread of infection and prevent the entry of infected animals into herds. Conflict of interestThe authors declare that there is no conflict of interest. Authors’ contributionKuraa, H.M., Youssef, Z.M.A., Mahmoud, F.S., and Malek, S.S. designed the study and helped in the ELISA procedure, data analysis, and interpretation. Youssef, Z.M.A., Mahmoud F.S., and Malek, S.S. examined animals and collected samples. Kuraa, H.M. wrote the manuscript. ReferencesÁlvarez-García, G., Frey, C.F., Ortega-Mora, L.M. and Schares, G. 2013. A century of bovine besnoitiosis: an unknown disease re-emerging in Europe. Trends Parasitol. 29, 407–415. Alzieu, J.P. and Jacquiet, P. 2015. Bovine besnoitiosis: from emergence to the need of control [Besnoitiosi bovina: dall'emergenza alla necessita del controllo]. Summa Anim. Reddito 10, 19–27. Alzieu, J.P., Dorchies, P., Schelcher, F. and Gottstein, B. 2007a. L’extension de la besnoitiose bovine en France. Le Point Vét. 276, 37–43. Alzieu, J.P., Cortes, H., Gottstein, B., Jacquiet, P., Dorchies, P., Schelcher, F. and L’Hostis, M.B. 2007b. La besnoitiose bovine: actualités épidémiologiques et diagnostiques. Bulletin des GTV Hors-série parasitisme des bovins, pp: 41–49. APHA. 2020. Seroprevalence survey for Besnoitia besnoiti in cattle and buffalo imported to mainland UK from Europe in 2018. Animal and Plant Health Agency. Available via www.gov.uk/government/publications Ashmawy, K.I. and Abu-Akkada, S.S. 2014. Evidence for bovine besnoitiosis in Egypt—first serosurvey of Besnoitia besnoiti in cattle and water buffalo (Bubalus bubalis) in Egypt. Trop. Anim. Health Prod. 46, 519–522. Basso, W., Schares, G., Gollnick, N.S., Rütten, M. and Deplazes, P. 2011. Exploring the life cycle of Besnoitia besnoiti—experimental infection of putative definitive and intermediate host species. Vet. Parasitol. 178, 223–234. Besnoit, C. and Robin, V. 1912. Sarcosporidiose cutanée chez une vache. Rev. Vét. 37, 649–663. Bigalke, R.D. 1968. New concepts on the epidemiological features of bovine besnoitiosis as determined by laboratory and field investigations. Onderstepoort J. Vet. Res. 35(1), 3–137. Bigalke, R.D. 1981. Besnoitiosis and globidiosis. In Diseases of cattle in the tropics. Current topics in veterinary medicine and animal science, 6. Eds., Ristic, M. and McIntyre, I. Dordrecht/The Hague, The Netherlands: Springer/Marunus Nijhoff, pp: 429–442. Bourdeau, P.J., Cesbron, N., Alexandre, F., Marchand, A.M., Desvaux, J.P. and Douart, A. Outbreak of bovine besnoitiosis, Besnoitia besnoiti in the west of France and its diagnosis by immunofluorescence assay. In Proceedings of the 9th European Multicolloquium of Parasitology, Valencia, Spain, 2004, pp: 459–460. Cortes, H., Leitão, A., Gottstein, B. and Hemphill, A. 2014. A review on bovine besnoitiosis: a disease with economic impact in herd health management, caused by Besnoitia besnoiti (Franco and Borges, 1916). Parasitology 141, 1406–1417. Diesing, L., Heydorn, A.O., Matuschka, F.R., Bauer, C., Pipano, E., de Waal, D.T. and Potgieter, F.T. 1988. Besnoitia besnoiti: studies on the definitive host and experimental infections in cattle. Parasitol. Res. 75, 114–117. Dubey, J.P., van Wilpe, E., Blignaut, D.J.C., Schares, G. and Williams, J.H. 2013. Development of early tissue cysts and associated pathology of Besnoitia besnoiti in a naturally infected bull (Bos taurus) from South Africa. J. Parasitol. 99, 459–466. Esteban-Gil, A., Grisez, C., Prevot, F., Florentin, S., Decaudin, A., Picard- Hagen, N., Berthelot, X., Ronsin, P., Alzieu, J.P., Marois, M., Corboz, N., Peglion, M., Vilardell, C., Liénard, E., Bouhsira, E., Castillo, J.A., Franc, M. and Jacquiet, P. 2014. No detection of Besnoitia besnoiti DNA in the semen of chronically infected bulls. Parasitol. Res. 113, 2355–2362. Fernández-García, A., Álvarez-García, G., Risco-Castillo, V., Aguado-Martínez, A., Marcén, J.M., Rojo-Montejo, S., Castillo, J.A. and Ortega-Mora, L.M. 2010. Development and use of an indirect ELISA in an outbreak of bovine besnoitiosis in Spain. Vet. Rec. 166, 818–822. Freudiger, I. 2008. La besnoitiose bovine: étude épidémiologique de l’épizootie des Alpes de-Haute-Provence et des Hautes-Alpes. Doctoral Thesis, Lyon, France, pp: 29–78. Frey, C.F., Gutiérrez-Expósito, D., Ortega-Mora, L.M., Benavides, J., Marcén, J.M., Castillo, J.A., Casasús, I., Sanz, A., García-Lunar, P., Esteban-Gil, A. and Álvarez-García, G. 2013. Chronic bovine besnoitiosis: intra-organ parasite distribution, parasite loads and parasite-associated lesions in subclinical cases. Vet. Parasitol. 197, 95–103. García-Lunar, P., Ortega-Mora, L.M., Schares, G., Gollnick, N.S., Jacquiet, P., Grisez, C., Prevot, F., Frey, C.F., Gottstein, B. and Álvarez-García, G. 2013. An inter-laboratory comparative study of serological tools employed in the diagnosis of Besnoitia besnoiti infection in bovines. Transbound. Emerg. Dis. 60, 59–68. Gazzonis, A.L., Álvarez-Garcia, G., Maggioni, A., Zanzani, S.A., Olivieri, E., Compiani, R., Sironi, G., Ortega-Mora, L.M. and Manfredi, M.T. 2017. Serological dynamics and risk factors of Besnoitia besnoiti infection in breeding bulls from an endemically infected purebred beef herd. Parasitol. Res. 116, 1383–1393. Gollnick, N.S., Scharr, J.C., Schares, G. and Langenmayer, M.C. 2015. Natural Besnoitia besnoiti infections in cattle: chronology of disease progression. BMC Vet. Res. 11, 35. Gollnick, N.S., Scharr, J.C., Schares, S., Bärwald, A., Schares, G. and Langenmayer, M.C. 2018. Naturally acquired bovine besnoitiosis: Disease frequency, risk and outcome in an endemically infected beef herd. Transbound. Emerg. Dis. 65, 833–843. Gonzalez-Barrio, D., Diezma-Diaz, C., Tabanera, E., Aguado-Criado, E., Pizarro, M., Gonzalez-Huecas, M., Ferre, I., Jimenez-Melendez, A., Criado, F., Gutierrez-Exposito, D., Ortega-Mora, L.M. and Álvarez-Garcia, G. 2020. Vascular wall injury and inflammation are key pathogenic mechanisms responsible for early testicular degeneration during acute besnoitiosis in bulls. Parasite. Vectors 13, 113. Gutiérrez-Expósito, D., Esteban-Gil, A., Ortega-Mora, L.M., García-Lunar, P., Castillo, J.A., Marcén, J.M. and Álvarez-García, G. 2014. Prevalence of Besnoitia besnoiti infection in beef cattle from the Spanish Pyrenees. Vet. J. 200, 468–470. Gutiérrez-Expósito, D., Ferre, I., Ortega-Mora, L.M. and Álvarez-García, G. 2017a. Advances in the diagnosis of bovine besnoitiosis: current options and applications for control. Int. J. Parasitol. 47, 737–751. Gutiérrez-Expósito, D., Ortega-Mora, L.M., García-Lunar, P., Rojo-Montejo, S., Zabala, J., Serrano, M. and Álvarez-García, G. 2017b. Clinical and serological dynamics of Besnoitia besnoiti infection in three endemically infected beef cattle herds. Transbound. Emerg. Dis. 64, 538–546. Jackson, P.G.G. and Cockcroft, P.D. 2002. Clinical examination of farm animals. Oxford, UK: Blackwell Publishing Company, pp: 9–28. Jacquiet, P., Liénard, E. and Franc, M. 2010. Bovine besnoitiosis: epidemiological and clinical aspects. Vet. Parasitol. 174, 30–36. Jacobs, D.E., Fox, M., Gibbons, L.M. and Hermosilla, C. 2016. Principles of veterinary parasitology. Chichester, UK; Hoboken, NJ: John Wiley & Sons, Ltd., pp: 240–259. Kula, D. and Gökpınar, S. 2021. Seroprevalence of Neospora caninum and Besnoitia besnoiti in Cattle in Oğuzlar Region. Turk. Parazitol. Derg. 45, 108–112. Liénard, E., Salem, A., Grisez, C., Prévot, F., Bergeaud, J.P., Franc, M., Gottstein, B., Alzieu, J.P., Lagalisse, Y. and Jacquiet, P. 2011. A longitudinal study of Besnoitia besnoiti infections and seasonal abundance of Stomoxys calcitrans in a dairy cattle farm of southwest France. Vet. Parasitol. 177, 20–27. Mehlhorn, H., Klimpel, S., Schein, E., Heydorn, A.O., Al-Quraishy, S. and Selmair, J. 2009. Another African disease in Central Europe: Besnoitiosis of cattle.I. Light and electron microscopical study. Parasitol. Res. 104, 861–868. Neve, V.C., Coltraro, M., Stamilla, A., Spadola, F., Puleio, R., Loria, G.R., Antoci, F., Cascone, G. and Salina, F. 2022. Investigation of an autochthonous outbreak of bovine besnoitiosis in Northwestern Sicily. Pathogens 11, 122. Nieto-Rodríguez, J.M., Calero-Bernal, R., Álvarez-García, G., Gutiérrez-Expósito, D., Redondo-García, E., Fernández-García, J.L. and Martínez-Estéllez, M.Á.H. 2016. Characterization of an outbreak of emerging bovine besnoitiosis in southwestern Spain. Parasitol. Res. 115, 2887–2892. Njagi, O.N., Ndarathi, C.M., Nyaga, P.N. and Munga, L.K. 1998. An epidemic of Besnoitiosis in cattle in Kenya. Onderstepoort J. Vet. Res. 65, 133–136. Öcal, N., Yağci, B.B. and Gökpinar, S.. Investigation as clinical and laboratory of besnoitiosis in cattle. 1st International Congress on Advances in Veterinary Sciences and Technics (ICAVST), 2016 Augt 25–29, Sarajevo, Bosnia and Herzegovina, pp: 109. Özdal, N., Oğuz, B., Orunç Kılınç, Ö., Karakuş, A. and Değer, S. 2019. Prevalence of ELISA-detected specific antibodies against Besnoitia besnoiti in cattle of the Eastern and Southeastern Anatolian regions, Turkey. Iran. J. Vet. Res., 20, 143–146. Papadopoulos, E., Arsenos, G., Ptochos, S., Katsoulos, P., Oikonomou, G., Karatzia, M.A. and Karatzias, H. 2014. First report of Besnoitia besnoiti seropositive cattle in Greece. J. Hell. Vet. Medical Soc. 65, 115–120. Rinaldi, L., Maurelli, M.P., Musella, V., Bosco, A., Cortes, H. and Cringol, G. 2013. First cross-sectional serological survey on Besnoitia besnoiti in cattle in Italy. Parasitol. Res. 112, 1805–1807. Ryan, E.G., Lee, A., Carty, C., O’Shaughnessy, J., Kelly, P., Cassidy, J.P., Sheehan, M., Johnson, A. and de Waal, T. 2016. Bovine besnoitiosis (Besnoitia besnoiti) in an Irish dairy herd. Vet. Rec. 178, 608. Schuurs, A.H. and Verheul, H.A. 1990. Effect of gender and sex steroids on the immune response. J. Steroid Biochem. 35, 157–172. Tainchum, K., Shukri, S., Duvallet, G., Etienne, L. and Jacquiet, P. 2018. Phenotypic susceptibility to pyrethroids and organop K. hosphate of wild Stomoxys calcitrans (Diptera: Muscidae) populations in southwestern France. Parasitol. Res. 117, 4027–4032. Talafha, A.Q., Al-Majali, A.M., Ababneh, M.M. and Abutarbush, S.M. 2015. Epidemiologic study on Besnoitia besnoiti infection in dairy herds in Jordan. Parasitol. Res. 114, 2491–2497. Waap, H., Nunes, T., Cortes, H., Leitão, A. and Vaz, Y. 2014. Prevalence and geographic distribution of Besnoitia besnoiti infection in cattle herds in Portugal. Parasitol. Res. 113, 3703-3711. Zhou, E., Silva, L.M.R., Conejeros, I., Velásquez, Z.D., Hirz, M., Gärtner, U., Jacquiet, P., Taubert, A. and Hermosilla, C. 2020. Besnoitia besnoiti bradyzoite stages induce suicidal-and rapid vital-NETosis. Parasitology 147, 401–409. | ||
| How to Cite this Article |
| Pubmed Style Kuraa HM, Youssef ZMA, Magied FSMA, Malek SS. Seroprevalence of Besnoitia besnoiti in Assiut governorate, Egypt. Open Vet J. 2022; 12(5): 754-761. doi:10.5455/OVJ.2022.v12.i5.21 Web Style Kuraa HM, Youssef ZMA, Magied FSMA, Malek SS. Seroprevalence of Besnoitia besnoiti in Assiut governorate, Egypt. https://www.openveterinaryjournal.com/?mno=32099 [Access: July 01, 2025]. doi:10.5455/OVJ.2022.v12.i5.21 AMA (American Medical Association) Style Kuraa HM, Youssef ZMA, Magied FSMA, Malek SS. Seroprevalence of Besnoitia besnoiti in Assiut governorate, Egypt. Open Vet J. 2022; 12(5): 754-761. doi:10.5455/OVJ.2022.v12.i5.21 Vancouver/ICMJE Style Kuraa HM, Youssef ZMA, Magied FSMA, Malek SS. Seroprevalence of Besnoitia besnoiti in Assiut governorate, Egypt. Open Vet J. (2022), [cited July 01, 2025]; 12(5): 754-761. doi:10.5455/OVJ.2022.v12.i5.21 Harvard Style Kuraa, H. M., Youssef, . Z. M. A., Magied, . F. S. M. A. & Malek, . S. S. (2022) Seroprevalence of Besnoitia besnoiti in Assiut governorate, Egypt. Open Vet J, 12 (5), 754-761. doi:10.5455/OVJ.2022.v12.i5.21 Turabian Style Kuraa, Huda Mohamed, Zainab Mohammed Ahmed Youssef, Fatma Saber Mahmoud Abdel Magied, and Safaa Said Malek. 2022. Seroprevalence of Besnoitia besnoiti in Assiut governorate, Egypt. Open Veterinary Journal, 12 (5), 754-761. doi:10.5455/OVJ.2022.v12.i5.21 Chicago Style Kuraa, Huda Mohamed, Zainab Mohammed Ahmed Youssef, Fatma Saber Mahmoud Abdel Magied, and Safaa Said Malek. "Seroprevalence of Besnoitia besnoiti in Assiut governorate, Egypt." Open Veterinary Journal 12 (2022), 754-761. doi:10.5455/OVJ.2022.v12.i5.21 MLA (The Modern Language Association) Style Kuraa, Huda Mohamed, Zainab Mohammed Ahmed Youssef, Fatma Saber Mahmoud Abdel Magied, and Safaa Said Malek. "Seroprevalence of Besnoitia besnoiti in Assiut governorate, Egypt." Open Veterinary Journal 12.5 (2022), 754-761. Print. doi:10.5455/OVJ.2022.v12.i5.21 APA (American Psychological Association) Style Kuraa, H. M., Youssef, . Z. M. A., Magied, . F. S. M. A. & Malek, . S. S. (2022) Seroprevalence of Besnoitia besnoiti in Assiut governorate, Egypt. Open Veterinary Journal, 12 (5), 754-761. doi:10.5455/OVJ.2022.v12.i5.21 |