| Research Article | ||
Open Vet. J.. 2025; 15(2): 939-953 Open Veterinary Journal, (2025), Vol. 15(2): 939-953 Research Article Ultrastructural and developmental anatomy of the peripheral olfactory organs of Dicentrarchus labrax inhabiting Egyptian Mediterranean waterSheren A. Al-Zahaby*, Al-Ahmady S. Al-Zahaby, Radwa A. Mohamed and Eman H. ElsheikhDepartment of Zoology, Faculty of Science, Zagazig University, Zagazig, Egypt *Corresponding Author: Sheren A. Al-Zahaby. Department of Zoology, Faculty of Science, Zagazig University, Zagazig, Egypt. Email: shalahmady [at] yahoo.com Submitted: 30/12/2024 Accepted: 07/02/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
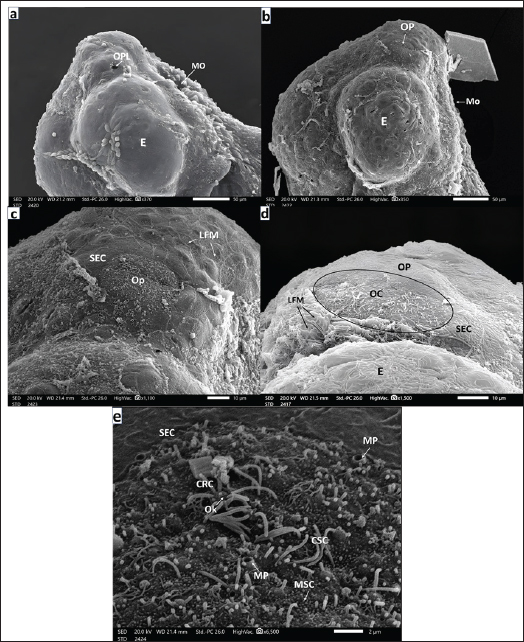
AbstractBackground: Olfaction regulates animal behavior; hence, its organs are present early in fish development. Due to their aquatic lifestyle, fish use olfaction to communicate chemical signals during gustation. Aim: In this study, we investigated the morphogenesis and ontogeny of the European seabass’s ( Dicentrarchus labrax, Linnaeus, 1758) olfactory organ. Methods: Scanning electron microscopy was used to study the development of these organs in fries, fingerlings, juveniles, and adults. Results: Both placodes were thickened and invaginated, generating simple olfactory pits with undifferentiated cells. The non-sensory and sensory olfactory cells (OC) emerged after the first day of hatching. In addition to occasional mucous goblet cells, ciliated receptor cells are more common than microvillous receptor cells. Long, thin, disordered kinocilium and small microvilli of ciliated and microvillous supporting cells cover some of the pit floor. The SEC-lined pit bottom deepens 45 days after hatching. Fries develop tube-shaped anterior incurrents and broader posterior excurrent nostrils by the 75th day post-hatching, along with a broad epidermal nasal bridge. In this stage and after fingerling formation at 90 days post-hatching, the bottom olfactory epithelium lining each chamber forms a multilamellar rosette of lamellae that radiates equally from the median raphe. Fully mature European seabass have 38–40 lamellae. The pseudostratified epithelia surrounding each lamella are sensory and nonsensory. The first one, scattered in islets between the second, has receptor (ciliated, microvillous, and rod-tipped) and supporting (ciliated and microvillar) cells, whereas the nonsensory epithelium has ciliated and microvillar supporting cells and masses of superficial epidermal cells. Goblet mucous cells and basal stem cells on the epithelium’s basal lamina are abundant in nonsensory areas but rare in sensory parts. Conclusion: These findings underscore the functional specialization of OCs, reflecting the organ’s pivotal role in chemical communication and environmental adaptation throughout the European seabass’ development. Keywords: Olfactory ontogeny, Olfactory placode, Olfactory receptor cells, Olfactory supporting cells. IntroductionThe European seabass Dicentrarchus labrax (Linnaeus, 1758) is a member of the Moronidae family (Yamamoto and Ueda, 1979). Nelson et al. (2016) classified the European seabass as part of the Moroniformes order. It is economically significant in most European and Mediterranean nations, including Egypt, where aquaculture is widely utilized. European seabass aquaculture is vital because of its role in raising global demand. It supports food security and exports while providing a rich protein and omega-3 fatty acid source (El-Shebly, 2009). Since the 1990s, aquaculture production has surpassed fisheries (FAO, 2018). This approach has been primarily realized in the Mediterranean basin (Vandeputte et al., 2019). European seabass is a demersal fish that lives in marine to slightly brackish water as a coastal migratory fish; in Egypt, it is locally known as “karous” (Mehanna, 2006). The European seabass has a slightly compressed, elongated body covered with small, rough, bony scales (Moretti et al., 1999). The feeding behavior of European seabass is related to their developmental stage, as the earlier larval stages rely on endogenous feeding (Diaz et al., 2002). As European seabass develop, they transition to exogenous feeding (Miller and Kendall, 2009). In contrast, larval stages feed on phyto- and zooplankton, while juveniles graze small crustaceans, and adults eat shrimp (Moretti et al., 1999). As a demersal fish, Dicentrarchus labrax hunts at any time of day, capturing both small and large varieties of invertebrates or fish that are abundant in the area. As it grows larger, it undergoes an ontogenetic shift to become a piscivory. As it grows in size, it loses its dominance over invertebrates. It is an opportunistic predator (Vandeputte et al., 2019). During the breeding season, Dicentrarchus labrax moves long distances to mate, eat, and spend the winter. Larvae drift ashore to nursery areas in lagoons and shallow bays for growth (Khedkar et al., 2003). The Mediterranean European seabass (Dicentrarchus labrax L.) spawn once a year, mainly in shallow coastal waters, with the breeding season typically occurring from December to March (Vinagre et al., 2009). Spawning is influenced by water temperature (8°C–15°C) and photoperiod, as eggs are rarely found where the water is colder than 8.0°C or warmer than 15°C (Moretti et al., 1999). In a wild environment, female fish complete maturation at the same time, and eggs are released at once. In the sea, females lay small pelagic eggs in the littoral areas and river estuaries in high saline seawater (approximately 30 ppm). These planktonic egg hatch between 4 and 6 days after fertilization (Beraud et al., 2018). The young are typically observed in groups, whereas adults tend to be more solitary (Watson et al., 2024). One chemical sense is olfaction, which is crucial for controlling behavior, especially in dark water. Along with gustation, olfaction is thought to be the main pathway by which fish send chemical signals (Hara, 1975). The distinction between these two sensory modalities, including olfaction and gustation, in fish is not always as distinctive as that in air-breathing vertebrates (Hara, 1975). The distinction between these two sensory modalities, including olfaction and gustation, in fish is not always as distinctive as that in air-breathing vertebrates (Hara, 1975). Olfaction is a long-distance sensory system that is crucial for processes such as foraging, coordination, social interactions, sex recognition, mating, and migration. Different actinopterygian species have different bodies and structures (Deng and Demisse, 2023), and their jobs may change at different stages of development depending on how the fish eat (Tytiuk et al., 2020). The outer parts of the olfactory system, such as the paired olfactory rosettes, are in two chambers dorsal to the mouth. The incurrent and excurrent nostrils connect these chambers to the outside environment (Hamdani and Døving, 2007). They are derived from ventrolateral olfactory placodal cells separated from the anterior neural plate (Hara, 2011). The lamellae are arranged on the median raphe (MR); they differ in their overall shape, anatomy, and ultrastructure, as well as in their number (Kasumyan, 2004). There is a columnar pseudostratified epithelium that covers the olfactory lamellae (OL) and goes all the way to the ends and protrusions of the tongue. There are superficial epidermal cells in these areas. The columnar pseudostratified epithelium needs to protect its surface from water stroking and thrust, according to Kuciel et al. (2013). The sensory epithelium comprises four main types of cells: olfactory receptor neurons, supporting cells, mucous cells, and basal cells. The nonsensory epithelium includes supporting, superficial epidermal, and goblet mucous cells. Olfactory receptor neurons are spherical cells with two poles that connect to the olfactory bulb and dendrites (Kim et al., 2019). There are different types of receptor cells at the tips of dendritic spines. These include ciliated, microvillous, crypt, and rod-tipped cells (Olivares and Schmachtenberg, 2019). In 2024, Al-Zahaby et al. reported that ciliated receptor neurons have short, thick sensory cilia that do not move, whereas Samajdar and Mandal (2016) reported that microvillous receptor neurons have many tiny microvilli. According to Hansen and Zeiske (1998), crypt receptor cells have a smell knob, microvilli, and cilia that are submerged in a small crypt below their surface. There is also a receptor-like cell that sticks out in the shape of a rod and has an actin-rich projection at the top (Cheung et al., 2021). All fish species have ciliated and microvillous receptor cells in their olfactory epithelium (Hara, 1975). Some teleosts, such as Sinocyclocheilus (Waryani et al., 2013), also have rod-tipped cells, but not many other teleosts do (Ghosh, 2021). Wakisaka et al. (2017) also identified kappe and ear-shaped receptor cells in adult Danio rerio. Cylindrical and nonsensory supporting cells encircle receptor cells such as neuroglia (Forni et al., 2011). They are either ciliated or microvillar (Garcia et al., 2021), and they are mostly on the epithelium’s outer layer. However, Yamamoto and Ueda (1978) reported that they can also be found in sensory areas. If you compare the cilia of ciliated non-sensory cells to those of ciliated sensory cells, you will notice that the cilia of ciliated non-sensory cells are much longer, thinner, and able to move around. The other type of supporting cells is microvillars, which are neuroglial cells. On their mostly flat top surface, they have short, thin actin microvilli. This is different from ciliated cells, which have long and thin cilia. Menco and Morrison (2003) reported that supporting cells may also secrete mucus and may be very important for maintaining ion homeostasis in mucus, which could have a significant effect on how olfactory receptors react to smells. Goblet mucous cells are unicellular universal glands; their name is derived from their cup-shaped form (goblet). They are packed with secretory mucus granules released through their orifices on the outer surface of the olfactory epithelium (Ridley and Thornton, 2018). The secreted mucus plays a crucial role in preventing foreign debris and pathogens from adhering to the epithelium mucosal surface (Ridley and Thornton, 2018). These cells are usually round or oval and form a broken monolayer on or near the basal lamina of the olfactory epithelium. Other OCs intersperse LSs, also known as basal cells, within their bases (Datta and Bandopadhyay, 1997). After damage, the olfactory epithelium can be divided and repaired into two groups: proper and globose basal cells. The round basal cells might help repair damaged olfactory epithelial cells by acting as stem cells (Child et al., 2018). However, basal cells do not divide and do not function as neurocompetent stem cells. Instead, stem cells are stored for later use (Jang et al., 2014). The top layer of skin cells is flattened and often sheds external cuticular substances, creating stable protrusions all over the teleost skin (Hawkes, 1974). These protrusions manifest as irregular, tiny, labyrinthic, and fingerprint-like microridges (Kuciel et al., 2013). These microridges stick together and release mucus, which protects the surface of the nose from the stroking and thrusting of water (Datta and Bandopadhyay, 1997). There is not a lot of information about the smell system of European seabass (Dicentrarchus labrax) or other related Perciformes and Moroniformes species, especially when they are in different stages of development. Thus, the purpose of this study was to use a scanning electron microscope to examine how the European seabass olfactory organ changes shape and develops, from the start of the olfactory placode to the growth of adult olfactory rosettes. The study also focused on the development of various olfactory epithelial cells. Material and MethodsThe embryos and adults of Dicentrarchus labrax L used in this studyTen samples were collected at each developmental stage. The samples came from fish naturally spawning in captivity in a broodstock of Bardawil Lake at the Egyptian Authority of Fish Production’s hatchery. Fertile eggs were obtained naturally or with hormonal injections under artificial thermal and photoperiodic cycles (Zanuy et al., 1986). The successfully produced natural eggs and yolk sac larvae were incubated at 15°C in the dark. Egg samples were then captured at different periods after fertilization to track ontogeny. The formation of the olfactory placode begins approximately 24 to 30 hours postfertilization (Diaz et al., 2002). Samples were collected and dissected every 6 hours starting 30 hours after fertilization. The olfactory placode appeared fully differentiated at the time of hatching, as observed by scanning electron microscopy. The specimens came from different stages of European seabass development, such as larvae (Fries-I at 35 days, Fries-II at 45 days), fingerlings (90 days after hatching), juveniles (9–12 months), and adults (3 years old). The samples were transported to the Laboratory of Experimental Zoology (Faculty of Science, Zagazig University, Egypt) in bags with sufficient airflow. We selected 10 specimens of each stage from different stages, such as larvae, Fries-I and Fries-II, fingerlings, juveniles, and adults, and prepared them for scanning electron microscopy (SEM). A buffer solution was used to clean the larvae. For the fingerlings, juveniles, and adults, fixative-soaked cotton was used to clean their sensory chambers. The rosettes were carefully excised and perfused with 0.1 M phosphate buffer. The specimens were fixed in 2.5% glutaraldehyde in phosphate buffer (pH 7.4) for 24 hours at 4°C and then rinsed in the same buffer for 10 min. The samples were then fixed in 1% osmium tetroxide in 0.1 M phosphate buffer (pH 7.4) for 2 hours at room temperature. They were then dried using a series of increasing amounts of acetone and isoamyl acetate (Newbury and Yakowitz, 1975). The specimens were dehydrated and then dried in a critical-point dryer (Hitachi 8CP2). The specimens were subsequently mounted on metal stubs and gold-coated (Zhou et al., 2006). Finally, the specimens were examined, scanned, and photographed using a Joel IT200 SEM, located at the Faculty of Science, Alexandria University, Egypt. Ethical approvalThis study was conducted according to the ethical guidelines of Zagazig University, Egypt. The study received ethical approval number: ZU-IACUC/1/F/76/2024; Institution: ZU-IACUC from Zagazig University. ResultsIn this study, 10 specimens were examined for each developmental stage. Observations were recorded in 6 specimens (0- and 6-hours post-hatching), 5 specimens (12 hours post-hatching), 7 specimens (18 hours post-hatching), and 9 specimens (24 hours post-hatching). Additionally, 7 specimens (Fry 1), 9 specimens (Fry 2), and 5 specimens (Fingerlings) were examined, with 8 specimens for both juvenile and adult stages. These findings reflect the developmental progression observed across all studied stages. European seabass larvaeAfter hatching, the larvae had thickened the olfactory placode (OPL) just in front of the eye placode (E) on each side. The skin cells that cover the whole body are flattened, as shown in Figure 1a. The olfactory pit (OP, Fig. 1b) is the structure formed when epidermal cells peel off over the OPL 6 hours after egg hatching. The OP, as shown in Figure 1b, had a rounded circumference owing to the complete sloughing of epidermal cells just anterior to the developing eye. By 12 hours post-hatching, the rounded OP was bounded by SECs with labyrinthic fingerprint microridges (LFMs, Fig. 1c). When larvae were 18 hours old, the OP got longer and showed new, undifferentiated (OCs, Fig. 1d). At 24 hours post-hatching, mucous pores (MPs) of goblet mucous cells (GMCs) and ciliated receptor cells (CRCs) with olfactory knobs (OKs) were observed, distributed between ciliated supporting cells (CSCs) and microvillar supporting cells (MSCs), with SECs also forming the boundary (Fig. 1e). European seabass friesAt 35 days post-hatching, after yolk sac absorption (YS), the European seabass fries were dependent on exogenous feeding. A SEM picture showed an OP that was getting deeper and more teardrop-shaped above the mouth opening (MO). The OP was surrounded by SECs with LFM (Fig. 2a, 2b). CRCs, rare microvillar receptor cells (MRCs) with minute microvilli, and MP of hidden GMCs were present in the OP (Fig. 2c, 2d). The nonsensory epithelial area (NSA) around the OP contained numerous MPs interspersed between SECs (Fig. 2e). At 45 days post-hatching, the OP elongated between the MO and E, and the medial rim of the epidermal layer protruded to form the future nasal bridge (NB, Fig. 3a, 3b). Higher magnification images show kinocilium (KC) from CSCs, MSCs with microvilli, and rare CRCs and MRCs (Fig. 3c, 3d). The bottom periphery of the OP was covered with SECs and LFM, with dispersed islets of CSCs, KC, and MP (Fig. 3e, 3f). The medial upper and lower rims eventually joined to form the NB, thereby completing the separation of the anterior incurrent nostril (InN) and posterior excurrent nostril (ExN, Fig. 3g, 4a).
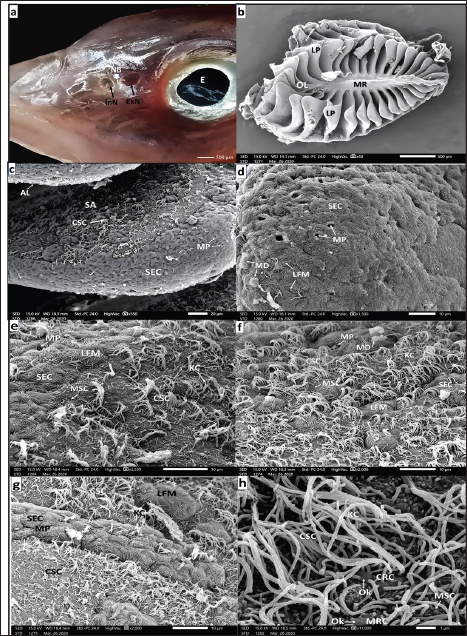
Fig. 1. a. SEM micrograph of a seabass just hatched larva showing: fairly peeling of superficial epidermal cells (SECs) covering an olfactory placode (OPL) just anterior to the eye (E) placode (Scale bar 50 µm). b. SEM micrograph of seabass larva (6-hours post-hatching) showing: Rounded olfactory pit (OP) after sloughing of SECs with labyrinthic microridges (LFMs) over the OPL just anterior eye (E) and the placoid of exfoliated SECs too (Scale bar 50 µm). c. SEM micrograph of seabass larva (12-hours. post- hatching) showing: Evident rounded olfactory pits (OP) bounded by flattened (SECs) of labyrinthic microridges (LFMs) (Scale bar 50 µm). d. SEM micrograph of a seabass larva (18-hours. post- hatching) showing: Elongated olfactory pit (OP) bounded by flattened (SECs) with labyrinthic microridges (LFM) and comprising newly formed undifferentiated OCs (Scale bar 10 µm). e. Magnified SEM micrograph of the olfactory pit (OP) of a seabass larva (24-hours post-hatching) showing: CRC with olfactory knob (OK), CSC with thin long kinocilium (KC), mucous cell pores (MP) of goblet mucous cell (GMSc), and microvillar supporting cell (MSC) spreading on the flat floor of the (OP) bounded by flattened (SECs) (Scale bar 2 µm).
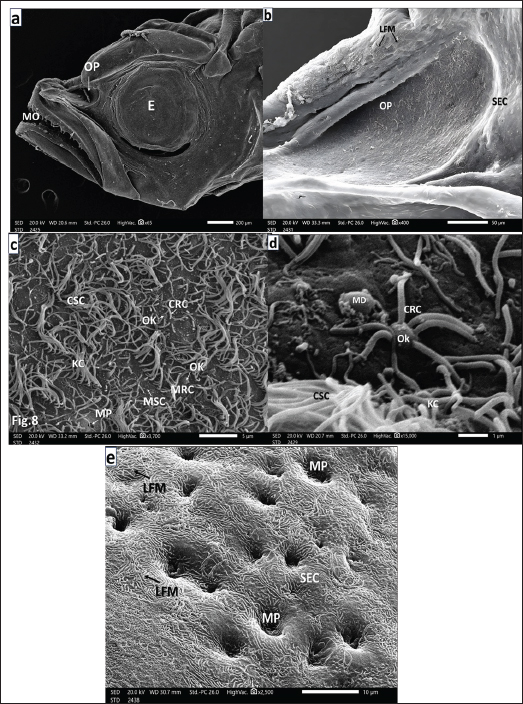
Fig. 2. a. SEM micrograph of the head of a seabass fry (35 days post-hatching) showing: Deepening olfactory pit (OP) above the mouth orifice (MO) and just anterior to the eyes (E) (Scale bar 200 µm). b. SEM micrograph of the deepening olfactory pit (OP) of a seabass fry (35 days post-hatching) Showing that it is bounded by SEC with labyrinthic microridges (LFMs) (Scale bar 50 µm).c. SEM micrograph of the olfactory pit (OP) bottom of seabass fry (35 days post-hatching) showing: Ciliated CRC, microvillous MRC both with their olfactory knob (OK) among ciliated CSCs with disarrayed long KC, mcrovillar MSC, and mucous cell pores (MP) (Scale bar 5 µm). d. Higher magnification SEM micrograph of the olfactory pit (OP) of seabass fry (35 days post-hatching) showing: CRC with short thick cilia emitted from the olfactory knob (OK), ciliated CSCs of long thin KC and mucus droplet (MD) (Scale bar 1 µm). e. Higher SEM micrograph at the periphery and around the olfactory pit (OP) of a sea bass fry (35 days post- hatching) showing: that this non-sensory area is packed with mucous cell pores (MP) among flattened SECs with their labyrinthic microridges (LFMs) (scale bar 10 µm). European seabass fingerlingsAt 90 days post-hatching, the European seabass fingerlings (3.5 cm) exhibited major developmental changes, with the formation of olfactory cavities (OCHs) and two nostrils. Seven OLs began to form olfactory rosettes (ORs). The left rosette was more developed and had a prominent lingual protrusion (LP; Fig. 4b), whereas the right rosette was less developed (Fig. 4c). The surrounding NSA was made of SECs with LFM and contained MP (Fig. 4d). The sensory area (SA) is populated by CSCs with KC and rare CRCs and MRCs (Fig. 4e, 4f). The NSA was primarily composed of GMCs and MPs interspersed between flattened SECs (Fig. 4g).
Fig. 3. a. SEM micrograph of a seabass head fry (45 days post-hatching) showing: More deepened olfactory pit (OP) in-between mouth opening (MO) and eye (E), with its medial upper and lower epidermal rim (ER) protruding inward, hanging over the olfactory pit to form the inter-nasal bridge (NB) (Scale bar 200 µm). b. SEM micrograph of the previous figure of seabass fry head (45 days post-hatching) showing: Elliptical deepening olfactory pit (OP) with medial upper and lower epidermal rims (ER) protruding inward hanging over the pit to form an inter-nasal bridge (NB) and then the roof of the olfactory chamber (OCH) (Scale bar 50 µm). c. SEM micrograph of a seabass fry olfactory pit bottom of 45 days post-hatching showing: Further deepening of the olfactory pit (OP) bottom and more elevation of its peripheries. The marginal nonsensory area (NSA) is partially enveloped within the disarrayed long thin KC of CSCs and the microvilli of MSCs. The olfactory pit is bounded by SECs of LFM embracing some MP of GMCs. But inward seldom detected CRCs with OK (Scale bar 10 µm). d. Magnified SEM micrograph of the previous peripheral area of the olfactory pit bottom 45 days post-hatching seabass fry showing: MSCs followed inward by CSCs with disarrayed long thin KC and seldom detected CRCs. The outer edge of the OP was covered with SECs and their LFM (Scale bar 2 µm). e. Magnified SEM micrograph of a seabass fry olfactory pit (45 days post-hatching) showing: Pit’s bottom periphery covered with SEC and LFM, separate islets of CSC with disarrayed long thin KC. Infrequently detected CRCs with their OK, some pores of GMCs and mucus droplets (MD) (Scale bar 5 µm). f. Higher magnification SEM micrograph of a seabass fry olfactory pit (45 days post-hatching) showing: Seldom noticed CRC and MRC were rarely observed with short thicker cilia and/or very short microvilli, respectively, radially emitted from OK dispersed among copious disarrayed long kinocilium of CSCs (Scale bar 1 µm). g. SEM micrograph of seabass fry head (75 days post-hatching) showing: Medially joining of the two medial upper and lower epidermal rims (ER) forming the inter nasal bridge (NB) separating the two nostrils, small anterior incurrent (InN), and considerably wider posterior excurrent (ExN) just anterior to the eye (E) (Scale bar 100 µm).
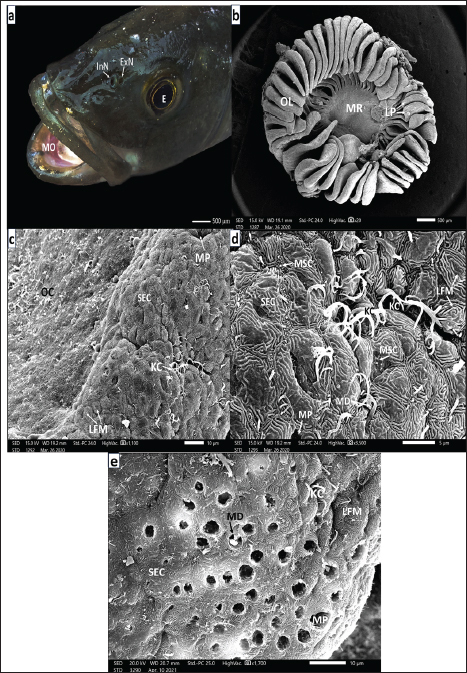
Fig. 4. a. SEM micrograph of a seabass fingerling head (90 days post-hatching) showing: Complete separation of the two nostrils, protruding rounded anterior InN and wider elongated posterior ExN above the MO and in front of E (Scale bar 500 µm). b. SEM micrograph of the right olfactory rosette of the same seabass fingerling (90 days post-hatching) setting on the bottom of OCH showing: seven less-developed (OL) arranged around less developed MR, but LP are not yet formed but appeared as cell thickenings (Scale bar 100 μm). c. SEM micrograph of the left olfactory rosette of seabass fingerling (90 days post-hatching) setting on the bottom of OCH: showing: seven more developed OL lined up around the MR and with obvious LP (Scale bar 100 μm).d. SEM micrograph of an OL periphery of seabass fingerling (90 days post-hatching) showing: non-sensory area (NSA) with SECs embracing many mucous cell pores (MP) followed inward by the sensory area packed with undifferentiated olfactory receptors and non-receptor cells (Scale bar 20 µm.) e. Magnified SEM micrograph of olfactory epithelium covering the sensory area of an OL of seabass fingerling (90 days post-hatching) embracing seldom observed CRCs with short thicker cilia emitted from OK concealed among supporting cells either ciliated (CRC) with disarrayed long thin KC or with very short microvilli of MRC (Scale bar 5 µm).f. More magnified SEM micrograph of olfactory epithelium covering the sensory area of an OL of seabass fingerling (90 days post- hatching) showing: CRCs short thicker cilia and MRCs cells of plentiful very short microvilli both emitted from olfactory knob (OK). These receptors are dispersed among disarrayed long KCs of CSCs and/or short microvilli of MSCs. Notice the spreading mucus droplets (MD) (Scale bar 2 µm). g. Magnified SEM micrograph of the olfactory epithelium of seabass fingerlings (90 days post-hatching) but at the border line between the nonsensory (NSA) and sensory (SA) areas the first one mainly comprises; GMCs denoted by their MP dispersed in-between mass of SECs with their LFM. Notice that a slight appearance appeared in the sensory sector, which embraces little supporting cells either MRCs or CSCs with very little disarrayed thin kinocilium (Scale bar 5 µm). European seabass juvenilesThe European seabass juveniles (10 cm, 9–12 months old) had nostrils that were completely separate from each other. The front InN was small, round, and raised, while the back ExN was wider and flatter (Fig. 5a). As the fish grew, the OR increased in size and its OLs showed prominent LP, characteristic of adults, as demonstrated later (Fig. 5b). The NSA comprised SECs and GMCs with MP, a transitional zone with CSCs and KC, and an SA comprised CRCs and MRCs (Fig. 5c–5f). The SA had CRCs with thick sensory cilia, MRCs, and very few rod-tipped cells (RCs), which were all OK (Fig. 5g, h). Fully grown adult European seabassThe adult Dicentrarchus labrax, like most teleost fish, has two paired peripheral olfactory organs that sit in the nasal chambers. The front part of the nose, called InN, was stock out, and the back part, called ExN, was flat. They are connected to the outside world through a nasal bridge (NB) (Fig. 6a). The position and shape of the nostrils ensured efficient in-and-out flow of water in the olfactory chamber. The olfactory rosettes (ORs) were composed of OLs with strong LP and an NSA composed of SECs and MP (Fig. 6b–d). The transitional zone between the NSA and SA included MRCs, CRCs, and rod-tipped cells (RCs) with single compound cilia (R), all of which were OK (Fig. 7a, b). The SA was mainly composed of CRCs with thick cilia, MRCs, and few RCs dispersed between MSCs and CSCs (Fig. 7c, d). DiscussionDuring development, the olfactory organs were the first chemosensory organs to form (Hansen and Zielinski, 2005), as they began to separate early in the fish life (Pashchenko and Kasumyan, 2015, 2016; Tytiuk et al., 2020). Olfaction is a crucial sensory faculties for the regulation of animal behavioral patterns (Yamamoto and Ueda, 1979). In addition, olfactory traces last longer because chemical molecules move more slowly in water than in air. This allows scents to travel long distances with the help of water currents. It is considered the major mediator of chemical signals with gustation in different fish (Hara and Zielinski, 1989). In this study on European seabass, we found that the olfactory organs begin as olfactory placode, with undifferentiated cell thickenings beneath the skin. The placodes are on both sides of the back of the head, in front of the optic cups. They are covered by a thin layer of epidermal cells. They are not the same as skin cells or brain cells, and they are thought to come from the side parts of the anterior neural plate in the back and side of the head (Whitlock and Westerfield, 2000).The first appearance of olfactory placode varied among different fish species according to the type of fish egg (Pashchenko and Kasumyan, 2016). The present study focused on the European seabass, which lay small, buoyant eggs in the ocean. The olfactory placode was activated on the first day that the larvae hatched. The same was true for the Perciformes, Amphiprion melanopus (Arvedlund et al., 2000). The European bitterling fish hide their eggs in invertebrate gills, and olfactory placodes form three days post-hatching (Pashchenko and Kasumyan, 2016). In most actinopterygians, olfactory placode arises from subepidermal cells that give rise to all OCs (Whitlock and Westerfield, 2000). However, Acipenser sturgeons come from two different types of cells: epidermal cells, which are nonsensory supporting cells, and subepidermal cells, which are sensory receptors (Zeiske et al., 2003). After six hours, the olfactory placode becomes thicker and sinks deeper into the mesenchymal tissue. They also get more wavy and look like simple olfactory pits with OCs that have not changed yet. At the same time, as in many other fish species (Pashchenko and Kasumyan, 2016; Tytiuk et al., 2020), the olfactory pit forms when the fish is still an embryo. However, in amphip, by the end of the first day after hatching, the OCs have changed into CRCs, MRCs, and sporadic GMCs, which can be identified by their pores. Some animals, such as the Ctenopharyngodon idella (Pashchenko and Kasumyan, 2015) and Ancistrus dolichopterus (Tytiuk et al., 2020), have pits surrounded by skin cells (SECs) that are shaped such as labyrinths and have microridges on them. In the case of Ctenopharyngodon idella (Pashchenko and Kasumyan, 2015) and Ancistrus dolichopterus (Tytiuk et al., 2020), epidermal cells (SECs) with labyrinthic microridges surround the pits. CRCs first appear in European seabass larvae 24 h post-hatching with 2-4 short, thick cilia. By 35 days post-hatching, and even in adults, they have 4-6 cilia. The sensor not only does the SRC differentiate by the end of the first day after hatching in Ancistrus dolichopterus (Tytiuk et al., 2020) and Oncorhynchus nerka (Rheinsmith et al., 2022), but in barfin flounder, it does not occur until day 14 after hatching (Yamamoto et al., 2004). Barfin flounder do not do this until day 14 after hatching, but Ancistrus dolichopterus (Tytiuk et al., 2020) and Oncorhynchus nerka (Rheinsmith et al., 2022) do it all the time. Around 35 days post-hatching, European seabass larvae lose their yolk sac and depend on exogenous feeding during the fry stage (Miller and Kendall, 2009). The olfactory pits become longer in European seabass fries. They are surrounded by SECs and have CRCs and MRCs at the bottom, along with thin kinocilium from CSCs, short microvilli from MSCs, and MP from GMCs. The bottom of the olfactory pit becomes deeper 45 days after hatching, and by day 75, lateral epidermal protrusions help form the nasal bridge. This creates two nostrils, one narrow anterior inflowing nostril and one much wider posterior exflowing nostril (Tytiuk et al., 2020). When the olfactory chambers are fully developed, the olfactory bottom epithelium starts to rise into a complex series of folds or lamellae. This behavior is more noticeable in European seabass fingerlings 90 days after hatch. As described in other teleosts by Diaz et al. (2002) and Hara (2011), they appear to be spread out from a central MR that goes from the back to the front on the bottom of the OCH. Generally, in Acanthopterygii, OL are symmetrically attached to a MR, forming a rosette shape (Hansen and Zielinski, 2005). As these lamellae grow in the middle, new ones appear at the base and move up the back, placing the oldest and biggest lamellae at the back (Hara, 2011). In European seabass, the number of lamellae increases with body size, from seven in fries to 31 in juveniles and 38–40 in adults. . In a similar manner, Rheinsmith et al. (2022) noted that olfactory rosette size and lamellar numbers steadily increase with increasing body size across life stages. Similarly, Rheinsmith et al. (2022). However, there is pronounced variability in the number of OL across fish species. Some species lack an OL, such as Oryzias latipes and Gambusia affinis (Kasumyan, 2004), whereas others have only one OL, such as Neogobius melanostomus (Belanger et al., 2003). Atherion elymus and Hemiramphus sajori each have three lamellae in each rosette (Kasumyan, 2004), but Anguilla vulgaris can have up to 100 OL (Al-Zahaby et al., 2024), for example. Thus, the number of OL is species-specific (Kasumyan, 2004). The adult European seabass have two layers of pseudostratified columnar epithelium that are split into sensory and nonsensory areas on the OL lamella (Hara, 2011). Islets of sensory areas, such as the ones in Cyprinus carpio (Kasumyan, 2004) and Sinocyclocheilus furcodorsalis (Waryani et al., 2013), are found in areas that are not sensory. Each area contains sensory receptors and supporting cells. There are goblet mucus cells, basal cells, and supporting, flattened, superficial epidermal cells in areas that are not sensed (Pashchenko and Kasumyan, 2015). Different species’ olfactory epithelia are built and made up in very different ways. This is because they eat and live in the environment (Hansen and Zeiske, 1993; Bhute and Baile, 2007; Samajdar and Mandal, 2016). CRCs are distinguished by their cilia lacking dynein arms and protruding from the apical olfactory knob (Hansen and Zeiske, 1998). These cilia exhibit the 9+2 microtubule arrangement (Zeiske et al., 2010). At 24 h after hatching, the CSCs, MSCs, and MP on the bottom of the olfactory pit in European seabass larvae display this arrangement. When European seabass fries are 35 to 45 days old (Hara and Zielinski, 1989), they lose most of their mucous receptor cells (MRCs) by 11 days old. This is also the case for Oncorhynchus mykiss fries. Both types of olfactory sensory neurons grow separately from basal stem cells, ensuring that they work independently (Datta and Bandopadhyay, 1997). The existing olfactory sensory neurons are specialized in the perception of different odorants (Hamdani et al., 2000). Amino acids are important smells for finding food. They are picked up by ciliated and microvillous OSNs, which are common in teleosts (Hansen and Zielinski, 2005). European seabass exhibit greater sensitivity to amino acid odorants in seawater than freshwater (Velez et al., 2024). Supporting cells in actinopterygians are classified as ciliated (CSCs) or microvillous (MSCs) based on the protrusions on their flat top surface. CSCs resemble respiratory epithelial cells in tetrapods (Muller and Marc, 1984). They bear long, thin, motile cilia with dynein arms, indicating their motility, and are thus termed kinocilium (Zeiske et al., 1992). In the European seabass (Dicentrarchus labrax), CSCs remain until OL form on the floor of the olfactory chamber 90 days after hatching. During the fingerling stage, they help to move the water. Subsequently, CSCs are replaced by MSCs, which remain throughout adulthood and are characteristic of cyclamate fish species. Supporting cells are either grouped or individual ciliated or microvillar cells (Muller and Marc, 1984). Their role in providing mechanical support to surrounding receptors is a recognized phenomenon (Datta and Bandopadhyay, 1997). Then, microvillar supporting cells (MSCs) perform glial functions (Hansen and Zeiske, 1998). Perhaps they also aid in the mechanical separation of hazardous materials entangled in the secreted mucus (Ghosh, 2021). In addition, we should look at the large, flattened superficial epidermal cells (SECs), which can be recognized by their unique labyrinthic microridges (LFMs). In all developmental stages of European seabass, actin-rich keratinocytes with highly distinctive labyrinthic microridges are observed. These tiny bumps, which cover the olfactory and eye placode, surround the olfactory pit and make up the lamellae’s edges or islets between sensory areas, preventing the epithelium from getting hurt or worn down (Depasquale, 2018). The microridges are organized in a pattern that is like a fingerprint, and it is unique to each cell. They can only form when cells touch each other, as observed in teleost epithelia (Hawkes, 1974; Doyle and Lee, 2005; Lam et al., 2015). Goblet mucus-secreting cells are another type of cell that can be found in the olfactory epithelia of European seabass at all stages of development. All vertebrates, including teleosts, have one-celled glands in their olfactory mucosa. They constantly grow new cells from pluripotent basal stem cells. They release glycoprotein mucus onto the epidermal surface and reach the olfactory epithelia (Dao and Le, 2023). This process provides protection to the olfactory cilia from mechanical stress and ensures optimal water flow across the OL. Thus, the even distribution of the mucus layer over the olfactory neurons makes it easier for the fish to smell things (Rygg et al., 2013). However, MP are absent in newly hatched European seabass larvae, with their first appearance observed 24 h after hatching at the bottom of the olfactory pit. By the end of its life, a European seabass contains clear MPs from goblet mucus-secreting cells, especially among SECs of labyrinthine microridges. These MPs are abundant in fries at 35 days post-hatching, as well as in fingerlings, juveniles, and fully grown individuals. This was also observed in Labeo rohita, where goblet mucous cells were not common in the larvae’s olfactory pit but were common in juveniles and adults (Bhute and Baile, 2007). For example, Hansen and Zeiske (1998) stated that the lack of goblet cells in carp larvae might be made up for by the presence of nonsensory supporting cells that might secrete substances and take them in. In their smelly skin, European seabass (Hara, 1975) and all other teleosts have ciliated and microvillous receptor cells. However, rod-tipped cells are only found in adult European seabass and other teleost species (Ghosh, 2021; Al-Zahaby et al., 2023, 2024; Elsheikh et al., 2024). Through retrograde degeneration, these cells were found to be different types of receptors, separating them from other types of receptor cells (Ichikawa and Ueda, 1977). In addition, Muller and Marc (1984) suggested that the adhesion of dendritic cilia could be a sign of senescence. They stated that the compound cilium of the rod cells was just a fixation error. Datta and Bandopadhyay (1997) refuted this, proposing that rod-tipped cells are not a distinct olfactory neuron subtype. Instead, their rods could result from CRC cilia fusion under environmental influence. Interestingly, rod-tipped cells are observed in zebrafish olfactory pits shortly after hatching, even in the absence of ciliated neurons (Cheung et al., 2021). The findings of this study, alongside recent research by Al-Zahaby et al. (2023, 2024) and Elsheikh et al. (2024), further corroborate Datta and Bandopadhyay’s hypothesis.
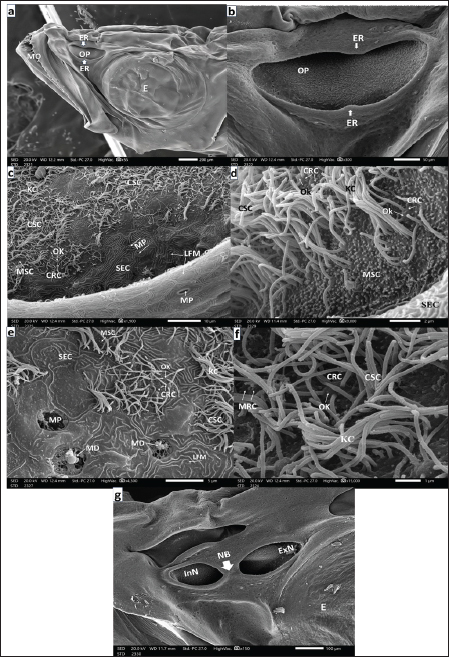
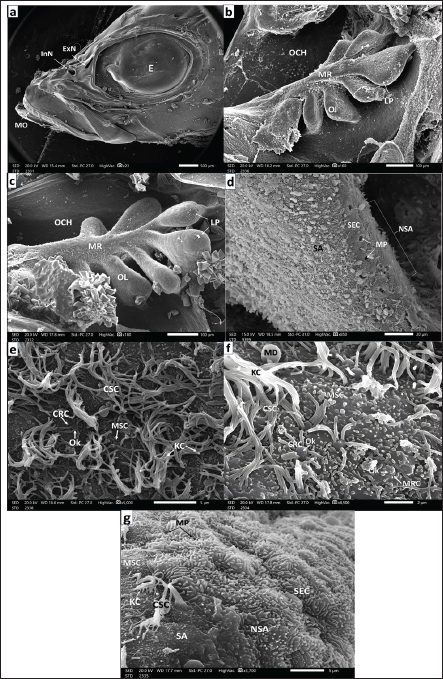
Fig. 5. a. Photograph of a seabass juvenile’s head (9–12-month post-hatching) showing: Complete separation of the two nostrils by the broad nasal bridge (NB) into a small rounded elevated anterior InN and a wider rounded flattened posterior ExN in front of the eye (E) (Scale bar 500 µm). b. SEM micrograph of an olfactory rosette of seabass juveniles (9-12-month post-hatching) showing: Well-developed 31 OL with prominent LP. Arranged on both sides of an elongated well-developed MR (Scale bar 500 µm). c. SEM micrograph of the LP of an OL and adjacent another one of seabass juveniles (9–12-month post-hatching) showing: The most marginal area with SECs and MPs in-between, followed inward with a transitional area of CSCs and thence sensory area SA in addition to the LP of adjacent lamella (AL) (Scale bar 20 µm). d. SEM micrograph of the LP peripheral edge with almost nonsensory epithelium of seabass juveniles (9–12-month post-hatching) showing: many mucous cell pores (MP) dispersed among the main constituents of SEC masses characterized by labyrinthic LFM. Notice the extruded mucus droplets (MD) (Scale bar 10 µm). e. SEM micrograph of the OL of seabass juveniles (9–12-month post-hatching) in the transitional zone between sensory and nonsensory areas but little to the latter showing: Aggregation of SECs penetrated with MP of GMCs as well dispersed MSCs and CSCs, but sensory receptor cells (SRCs) are hidden among disarrayed KC of CSCs (Scale bar 10 µm). f. SEM micrograph of the OL of seabass juvenile (9–12-month post-hatching) in the transitional zone but more in the sensory area showing: In addition, aggregation SECs dispersed in isolated islets penetrated with MP of GMCs. but the SRCs are hidden also among disarrayed thin long KC of CSCs (Scale bar 10 µm). g. SEM micrograph of the OL of seabass juvenile (9–12-month post-hatching) also in a transitional zone showed: CSCs with disarrayed KCs appeared in islets in between extending strands of flattened SEC aggregations penetrated with MP, but the seldom detected SRCs may be hidden too among copious of thin long KCs of CSCs (Scale bar 10 µm). h. Magnified SEM micrograph of lamella’s sensory area of a seabass juvenile (9–12-month post-hatching) showing: CRCs with short sensory cilia, MRCs with minute microvilli, both the two cells are with OK and hidden among copious KC of CSC (Scale bar 1 µm).
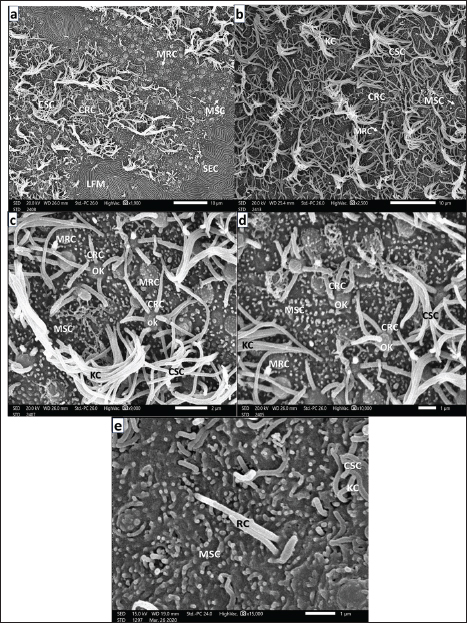
Fig. 6. a. Photograph of a full-grown seabass’s head (3 years old) showing: The left pair of nostrils, anterior outstanding of the elevated back margin InN, and posterior almost wider flat ExN above and in front of the eye (E) and above the posterior corner the mouth (MO) (Scale bar 500 µm). b. SEM micrograph of an olfactory rosette of full-grown seabass (3 years old) showing: Olfactory rosette of 38–40 lamellae (OL) with prominent LP. The lamellae are arranged around the oval MR (Scale bar 500 µm). c. Magnified SEM micrograph of the nonsensory olfactory epithelium covering the periphery of the OL of adult seabass (3 years old) showing; Mucous cell pores (MP) and very little disarrayed thin KC dispersed in-between masses of SECs with their prominent LFM. Inwardly, the transitional zone of undifferentiated OCs (Scale bar 10 µm). d. More magnified SEM micrograph of the previous nonsensory epithelium covering the olfactory lamella’s periphery of the seabass showing; MPs, MSCs, and very little long KC of CSCs dispersed in-between masses of SECs with their labyrinthic LFM (Scale bar 5 µm). e. Magnified SEM micrograph of the most periphery of the OL of adult seabass (3 years old) showing: Abundant MP, some with MD dispersed among masses of SECs with their prominent LFM, and very little KC of hidden CSCs (Scale bar 10 µm).
Fig. 7. a. SEM micrograph of the transitional epithelium between sensory (SA) and nonsensory (NSA) areas of adult seabass (3 years old) showing: Islets of SECs with their LFM, abundant MRCs but little CRCs both with OK dispersed among MSCs and CSCs of disarrayed KC (Scale bar 10 µm). b. SEM micrograph of the ciliary zone of the adult seabass’s OL (3-years old) showing: seldom CRCs, MRCs both with prominent OK dispersed in-between overall CSCs of disarrayed long thin KC and very little MSCs (Scale bar 10 µm). c. Magnified SEM micrograph of olfactory sensory areas of adult seabass (3 years old) showing: CRCs of thick sensory cilia, many MRCs both are with prominent OK and dispersed among overall MSCs and little CSCs of disarrayed long thin KC (Scale bar 2 µm). d. Another magnified SEM micrograph of the sensory areas of adult seabass (3 years old) showing: CRCs of short thick sensory cilia, many MRCs, both neurons with prominent OK and dispersed among the overall MSC and little CSCs of long disarrayed KC (Scale bar 1µm). e. Magnified SEM micrograph of the same adult seabass (3 years old) showing: Two adjacent rod-tipped cells (RC), each with a single compound cilium (R) emitted from prominent OK observed in-between overall MSCs and some CSCs of long thin KC (Scale bar 1µm). ConclusionThis study showed how the ultrastructure of the olfactory organ of Dicentrarchus labrax changes from hatching to adulthood. By 90 days after hatching, multilamellar rosettes have formed, indicating that the fish’s olfactory structure is fully developed. The number of lamellae increases with fish size, reaching 38–40 in adults. Goblet mucous cells, which are predominantly found in nonsensory areas, play a lesser role in sensory regions. These results indicate that OCs are functionally specialized. This shows that the organ has played a key role in chemical communication and adaptation to its environment throughout the development of the European seabass. Future research could look into how advanced methods such as nanotechnology can be used to create targeted treatments for fish diseases and how genetic engineering can be used to make aquaculture species more resistant to disease. It would be helpful to conduct experiments to examine the effects of pathogen exposure over time and to try out new treatments in a controlled setting. These results could also be used to develop practical plans, such as nanotechnology-based therapies, that would greatly improve the health and longevity of fish in aquaculture systems. AcknowledgmentsWe would like to thank the Zoology Department, Faculty of Science, Zagazig University, Zagazig, Egypt, for technical support and helpful assistance. Conflict of interestThe authors have not declared any conflict of interest. FundingThis study received no funding. Authors’ contributionThe practical part and its completion were conducted by both Sheren A. Al-Zahaby and Radwa A. Mohamed. The research writing was performed by Sheren A. Al-Zahaby, Radwa A. Mohamed, and E. H. Elsheikh. All the work was under the supervision, guidance, and review of Al-Ahmady S. Al-Zahaby. Data availabilityThe original data presented in this study are included in the manuscript. For inquiries, please contact the corresponding author. ReferencesAl-Zahaby, S.A., Farag, M.R., Alagawany, M., Taha, H.S.A., Varoni, M.V., Crescenzo, G. and Mawed, S.A. 2023. Zinc oxide nanoparticles (ZnO-NPs) induce cytotoxicity in the zebrafish olfactory organs via activating oxidative stress and apoptosis at the ultrastructure and genetic levels. Animals 13(18), 2867. Al-Zahaby, S.A., Hassan, S.S. and Elsheikh, E.H. 2024. Ultramicroscopic organization of the exterior olfactory organ in Anguilla vulgaris in relation to its spawning migration. Open Vet. J. 14(1), 512–524. Arvedlund, M., Larsen, K. and Winsor, H. 2000. The embryonic development of the olfactory system in Amphiprion melanopus (Perciformes: Pomacentridae) related to the host imprinting hypothesis. J. Mar. Biol. Assoc. 80(6), 1103–1109. Belanger, R.M., Smith, C.M., Corkum, L.D. and Zielinski, B.S. 2003. Morphology and histochemistry of the peripheral olfactory organ in the round goby, Neogobius melanostomus (Teleostei; Gobiidae). J. Morphol. 257(1), 62–71. Beraud, C., van der Molen, J., Armstrong, M., Hunter, E., Fonseca, L., and Hyder, K. 2018. The influence of oceanographic conditions and larval behaviour on settlement successThe European sea bass, Dicentrarchus labrax (L.). ICES J. Mar. Sci. 75(2), 455–470. Bhute, Y.V. and Baile, V.V. 2007. Organization of the olfactory system of the Indian Major Carp, Labeo rohita (Hamilton): a scanning and transmission electron microscopic study. J. Evol. Biochem. Physiol. 43(3), 342–349. Cheung, K.Y., Jesuthasan, S.J., Baxendale, S., van Hateren, N.J., Marzo, M., Hill, C.J. and Whitfield. T.T. 2021. Olfactory rod cells: a rare cell type in the larval zebrafish olfactory epithelium with a large actin-rich apical projection. Front. Physiol. 12, 626080. Child, K.M., Herrick, D.B., Schwob, J.E., Holbrook, E.H. and Jang, W. 2018. The neuroregenerative capacity of olfactory stem cells is not limitless: implications for aging. J. Neurosci. 38(31), 6806–6824. Dao, D.P.D. and Le, P.H. 2023. Histology, goblet cells. In: StatPearls [Internet]. Treasure Island, FL: Stat Pearls Publishing, 2024, DOI: https://www.ncbi.nlm.nih.gov/sites/books/NBK553208/ Datta, N.C. and Bandopadhyay, S. 1997. Ultrastructure of cell types of the olfactory epithelium in a catfish, Heteropneustes fossilis (Bloch). J. Biosci. 22(2), 233–245. Deng, G.T. and Demisse, B.T. 2023. Freshwater fish migration: fisheries management strategy insight. In Sustainable wildlife management. Eds., Khan Perveen, F. London, UK: Intechopen; doi: 10.5772/intechopen.110027 Depasquale, J.A. 2018. Actin microridges, actin and actin-rich structures. Anatom. Record. 301(12), 2037–2050. Diaz, J.P., Prie-Granie, M., Blasco, C., Noell, T. and Connes, R. 2002. Ultrastructural study of te olfactory organ in adult and developing European seabass, Dicentrarchus labrax. Can. J. Zool. 80(9), 1610–1622. Doyle, A.D. and Lee, J. 2005. Cyclic changes in keratocyte speed and traction stress arise from Ca2+- dependent regulation of cell adhesiveness. J. Cell Sci. 118(2), 369–379. El-Shebly, A.A. 2009. Aquaculture potential of sea bass (Dicentrarchus Labrax) in brackish water fish farms in Egypt. J. Appl. Sci. Res. 5(7), 765–769. Elsheikh, E.H., Hassan, S.S. and Al-Zahaby, S.A. 2024. Olfactory epithelium organization of the grass carp (Ctenopharyngodon idella) at the ultrastructural level: SEM and TEM observations. J. Adv. Vet. Res. 14(5), 793–798. FAO. 2018. Fisheries statistics and information. In: FAO Fisheries and Aquaculture Department. FAO, Rome. Available via http://www.fao.org/fishery/statistics/en. Forni, P.E., Taylor-Burds, C., Melvin, V.S., Williams, T. and Wray, S. 2011. Neural crest and ectodermal cells intermix in the nasal placode to give rise to GnRH-1 neurons, sensory neurons, and olfactory ensheathing cells. J. Neurosci. 31(18), 6915–6127. Garcia, P.A.T., Nevitt, G.A., Pesavento, J.B. and The, S.J. 2021. Gross morphology, histology, and ultrastructure of the olfactory rosette of a critically endangered indicator species, the Delta Smelt, Hypomesus transpacificus. J. Comp. Physiol. A 207, 597–616. Ghosh, S.K. 2021. The olfactory organ of schilbid catfish Eutropiichthys vacha (Hamilton, 1822): morphological and ultrastructural studies. J. Basic Appl. Zool. 82(4), 1–11. Hamdani, E. and Døving, K. 2007. The functional organization of the fish olfactory system. Progress Neurobiol. 82, 80–86. Hamdani, E.H., Stabell, O.B., Alexander, G. and Døving, K.B. 2000. Alarm reaction in the crucian carp is mediated by the medial bundle of the medial olfactory tract. Chem. Senses, 25, 103–109. Hansen, A. and Zeiske, E. 1993. Development of the olfactory organ in the zebrafish, Brachydanio rerio. J. Comp. Neurol. 333(2), 289–300. Hansen, A. and Zeiske, E. 1998. The peripheral olfactory organ of the zebrafish, Danio rerio: an ultrastructural study. Chem. Senses. 23(1), 39–48. Hansen, A. and Zielinski, B.S. 2005. Diversity in the olfactory epithelium of bony fishes: development, lamellar arrangement, sensory neuron cell types and transduction components. J. Neurocytol. 34(3–5), 183–208. Hara, T.J. 1975. Olfaction in fish. Progress Neurobiol. 5(4), 271–335. Hara, T.J. 2011. Morphology of the olfactory (smell) system in fishes. In: Farrell AP (ed) Encyclopedia of fish physiology. San Diego, CA: Academic Press, pp: 194–207. Hara, T.J. and Zielinski, B.S. 1989. Structural and functional development of the olfactory organ in teleosts. Trans. Amer. Fisher. Soc. 118(2), 183–194. Hawkes, J.W. 1974. The structure of fish skin. I. General organization. Cell Tissue Res. 149(2), 147–158. Ichikawa, M. and Ueda, K. 1977. Fine structure of the olfactory epithelium in the goldfish, Carassius auratus. A study of retrograde degeneration. Cell Tissue Res. 183, 445–455. Jang, W., Chen, X., Flis, D., Harris, M. and Schwob, J.E. 2014. Label-retaining, quiescent globose basal cells are found in the olfactory epithelium. J. Comp. Neurol. 522(4), 731–749. Kasumyan, A.O. 2004. The olfactory system in fish: Structure, function and role in behavior. J. Ichthyol. 44(2), S180–S223. Khedkar, G.D., Jadhao, B.V., Chavan, N.V. and Khedkar, C.D. 2003. Fish demersal species of temperate climates. In: Encyclopedia of foods and nutrition, 2nd ed. Eds., Caballero, B., Trugo, L. and Finglas, P.M. London, UK: Elsevier and Academic Press Publishers. Kim, H.T., Yun, S.W. and Park, J.Y. 2019. Anatomy, ultrastructure and histology of the olfactory organ of the largemouth bass, Micropterus salmoides, Centrarchidae. Appl. Microsci. 49, 18. Kuciel, M., Lauriano, E.R., Silvestri, G., Zuwała, K., Pergolizzi, S., and Zaccone, D. 2013. The structural organization and immunohistochemistry of G-protein alpha subunits in the olfactory system of the air-breathing mudskipper, Periophthalmus barbarus (Linnaeus, 1766) (Gobiidae, Oxudercinae). Acta Histochem. 116(1), 70–78. Lam, P., Mangos, S., Green, J.M., Reiser, J. and Huttenlocher, A. 2015. In Vivo imaging and characterization of actin microridges. PLoS One 10(1), e0115639. Mehanna, S.F. 2006. Fisheries regulations based on yield per recruit analysis for the spotted Seabass, Dicentrarchus punctatus (Moronidae) at Bardawil Lagoon, Mediterranean coast of Sinai, Egypt. Egy. J. Aquat. Biol. Fishier. 10(4), 129–145. Menco, B.P. and Morrison, E.E. 2003. Morphology of the mammalian olfactory epithelium form, Fine Structure, Function and Pathology. In: Handbook of Olfaction and Gustation, 2nd ed. Ed., Doty R.L. New York, NY: Marcel Dekker, Inc. pp: 17-49. Miller, B. and Kendall, A.W. 2009. Early life history of marine fishes. 1st ed, Berkeley, CA: Univesrsity of California Press, pp: 376. Moretti, A., Fernandez-Criado, M.P., Cittolin, G. and Guidastri, R. 1999. Manual on hatchery production of seabass and gilthead seabream. Volume 1. Rome: FAO. pp: 194. Muller, J.F. and Marc, R.E. 1984. Three distinct morphological classes of receptors in fish olfactory organs. J. Comp. Neurol. 222(4), 482–495. Nelson, J.S., Grande, T. and Wilson, M. 2016. Fishes of the world. 5th ed. Hoboken, NJ: Wiley, pp: 495–497. Newbury, D.E. and Yakowitz, H. 1975. Specimen preparation, special techniques and applications of the scanning electron microscope. In: Practical scanning electron microscopy-electron and ion microprobe analysis. 1st ed, Eds., Goldstein, J.I. and Yakowitz, H. New York and London: Plenum Press, Chapt. VI, pp: 211–262. Olivares, J. and Schmachtenberg, O. 2019. An update on anatomy and function of the teleost olfactory system. Peer J. 7, e7808. Pashchenko, N.I. and Kasumyan, A.O. 2015. Scanning electron microscopy of development of the olfactory organ in ontogeny of grass carp, Ctenopharyngodon idella. J. Ichthyol. 55(6), 880–899. Pashchenko, N.I. and Kasumyan, A.O. 2016. Development of the olfactory organ in the ontogeny of carps (Cyprinidae). J. Ichthyol. 57(1), 136–151. Rheinsmith, S.E., Quinn, T.P., Dittman, A.H. and Yopak, K.E. 2022. Ontogenetic shifts in olfactory rosettemorphology of the sockeye salmon, Oncorhynchus nerka. J. Morphol. 284, e21539. Ridley, C. and Thornton, D.J. 2018. Mucins: the frontline defence of the lung. Biochem. Soc. Transform. 46(5), 1099–1106. Rygg, A.D. van Duin, A.C. and Craven, B.A. 2013. Molecular dynamics simulations of water/mucus partition coefficients for feeding stimulants in fish and the implications for olfaction. PLoS One, 8, e72271. Samajdar, I. and Mandal, D.K. 2016. Histological organization and ultrastructures of the apical surface of the olfactory epithelium of a carp, Labeo bata (Hamilton). Inter. J. Pure Appl. Zool. 4(2), 134–141. Tytiuk, O.K., Sapoznikov, O., Tichy, F., Klouckova, M. and Stepanyuk, Y. 2020. Morphogenesis of olfactory organ of Bushymouth catfish, Ancistrus dolichopterus (Teleostei: Loricariidae) before switching to exogenous feeding. Zool. Sci., 37, 1–12. Vandeputte, M., Gagnaire, P.A. and Allal, F. 2019. The European seabass: a key marine fish model in the wild and in aquaculture. Animal Genet., 50(3), 195–206. Velez, Z., Hubbard, P.C., Alves, A., Costa, R. and Guerreiro, P.M. 2024. Environmental salinity modulates olfactory sensitivity in the euryhaline European seabass, Dicentrarchus labrax, acclimated to seawater and brackish water. J. Exper. Biol. 227, 3. Vinagre, C., Ferreira, T., Matos, L., Costa, M.J. and Cabral, H. 2009. Latitudinal gradients in growth and spawning of sea bass, Dicentrarchus labrax, and their relationship with temperature and photoperiod. Estuarine Coastal Shelf Sci. 81(3), 375–380. Wakisaka N., Miyasaka N., Koide T., Masuda M., Hiraki-Kajiyama T. and Yoshihara Y. 2017. An Adenosine receptor for olfaction in fish. Current Biol., 27: 1437–1447. http://dx.doi.org/10.1016/j.cub.2017.04.014 Waryani, B., Zhao, Y., Zhang, C., Dai, R. and Abbasi, A.R. 2013. Anatomical studies of the olfactory epithelium of two cave fishes Sinocyclocheilus jii and S. furcodorsalis (Cypriniformes: Cyprinidae) from China. Pak. J. Zool. 45(4), 1091–1101. Watson, J.W., Radford, Z., Bannister, H., Bradley, R., Brown, M., Ciotti, B.J., Goodwin, D., Graham, J.A., Nash, R.D., Roche, W.K., Wogerbauer, C. and Hyder, K. 2024. Assessing the coherence in biological and environmental drivers of young sea bass abundance across important estuarine nursery areas of the northern European sea bass stock. Front. Mar. Sci. 10, 1209311. Whitlock, K.E. and Westerfield, M. 2000. The olfactory placodes of the zebrafish form by convergence of cellular fields at the edge of the neural plate. Development, 127, 3645–3653. Yamamoto, M. and Ueda, K. 1978. Comparative morphology of fish olfactory epithelium. II- Clupeiformes. DOI: https://api.semanticscholar.org/CorpusID:88009432 Yamamoto, M. and Ueda, K. 1979. Comparative morphology of fish olfactory epithelium. X. Perciformes, Beryciformes, Scorpaeniformes, and Pleuronectiformes. J. Fac. Sci. Tokyo Univ. Zool. 14(3), 273–297. Yamamoto, Y., Mori, M., Amano, M., Yamanome, T., Saito, S., Taniguchi, K., Yamamori, K. and Taniguchi, K. 2004. Morphogenesis of the olfactory pit in a flatfish, barfin flounder (Verasper moseri). J. Vet. Med. Sci. 66(10), 1275–1278. Zanuy, S., Carillo, M., Ruiz, F. 1986. Delayed gametogenesis and spawning of seabass (Dicentrarchus labrax L.) kept under different photoperiod and temperature regimes. Fish Physiol. Biochem. 2, 53–63. Zeiske, E., Kasumyan, A., Bartsch, P. and Hansen, A. 2003. Early development of the olfactory organ in sturgeons of the genus Acipenser: a comparative and electron microscopic study. Anat. Embryol. 206(5), 357–372. Zeiske, E., Theisen, B. and Breucker, H. 1992. Structure, development and evolutionary aspects of the peripheral olfactory system. In: Fish chemoreception, (Fish and Fisheries 6. Series ed. T.J. Pitcher), Ed., Hara, T.J. London, UK: Chapman & Hall, pp: 13–39. Zhou, W., Apkarian, R., Wang, Z.L. and Joy, D. 2006. Fundamentals of scanning electron microscopy (SEM). In: Scanning microscopy for nanotechnology. Eds., Zhou, W., Wang, Z.L. New York, NY: Springer. doi: https://doi.org/10.1007/978-0-387-39620-0_1 | ||
| How to Cite this Article |
| Pubmed Style Al-zahaby SA, Al-zahaby AS, Mohamed RA, Elsheikh EH. Ultrastructural and developmental anatomy of the peripheral olfactory organs of Dicentrarchus labrax inhabiting Egyptian Mediterranean water. Open Vet. J.. 2025; 15(2): 939-953. doi:10.5455/OVJ.2025.v15.i2.43 Web Style Al-zahaby SA, Al-zahaby AS, Mohamed RA, Elsheikh EH. Ultrastructural and developmental anatomy of the peripheral olfactory organs of Dicentrarchus labrax inhabiting Egyptian Mediterranean water. https://www.openveterinaryjournal.com/?mno=240419 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.43 AMA (American Medical Association) Style Al-zahaby SA, Al-zahaby AS, Mohamed RA, Elsheikh EH. Ultrastructural and developmental anatomy of the peripheral olfactory organs of Dicentrarchus labrax inhabiting Egyptian Mediterranean water. Open Vet. J.. 2025; 15(2): 939-953. doi:10.5455/OVJ.2025.v15.i2.43 Vancouver/ICMJE Style Al-zahaby SA, Al-zahaby AS, Mohamed RA, Elsheikh EH. Ultrastructural and developmental anatomy of the peripheral olfactory organs of Dicentrarchus labrax inhabiting Egyptian Mediterranean water. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 939-953. doi:10.5455/OVJ.2025.v15.i2.43 Harvard Style Al-zahaby, S. A., Al-zahaby, . A. S., Mohamed, . R. A. & Elsheikh, . E. H. (2025) Ultrastructural and developmental anatomy of the peripheral olfactory organs of Dicentrarchus labrax inhabiting Egyptian Mediterranean water. Open Vet. J., 15 (2), 939-953. doi:10.5455/OVJ.2025.v15.i2.43 Turabian Style Al-zahaby, Sheren A., Al-ahmady S. Al-zahaby, Radwa A. Mohamed, and Eman H. Elsheikh. 2025. Ultrastructural and developmental anatomy of the peripheral olfactory organs of Dicentrarchus labrax inhabiting Egyptian Mediterranean water. Open Veterinary Journal, 15 (2), 939-953. doi:10.5455/OVJ.2025.v15.i2.43 Chicago Style Al-zahaby, Sheren A., Al-ahmady S. Al-zahaby, Radwa A. Mohamed, and Eman H. Elsheikh. "Ultrastructural and developmental anatomy of the peripheral olfactory organs of Dicentrarchus labrax inhabiting Egyptian Mediterranean water." Open Veterinary Journal 15 (2025), 939-953. doi:10.5455/OVJ.2025.v15.i2.43 MLA (The Modern Language Association) Style Al-zahaby, Sheren A., Al-ahmady S. Al-zahaby, Radwa A. Mohamed, and Eman H. Elsheikh. "Ultrastructural and developmental anatomy of the peripheral olfactory organs of Dicentrarchus labrax inhabiting Egyptian Mediterranean water." Open Veterinary Journal 15.2 (2025), 939-953. Print. doi:10.5455/OVJ.2025.v15.i2.43 APA (American Psychological Association) Style Al-zahaby, S. A., Al-zahaby, . A. S., Mohamed, . R. A. & Elsheikh, . E. H. (2025) Ultrastructural and developmental anatomy of the peripheral olfactory organs of Dicentrarchus labrax inhabiting Egyptian Mediterranean water. Open Veterinary Journal, 15 (2), 939-953. doi:10.5455/OVJ.2025.v15.i2.43 |