| Review Article | ||
Open Vet. J.. 2025; 15(10): 4847-4864 Open Veterinary Journal, (2025), Vol. 15(10): 4847-4864 Review Article The role of heat shock protein 70 on oocyte apoptosis during vitrificationRimayanti Rimayanti1*, Aswin Rafif Khairullah2, Sri Pantja Madyawati1, Fedik Abdul Rantam3, Imam Mustofa1, Widjiati Widjiati4, Pudji Srianto1, Adeyinka Oye Akintunde5, Bima Putra Pratama6, Riza Zainuddin Ahmad2, Bantari Wisynu Kusuma Wardhani7, Andi Thafida Khalisa8, Syahputra Wibowo91Division of Veterinary Reproduction, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia 2Research Center for Veterinary Science, National Research and Innovation Agency (BRIN), Bogor, Indonesia 3Division of Veterinary Microbiology, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia 4Division of Veterinary Anatomy, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia 5Department of Agriculture and Industrial Technology, Babcock University, Ilishan Remo, Nigeria 6Research Center for Agroindustry, National Research and Innovation Agency (BRIN), South Tangerang, Indonesia 7Research Center for Pharmaceutical Ingredients and Traditional Medicine, National Research and Innovation Agency (BRIN), Bogor, Indonesia 8Faculty of Military Pharmacy, Universitas Pertahanan, Bogor, Indonesia 9Eijkman Research Center for Molecular Biology, National Research and Innovation Agency (BRIN), Bogor, Indonesia *Corresponding Author: Rimayanti Rimayanti. Division of Veterinary Reproduction, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia. Email: rimayanti [at] fkh.unair.ac.id Submitted: 14/04/2025 Revised: 25/08/2025 Accepted: 12/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
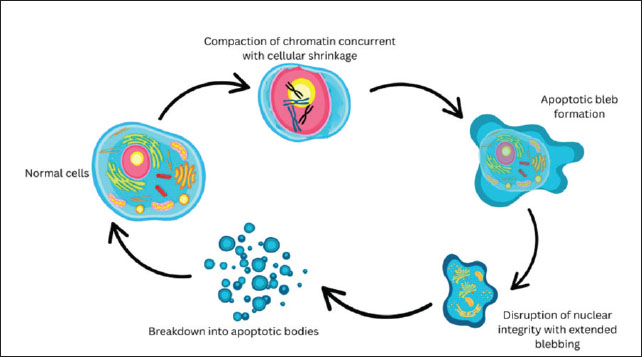
AbstractThe cryopreservation of animal and human oocytes has potential for developing assisted reproductive technologies (ARTs), especially as oocyte bank. One of the techniques used for oocyte cryopreservation is vitrification to avoid the critical temperature and reduce cell damage caused by the formation of ice crystals. Although the oocyte vitrification method is practical, applicable, efficient, inexpensive, and simple, further research is required because of the negative effects of temperature stress. Extreme temperature due to temperature changes from hot-cold-hot in the series of vitrification processes needs to be aware of damage to the structure of cell proteins and DNA. Extreme temperatures can cause stress to oocytes. Stress cells increase the need for heat shock protein 70 (HSP70) as a stress protein. The inductive or synthesized response of HSP70 is considered a protective mechanism of the cells to stress conditions, such as an extreme temperature change. The role of HSP70 in cell protection against extreme temperature changes during oocyte vitrification needs to be assessed by adding HSP70 supplementation into the cryoprotectant vitrification. In conclusion, HSP70 plays a pivotal role in cell protection against stress cells during oocyte vitrification. Fresh oocytes, in vitro maturated oocytes, and postwarming oocytes exhibited HSP70 and Cytochrome c expression. Supplementation of HSP70 into the vitrification medium was expected to suppress the activation of caspase 3, thereby maintaining the viability of oocytes during vitrification and decreasing the incidence of apoptosis after warming. Keywords: Apoptosis, Caspase 3, Cytochrome c, In vitro, Oocyte maturation, Endangered species, Extinction risk. IntroductionOne way to preserve germplasm is by freezing oocytes as an alternative to preserving embryos (Konc et al., 2014). For animal husbandry and veterinary medicine, oocyte cryopreservation has the potential for the development of assisted reproductive technology (ART), especially as an oocyte bank for the preservation of pure lines of rare or conserved animals or endangered species, as well as the provision of oocytes for embryo transfer with various reproductive biotechnology techniques (Tharasanit and Thuwanut, 2021). The success of oocyte cryopreservation is important not only for the preservation or conservation of endangered species but also for the availability of material for research development (Bhat and Sofi, 2021). The risk that needs to be considered in this cryopreservation process is temperature stress, which occurs when returning to normal temperature after thawing or rewarming (Baust et al., 2009). The viability of vitrified oocytes, determined based on their normal morphology, is quite high, but these oocytes do not necessarily have good quality to be able to carry out the fertilization process for the in vitro fertilization (IVF) program, grow and develop (Doyle et al., 2016). This shows that the quality of vitrified oocytes varies even though their viability is quite high. Temperature stress can trigger changes in gene expression, including the induction of heat shock proteins (HSPs) (Boopathy et al., 2022). HSPs are produced not only during the induction of heat shock or heat stress but are also produced continuously under normal conditions (Scieglinska et al., 2019). The response to the increase and induction of HSP70 is thought to be a protective mechanism. Cell protection by HSP70 will refold the protein so that it can function normally. Temperature stress on vitrified oocytes can also cause DNA damage (Souza-Cácares et al., 2019). Cells can provide protection through repair mechanisms and apoptosis (Elmore, 2007). For vitrified oocytes that cannot be repaired, they will be destroyed through apoptosis mechanisms or experience failure in the fertilization and growth process, while those that are able to survive will continue to grow and develop. HSPs can also control the apoptosis mechanism that occurs in these cells (Ledwaba et al., 2025). Oocytes that experience stress due to a series of vitrification processes will cause mitochondria to release cytochrome c, resulting in the activation of apoptosis protease activating factor 1 (APAF-1) and proapoptotic caspase 9 (Wen et al., 2025). The activation of APAF-1 and caspase 9 will be followed by the activation of caspase 3 to induce apoptosis (Bratton and Salvesen, 2010). However, because oocytes experiencing stress have the ability to produce HSP70 during the cell growth stage and store it when they reach oocyte maturation, the HSP70 found in these oocytes will suppress the activation of APAF-1, caspase 9, and caspase 3 so that the apoptosis process does not occur in the oocytes and the viability of the oocytes after thawing is high (Le Masson and Christians, 2011). Conceptually, the cell protection mechanism carried out by HSP70 due to temperature stress in vitrified oocytes, with the ability to assist protein folding and inhibit apoptosis, can be used to explain the cell protection ability, so that it influences the variation in the quality of viable vitrified oocytes (Li et al., 2000). However, the mechanism of variation in the quality of viable vitrified oocytes remains unclear. The unclear mechanism regarding the variation in the quality of vitrified oocytes, even though their viability is quite high, needs to be given attention because it concerns the success of the ART program, as well as the quality of individuals resulting from the ART program to grow and develop healthily in the future. This review article aims to explain the theoretical concept of the involvement and role of HSP70 in vitrification-based oocyte preservation techniques. It is hoped that future laboratory exploration and application opportunities can be carried out to save endangered germplasm and for developing superior livestock seeds. The extrinsic pathway of apoptosis mediated by death receptors can also contribute to cell death under certain conditions (Jan and Chaudhry, 2019). This pathway is typically triggered by ligands such as FasL and TNF-α, which bind to cell surface receptors, activating the death-inducing signaling complex (DISC) and ultimately caspase-8, which can interact directly or indirectly with the mitochondrial pathway via the Bid protein (Mustafa et al., 2024). Exposure to extreme temperatures or osmotic stress during cryopreservation can increase the expression of Fas receptors or TNF-R on the oocyte membrane, suggesting that these pathways may be relevant under certain extreme conditions (Jung and Ryu, 2023; Hai et al., 2024). However, the role of HSP70 in the extrinsic pathway has not been fully elucidated, although evidence suggests that HSP70 may also inhibit caspase-8 activation or stabilize the cell membrane against death receptor activation (Giffard et al., 2008). VitrificationThe cryopreservation method that has received much attention in the last decade is vitrification. The vitrification method is a freezing procedure for preserving cells or tissues, which is considered radical because to avoid the formation of ice crystals, both intracellular and extracellular, the cells or tissues are dipped directly into liquid nitrogen at a temperature of −196°C and using high concentration cryoprotectants (30%–50%), which are feared to cause toxicity to the frozen cells (Valcarce et al., 2013). This method aims to eliminate ice crystal formation and simplify the cryopreservation process (Chang and Zhao, 2021). Many studies have been conducted to shorten the time of conventional freezing procedures and reduce the high costs by using programmable freezing equipment (Bojic et al., 2021; Chen et al., 2022; Pezo et al., 2023). Compared with conventional freezing methods, vitrification is an efficient cryopreservation method because it is much simpler, easier to perform, and cheaper because it does not require expensive, sophisticated equipment and does not require a certain amount of time in the freezing process (Araújo-Lemos et al., 2015). A practical and applicable method is needed in the field, especially for the interests of the world of veterinary medicine and animal husbandry, so that this vitrification method can be used as an alternative (Sanchez-Osorio et al., 2010). The mechanism that occurs in the vitrification method is the solution’s solidification due to increased viscosity during freezing, so that the solution appears to become glass-like (Roque-Borda et al., 2021). Therefore, instead of the slow dehydration process in conventional freezing procedures, the material to be frozen by the vitrification method is placed in a hyperosmolar medium or a high concentration of cryoprotectant (Jaiswal and Vagga, 2022). After the material is dipped directly into liquid nitrogen, rapid dehydration occurs, no intra-extracellular ice crystals are formed, and the solution becomes glass-like (Isachenko, 2004). Increasing the cooling rate at the critical temperature (20°C–12°C) is a determining factor in the success of bovine oocyte cryopreservation and is not due to the reduction in the formation of ice crystals during vitrification (Díez et al., 2012; Pardede et al., 2023). The detrimental effects of vitrification are caused by toxicity and osmotic injury when cells are exposed to high concentrations of cryoprotectants, thereby affecting the survival ability of these cells (Prentice-Biensch et al., 2012). Vitrification solutions must contain one or more high-concentration intracellular cryoprotectants, each solution requires a physiological salt component, and macromolecules must be added to the solution to increase the tissue’s ability to withstand freezing conditions, such as glass (Kasai and Mukaida, 2004; Wang et al., 2022). The freezing and re-thawing stages are critical in the cell freezing process (Cottle et al., 2022). The cryoprotectants used are divided into two types: micromolecular cryoprotectants, which function as intracellular cryoprotectants, and macromolecular cryoprotectants, which function as extracellular cryoprotectants (Bissoyi et al., 2023). Intracellular cryoprotectants have small molecular sizes, so they can enter and exit through cell membranes, whereas macromolecular cryoprotectants have large molecules, so they cannot penetrate cell walls to balance osmosis between inside and outside the cell (Pegg, 2015). The type and concentration of cryoprotectant, the length and temperature of exposure of cells to cryoprotectant before being dipped in liquid nitrogen and at the time of re-thawing, and the level of cell maturity all influence the viability of vitrified cells (Meneghel et al., 2020). The approach taken to avoid chilling injury by increasing the freezing speed is carried out using several methods, including Open Pulled Straw (OPS), flexipet-denuding pipette (FDP), microdrops, EM copper grid, nylon mesh, cryoloop, and minimum volume cooling using crypto (Liebermann and Tucker, 2002). Currently, there is no universal vitrification procedure standard. Researchers need to obtain consistent results using a standard vitrification procedure that can be applied for cryopreservation at various stages of cell division. Oocyte vitrificationRapid developments in gamete cryopreservation have occurred. Frozen semen and embryo cryopreservation have been widely used in recent decades (Estudillo et al., 2021). Moreover, embryo cryopreservation has been widely used as an assisted reproductive technology in IVF programs for livestock and humans (Konc et al., 2014). Confidence in embryo cryopreservation techniques continues to increase because embryo viability is relatively consistent, and application in livestock is commercially acceptable. The provision of embryos using cryopreservation techniques, which have been widely used in human IVF programs, has apparently caused problems beyond technical matters because it is considered unethical and is prohibited in several countries (Bankowski et al., 2005). Therefore, research is directed at oocyte cryopreservation as an alternative to embryo preservation. The provision and storage of oocytes for human IVF programs is related to saving IVF costs and the availability of oocyte cells in the event of failure in the IVF program, as well as storing oocytes before a woman undergoes therapy or chemotherapy, which could damage her oocyte cells (Das and Son, 2023). Pai et al. (2021) also stated that oocyte cryopreservation can improve medical assistance for couples who have difficulty conceiving by forming an oocyte bank to increase the chances of ART success. In the field of animal husbandry, oocyte cryopreservation can be used as an alternative for providing gametes and developing the field of reproductive biotechnology, for example, cloning by nuclear transfer and production of transgenic animals (Huang et al., 2018). Unlike embryo cryopreservation, the problems associated with oocyte cryopreservation have not yet been resolved. Studies on oocyte cryopreservation often have unpredictable results, and the commercial use of this cryopreservation procedure is still very limited and challenging due to the complex structure of oocytes (Chronopoulou et al., 2021). The properties of oocytes and embryos, such as size, shape, permeability, quality, and sensitivity, determine the most appropriate conditions for successful cryopreservation, depending on the species, developmental stage, and origin. Cryopreservation conditions are different for each oocyte and embryo (Rienzi et al., 2017). In early studies, oocyte cryopreservation was mostly performed using slow and conventional freezing procedures (Konc et al., 2008; Argyle et al., 2016). The fertilization ability of frozen and thawed bovine oocytes was lower than that of fresh or unfrozen oocytes (Xu et al., 2022; Park et al., 2022). This is likely due to disruption of the plasma membrane, extensive disorganization of the ooplasm, damage to the spindle, and structural changes in the zona pellucida during the clotting process (Litscher et al., 2009). Meanwhile, according to Gil et al. (2014) it is suspected that the factor that causes oocytes to lose their viability is related to the gradual freezing process to −7°C, then continuing to −35°C. These researchers concluded that the significant loss of oocyte viability during the cryopreservation process was due to freezing and ice crystal formation. Efforts to develop oocyte freezing techniques continue to be conducted by simplifying the procedure. Feng et al. (2025) and Zhou et al. (2016) conducted studies on freezing mouse oocytes using vitrification techniques, and the results showed good development in mouse oocytes fertilized in vitro after the vitrification process. Hochi et al. (1994) studied the viability of immature horse oocytes after cryopreservation using the vitrification method. The cryoprotectants used were 40% ethylene glycol, 18% ficoll 70, and 0.3 M sucrose. These immature oocytes were frozen by vitrification after being exposed for 10–20 min in ethylene glycol at a temperature of 20°C–30°C. The results show that oocytes are alive and developing until they reach the metaphase II stage after 32 h of in vitro maturation. Rienzi et al. (2010) showed that the normal morphology of vitrified oocytes was significantly higher than that of slow-frozen oocytes. The developmental competence of oocytes after fertilization did not differ between the two cryopreservation techniques, although spindle disorders were more common in the slow-frozen oocyte group. The vitrification method showed a better frequency of spindle development to normal shape after thawing than slow freezing. Concentration and type of cryoprotectant have an important influence on the development competence of vitrified oocytes. As proven by García-Martínez et al. (2021) and Du et al. (2024) demonstrated the use of ethylene glycol cryoprotectants with various formulas and concentrations for oocyte vitrification. Yassin et al. (2022) showed that 40% and 50% ethylene glycol cryoprotectants had a better effect on the viability of mature bovine oocytes after the vitrification process than 20% and 30% ethylene glycol. The use of EG as an intracellular cryoprotectant in vitrification media is believed to be a major factor in increasing fertilization success, as also expressed by Chi (2002). In addition to conventional cryoprotectants, recent studies have indicated that the protective efficacy of HSP70 supplementation is also concentration-dependent. Low concentrations may be insufficient to stabilize protein structures and inhibit apoptosis, whereas excessive levels could disrupt cellular homeostasis or induce cytotoxic effects. Therefore, establishing an optimal HSP70 concentration gradient in vitrification solutions is crucial to maximize oocyte survival and developmental competence while minimizing unintended side effects (Wang et al., 2012 ; Li et al., 2024). There are also reports that adding sugar to vitrification media will increase survival after re-thawing and at the embryo development stage (Elnahas et al., 2010). McWilliams et al. (1995) and Huang et al. (2008) also proved that various saccharides added as extracellular cryoprotectants (non-permeating saccharides) can be osmotic buffers for the recovery of oocyte, zygote, and embryo conditions after vitrification. The exposure time of the cryoprotectant before the vitrification process also affects the oocyte viability. Dhali et al. (2000) showed that an exposure time of 3 min can be used for vitrification of buffalo oocytes, while García-Martínez et al. (2022) reported that exposure times of 10, 20, and 40 min showed good results on the morphology of bovine oocytes compared with an exposure time of 5 min before quick freezing. Meanwhile, Otoi et al. (1998) showed different results. In the cryopreservation of mature bovine oocytes using the vitrification method, exposure times of 1, 5, and 10 min were not significantly different. The maturity level of the oocytes undergoing the freezing process also influences the success of vitrification. Rodriguez-Villamil et al. (2016) investigated the potential survival of bovine oocytes at several levels of maturity after the vitrification and re-thawing process and concluded that the survival of immature bovine oocytes is lower than that of mature bovine oocytes. All mature oocytes in metaphase II are sensitive to damage due to the freezing process because the spindle that holds the chromosomes depolymerizes when it reaches a lower temperature (Gomes et al., 2008). Cooling may increase the incidence of aneuploidy because chromosome reunification may not be precise in the newly reformed spindle, although the spindle returns to its original state when the temperature returns to normal (Duncan et al., 2009). Although the spindle of mature oocytes is damaged due to the freezing process, the survival of mature oocytes is still better than that of immature oocytes (Martínez-Burgos et al., 2011). Yamanaka et al. (2007) also stated that oocytes in the germinal vesicle phase (GV stage) are more sensitive to cryoprotectants than oocytes that have been matured in vitro. Zhu et al. (1998) performed cryopreservation on mature bovine oocytes using the step-freezing and vitrification technique. The cryoprotectants used were 1.6 M propanediol and 0.5 M sucrose with exposure times of 10 and 20 min, respectively. Although most oocytes (86.1%) were viable as determined based on their normal morphology, their developmental competence was severely impaired. The average fertilization and division rates after vitrification were only 30% and 7.4%, respectively. Ultrastructural observation by electron microscopy showed a more severe subcellular damage in step freezing compared with that in vitrified oocytes. Severe damage was observed in the mitochondria and vesicles. Meanwhile, the cortical granules underwent exocytosis in the 2 freezing methods. Decreased developmental capacity after cryopreservation is mainly due to zona pellucida hardening and cortical granule exocytosis. Cortical granules are one of the determining elements of the fertilization ability of frozen bovine oocytes. Vitrification also damages microtubules and mitochondria (Gutierrez-Castillo et al., 2022). Considering such conditions, various techniques and methods are being developed to minimize the negative effects of the freezing process on the viability and development ability of cells. One technique to reduce the occurrence of damage due to freezing on embryos and oocytes is by pulling the straw so that it shrinks, lengthens, and its walls become thinner (Estudillo et al., 2021). This technique is known as Open Pulled Straw (OPS) and was carried out and patented by Vajta et al. (1998) and tried by Al Yacoub et al. (2010) with satisfactory success. This OPS technique can cause the freezing speed to occur very quickly, making it possible to avoid damage due to freezing and reduce toxicity and osmotic impact (Amini and Benson, 2023). Vajta et al. (1998) reported that of 184 vitrified oocytes, 25% developed into blastocysts after in vitro fertilization and were followed by pregnancy. However, de De Leon et al. (2012) still doubted the beneficial effects of this technique. Interspecies variability in vitrification success is an important factor to consider when interpreting results. For example, murine oocytes tend to have a higher tolerance to vitrification than bovine oocytes, which are more structurally and physiologically complex. Bovine oocytes have a larger cytoplasmic volume, higher metabolic activity, and a spindle microtubule and organelle arrangement that is more susceptible to extreme temperature and osmotic changes, making them more susceptible to damage during flash freezing (Kyogoku and Kitajima, 2023). Furthermore, differences in the plasma membrane composition and expression of protective proteins (such as HSP70, aquaporins, and endogenous antioxidants) can also influence post-vitrification survival (De Maio and Hightower, 2021). Therefore, while murine model results can provide valuable baseline information, direct extrapolation to other species, particularly larger animals like cattle or humans, should be approached with caution. Cross-species validation, taking into account specific oocyte conditions, such as maturation stage, vitrification medium, and rewarming protocol, is necessary to ensure the consistency and biological acceptability of the results (Costa-Borges et al., 2025). Cellular Response to StressThe ability of cells to adapt and survive stressful conditions is a fundamental requirement for cell survival. Injury agents can come from inside (endogenous) or outside the cell (exogenous). Endogenous injury is caused by genetic abnormalities, such as gene mutations, chromosome abnormalities, and transcription and translation errors (Miller and Zachary, 2017). Meanwhile, exogenous damage is due to the influence of agents such as chemical substances, physical agents, microorganisms, radiation, temperature, pH, nutritional deficiencies, loss of vital components (hypoxia, ischemia, and starvation), and immunological reactions (Miller and Zachary, 2017). These various injury agents are stressors that can cause mild or severe imbalances, which may or may not be tolerated by the body and ultimately result in cell death (Kalogeris et al., 2012). Cell death can occur through two mechanisms, namely, apoptosis and necrosis. Apoptosis is a programed cell death (PCD) that occurs physiologically normally, for example, in the process of cell replacement in damaged cells, maintaining cell or tissue homeostasis, embryogenesis, induction and maintenance of the immune system, and cell atrophy due to hormonal processes (Elmore, 2007). Apoptosis mechanisms can also occur due to pathological conditions such as radiation, environmental toxicants, oxidative stress, and temperature stress (Redza-Dutordoir and Averill-Bates, 2016). Meanwhile, necrosis is cell death caused by infection and severe injury (Yang et al., 2015). Excessive stress induces changes in gene expression as a cell response to conditions outside normal limits. According to Almanza et al. (2019), this stress condition induces apoptosis. In cold shock conditions, a proapoptotic effect occurs, which depends on the extent of exposure, intracellular Ca2+ level, cell cycle phase, and cytoskeletal stability. The changes in cells undergoing apoptosis are as follows: Cells experience dehydration so that their shape and size shrink, chromatin condensation starts from the edge of the nucleus along with the disappearance of the nuclear membrane, chromatin fragmentation is a characteristic of apoptosis, the formation of "apoptotic bodies" which are cell membranes that envelop chromatin fragments, cytoplasm, and organelles that remain functional (intact organelles), the process of phagocytosis of "apoptotic bodies" by the phagocyte system (Elmore, 2007). Figure 1 shows the apoptosis process.
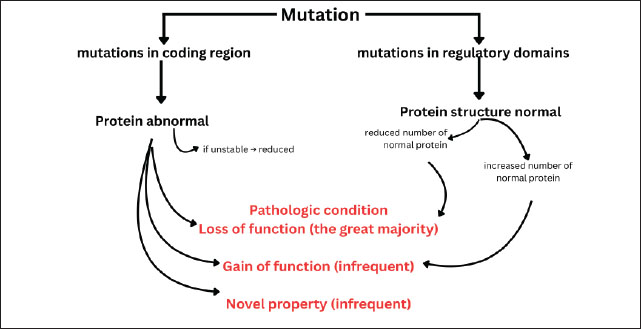
Fig. 1. Morphological changes during apoptosis. Normal cells undergo chromatin compaction concurrent with cellular shrinkage, followed by apoptotic bleb formation. Subsequent disruption of nuclear integrity with extended membrane blebbing leads to cell fragmentation into apoptotic bodies, which are ultimately phagocytosed by surrounding cells or macrophages. The occurrence of apoptosis is marked by a typical biochemical marker, namely DNA fragmentation, which was confirmed using the TdT-mediated dUTP nick end labeling method (Denton and Kumar, 2015). Several pathological conditions, such as radiation, environmental toxicants, oxidative stress, and temperature stress, can initiate apoptosis in mammalian oocytes (Roth and Hansen, 2004). Similarly, oocytes are exposed to several stressors during cryopreservation, such as temperature, osmotic, and toxicity, which are never experienced physiologically (Comizzoli et al., 2008). Using the TUNEL method, it is possible to determine which oocytes have undergone apoptosis from the visible nuclear color (Ghafari et al., 2007). Cells undergoing apoptosis will have a brown nucleus and a granular appearance due to DNA fragmentation (Schulze-Osthoff et al., 1994). Apoptosis occurs through several pathways. The pathway that leads to apoptosis can be passed through the interaction between the death ligands, namely Fas ligand (FasL) and Tumor Necrosis Factor α (TNF α) with their receptors. Fas receptors and TNF receptors will trigger the activation of proteases (Arguello and Haisma, 2021). Activation of procaspase 8 will break down Bid, causing the release of cytochrome c from mitochondria, which will trigger the formation of the APAF-1 and caspase 9 complex, and protease activity will continue until it ends in the destruction of the cell structure or apoptosis (Li et al., 1997). In addition, apoptosis can also occur if mitochondria are damaged so that the MPTP opens. The opening of this MPTP will cause the secretion of cytochrome c and other molecules, namely APAF-1, which together with caspase 9 will activate caspase 3 (Yapryntseva et al., 2024). Temperature stress on vitrified oocytes can also cause DNA damage. DNA-encoded genetic information is a "blueprint" for protein production (Walker and Biase, 2020). Therefore, the function of a gene can be inferred from its nucleotide sequence. For example, the suspected amino acid sequence can be compared with the known gene’s protein sequence. Significant sequence similarities indicate the same protein function (Pearson, 2013). If there is an error in encoding the genetic information of this protein synthesis, the protein will not normally perform its biological function. This damage can be small-scale damage that is classified as a point mutation, frameshift mutation, or damage to macrochromosomal structures, such as translocations and deletions (Chatterjee and Walker, 2017). Although both belong to the HSP70 protein family, fundamental differences exist between the inducible form of HSP72 and the constitutive form of HSC70. HSP72 is the induced form specifically in response to cellular stress, such as heat, oxidative stress, or vitrification. Its expression is controlled by heat shock factor 1 (HSF1) and increases sharply when cells encounter extreme conditions (Volloch et al., 2000). HSP72 functions primarily to protect cells from damage by assisting in the refolding of denatured proteins, preventing protein aggregation, and inhibiting apoptotic pathways (Park et al., 2001). In contrast, HSC70 is a constitutively expressed form, indicating that it is always present in cells even in the absence of stress. Its primary role is as a housekeeping protein, such as assisting in the folding of new proteins, interorganelle protein transport, and misfolded protein degradation (Chen and Brown, 2007). In the context of stress responses, HSP72 acts as an emergency protector, whereas HSC70 ensures protein stability and daily function (Xu et al., 2011). The two work together to maintain cellular homeostasis. Notably, HSP70 expression is not uniform across oocyte developmental stages. During the germinal vesicle (GV) stage, oocytes actively synthesize and accumulate HSP70, providing a reservoir of protective proteins that can be mobilized under stress conditions. In contrast, metaphase II (MII) oocytes largely rely on pre-accumulated HSP70 stores and show limited inducibility, which may explain their higher susceptibility to cryoinjury during vitrification. This stage-dependent pattern highlights the importance of timing and developmental context in determining the protective efficacy of HSP70 during cryopreservation (Neuer, 2000; Lánská et al., 2006; Miętkiewska et al., 2022). The effects of mutations on protein function are shown in Figure 2. Mutations that occur in the coding region produce proteins with abnormal structures that will lose or increase their function or have new functions but cause abnormalities or diseases (Gerasimavicius et al., 2022). Mutations in regulatory domains can cause proteins to lose or increase their function. Mutations in the coding region or regulatory domains can reduce the amount of protein (Vihinen, 2021).
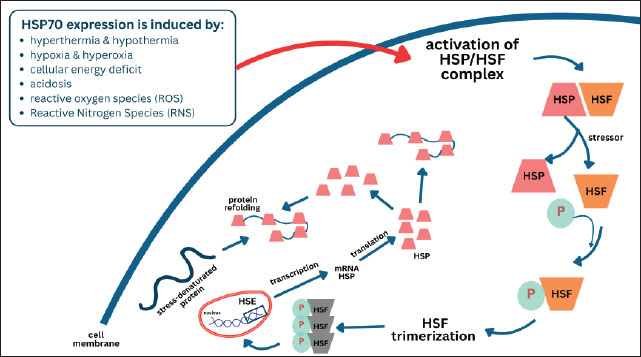
Fig. 2. Mechanisms by which mutations lead to the development of pathological conditions. Mutations in coding regions often produce abnormal proteins that cause function loss, function gain, or novel properties. Mutations in regulatory domains typically maintain protein structure but alter protein levels, which can also result in disease. Heat shock proteins 70One of the responses of cells when experiencing stress, whether in the form of physiological or pathological stress, is the formation of a group of proteins called HSPs (Hu et al., 2022). Exogenous injury agents in the form of temperature stress can trigger changes in gene expression, including the induction of HSPs (Singh et al., 2024). Sonna et al. (2002) stated that there are actually around 100 genes that are expressed when exposed to heat stress and around 20 genes when exposed to cold stress, and HSP genes are expressed in both cells exposed to heat and cold stress. HSP 70 is one of the most sensitive HSPs to temperature changes and is “highly conserved” (Xu et al., 2020). This protein is an ATP-binding protein, and there are at least 4 proteins in the HSP70 group: HSP72, HSP73, HSP75, and HSP78 (Singh et al., 2024). These proteins are synthesized in response to various stimuli. For example, the 73-kDa protein (HSP73 or Hsc 70) is constantly produced, or what is known as constitutive, whereas the 72-kDa protein (HSP72 or HSP70) is highly inducible and its synthesis will increase in response to various stressors (Shin et al., 2004). Therefore, the production of group with HSP is not only due to induction from heat shock or heat stress but is also produced continuously even under normal conditions (Scieglinska et al., 2019). Physiological conditions include development, growth, differentiation, cell cycle, and the presence of oncogenes/protooncogenes, whereas environmental stress is caused by changes in temperature, oxidative stress, nutritional deficiencies, ultraviolet radiation, exposure to chemicals, viral infections, and ischemia-reperfusion conditions (Latchman, 2001). Pathological conditions that stimulate the emergence of HSPs include tissue injury/repair of damaged tissue, neuronal injury, aging process, ischemic/hypoxic, oxidant injury, hypertrophy, fever/inflammation, viral or bacterial infection (Tytell and Hooper, 2001). Therefore, group with HSP play a role in protecting against several stressful conditions. HSPs have biochemical activity as molecular chaperones (HSP40, HSP60, HSP70, and HSP90), repairing proteins that experience misfolding, unfolding, or abnormal folding, protein synthesis, transport, and protein translocation processes, and preventing protein aggregation by participating in protein synthesis, protein folding, transport, and protein translocation processes (Singh et al., 2024). HSP genes are activated after stimulation of the transcription factor, namely HSF (Gomez-Pastor et al., 2018). The central process of HSF activation depends on the balance between the binding of free HSP molecules to HSF and stress-mediated unfolding proteins (Szyller and Bil-Lula, 2021). However, it is not known exactly how stressors can activate HSF. Increased protein damage or abnormalities induce the emergence of a group with HSP (Kalmar and Greensmith, 2009; Hashikawa et al., 2021). The HSP70 activation pathway is initiated when cells experience various physiological or environmental stresses, such as hyperthermia, hypothermia, hypoxia, energy deficit, accumulation of reactive oxygen species (ROS), or acidosis (Wang et al., 2012). In this initial stage, the stressor causes denaturation of intracellular proteins, which then triggers the release of HSF1 from an inactive complex with HSP70 in the cytoplasm (Singh et al., 2024). Once released, HSF1 undergoes activation through phosphorylation, which then induces HSF1 trimerization into its active form. This HSF1 trimer then translocates to the cell nucleus and binds to a specific DNA element called the HSE in the HSP gene promoter (Verma et al., 2014). This binding induces transcription of the HSP70 gene, producing mRNA that is then translated into the HSP70 protein in the cytoplasm. Once synthesized, HSP70 plays a crucial role as a molecular chaperone, stabilizing and assisting in the refolding of denatured proteins, preventing protein aggregation, and maintaining cellular integrity (Singh et al., 2025). Furthermore, HSP70 has anti-apoptotic functions, including inhibiting the release of cytochrome c from mitochondria, interfering with the activation of the apoptosome complex (APAF-1 and caspase-9), and suppressing the activation of caspase-3, the executor of apoptosis (Jäättelä et al., 1998). The scope of HSP70 supplementation in cryobiology has expanded considerably in recent years, with several studies confirming its role in diverse cell types beyond oocytes. In spermatozoa, exogenous HSP70 improves post-thaw motility, maintains acrosomal integrity, and reduces ROS-induced DNA damage, thereby enhancing fertilization potential (Zhou et al., 2016). Similarly, HSP70 supplementation stabilizes the cytoskeleton and improves blastocyst development rates after vitrification and warming, underscoring its importance in early embryogenesis (Stamperna et al., 2021). HSP70 enrichment during cryopreservation was associated with better preservation of mitochondrial function and reduced apoptotic signaling in somatic cells, including stem cells and fibroblasts (Costa-Borges et al., 2025). Collectively, these findings indicate that the protective action of HSP70 is not limited to oocytes but rather represents a broadly conserved cytoprotective mechanism across mammalian cell types. This growing body of evidence highlights the translational potential of HSP70 as a universal supplement in cryopreservation protocols, although optimization strategies may need to be tailored according to cell type and physiological context. The intrinsic apoptosis pathway is initiated by various forms of cellular stress that cause mitochondrial damage and the release of cytochrome c into the cytoplasm (Wu and Bratton, 2013). Then, cytochrome c binds to apoptotic protease-activating factor-1 (APAF-1) and ATP, forming a complex called the apoptosome (Zhou et al., 2015). This complex recruits and activates caspase-9, which in turn activates caspase-3, the primary apoptosis executor responsible for structural protein degradation, DNA breakdown, and ultimately cell death (Prokhorova et al., 2015). HSP70 plays a crucial role in interrupting this pathway at several critical points. First, HSP70 can inhibit the release of cytochrome c from the mitochondria by stabilizing the mitochondrial membrane and preventing the formation of the permeability transition pore (Stankiewicz et al., 2005). Second, once cytochrome c is already in the cytoplasm, HSP70 can directly bind APAF-1, thereby inhibiting apoptosome formation (Bratton and Salvesen, 2010). Caspase-9 activation is also blocked by preventing the formation of this complex. Third, HSP70 also indirectly inhibits caspase-3 activation by suppressing the expression of pro-apoptotic factors or by interacting with other proteins that regulate the caspase pathway (Lanneau et al., 2008). Figure 3 describes the HSP70 expression mechanism. If stress occurs in cells under physiological conditions, such as hyperthermia, hypothermia, hypoxia, hyperoxia, viral infection, acidosis, ROS, energy loss, and ischemia-reperfusion, the inductive form of HSP70 will be activated, and its expression in cells will increase (Szyller and Bil-Lula, 2021). HSF-1 found in the cytosol, is bound to group with HSP in an inactive state. The presence of stressors activates HSF-1 and causes HSF-1 to separate from HSP70 (Kmiecik et al., 2020). Protein kinase phosphorylates HSF-1 and trimerizes it in the cytosol. This HSF-1 trimer complex enters the cell nucleus and binds to the HSE in the promoter region of the HSP70 gene. HSP70 mRNA undergoes transcription and enters the cytoplasm to be synthesized and translated into new HSP70 (Masser et al., 2019).
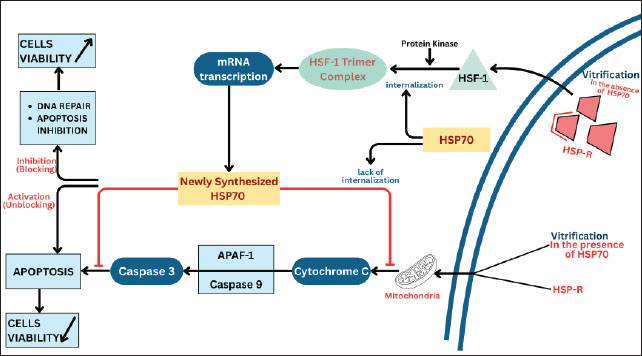
Fig. 3. Activation of the HSF/HSP complex and HSP70 induction. Stressors activate HSF, resulting in its trimerization and binding to HSE, which drives HSP70 transcription. The synthesized HSP70 assists protein refolding and protects cells from stress-induced damage. HSP genes exhibit several properties that allow them to be translated into proteins in situations where other genes’ translation is inhibited. This property is illustrated in the graded response of Drosophila cells exposed to hyperthermia (Pirkkala et al., 2001). Moderate temperature increases (29°C–30°C) cause increased synthesis of HSP mRNA. At higher temperatures (34°C–36°C), normal mRNA translation into protein is inhibited, but HSP mRNA is not. At higher temperatures (37°C–39°C), normal splicing of precursor mRNA is inhibited, but the transcription of most heat-inducible genes escapes inhibition because they lack introns and do not require splicing (Boopathy et al., 2022). The temperature at which HSP synthesis begins varies among species. It is usually several degrees above the normal body temperature of the species (Tomanek, 2010). Protein synthesis involves a very complex process consisting of transcription and translation processes. Protein folding is related to the methylation process that occurs during translation (Waudby et al., 2019). In the translation process, an adaptor factor that acts as a messenger for mRNA to be encoded in the ribosome is needed. This adaptor factor is played by tRNA, which consists of a single chain and comprises 73–93 ribonucleotides (Tamura, 2015). The 5’ end is phosphorylated, and the 3′ end has one free hydroxyl group. This tRNA contains several nucleotides with bases that are not commonly found in other biological units, such as inosine, pseudouridine, dihydrouridine, ribothymidine, and guanosine methyl derivatives (Helm, 2006). Most of these bases result from enzymatic modification by an RNA precursor that catalyzes the methylation of nucleotides A, U, G, and C (Moore et al., 2013). Methylation is performed to prevent base pairs that interfere with the role of tRNA as an adaptor molecule (Wu et al., 2024). Methylation also allows the emergence of hydrophobic properties in certain parts of the tRNA molecule that will play an important role in the interaction with synthetase enzymes and ribosomal proteins (Gomez and Ibba, 2020). The methylation process is required by many proteins to perform their cellular functions (Małecki et al., 2022). Some possible mechanisms for cell protection include the binding of HSP70 to parts of the unfolded protein that are rich in hydrophobic residues produced by the methylation process (Mayer and Bukau, 2005). The energy of ATP hydrolysis is then used to release the HSP from its substrate, and the resulting changes are thought to increase the likelihood of the denatured protein refolding to its original shape (Szabo et al., 1994). Braakman and Hebert (2013) stated that there is a protein folding process from polypeptides that have been in the ER with the help of HSPs and ATP. HSP70 has an affinity for the hydrophobic part of denatured or mutated proteins, even in normal proteins. HSP70 maintains certain proteins in an unfolded state until these proteins undergo transport through the membrane into the ER or mitochondria (Saibil, 2013). Oocyte protection by HSP70 during vitrificationThe negative effects of the vitrification method on oocytes are in the form of cell stress caused by osmotic injury, toxic injury, and temperature injury (Chang et al., 2022). The presence of osmotic damage, cryoprotectant toxicity, and damage due to changing temperatures from hot to cold to reheating in the vitrification process series, namely in vitro maturation, vitrification, and rewarming or thawing, necessitates attention to damage to cell organelles and cell protein structures (Li et al., 2024). Damage to cell organelles and cell protein structures greatly affects oocyte viability, both after vitrification, during IVF, and during the embryo development phase (López et al., 2021). Until now, how the HSP70 protection mechanism works against cell stress is unknown. Cell stress and the number of denatured or damaged proteins increase the need for a group with HSP (Hu et al., 2022). It is suspected that the cell protection mechanism carried out by HSP70 due to temperature stress on the cell stress with the ability to help protein folding and inhibit apoptosis (Mosser et al., 2000). Several studies have been conducted to prove the expression of HSP70 under extreme temperature conditions. Vendrell-Flotats et al. (2017) evaluated the effects of heat stress on oocyte function after in vitro maturation as assessed by cell development and division, then related to the effects on membrane integrity and protein synthesis. The results showed that oocyte thermolability may be related to changes in protein synthesis and the absence of the ability to induce group with HSP. Furthermore, heat stress in bovine embryos induces changes in protein synthesis and possibly gene expression in the developing 2-cell embryo. Cumulus cells play a thermoprotective role during maturation. The presence of HSP70 was also reported by Lang et al. (2000). The results showed that the expression of HSP70 mRNA controls the condition of the embryo so that it responds to heat stress by inducing HSP70 mRNA. The presence of HSP70 in oocytes and embryos showed that the distribution of HSP70 in the ooplasm of mature oocytes and immature oocytes was not affected by exposure to high temperatures, and this protein is adjacent to the spindle, indicating that HSP70 possibly plays a role in stabilizing the spindle structure. HSP70 is distributed in the cytoplasm of 8-cell embryos but appears as aggregates in some embryos exposed to high temperatures. Blastocysts are more resistant to high temperatures. From the research conducted, it appears that HSP70 is induced as a mechanism of cell protection against an environment that is unfavorable for cell viability in oocytes and embryos exposed to high temperatures. The role of group with HSP in protecting organisms from environmental stress is illustrated by the increased synthesis of this protein. Matwee (2001) demonstrated that HSPs play an important role in the fertilization and early development of mammalian embryos by demonstrating that the presence of anti-HSP70 antibodies significantly reduced the strong binding of spermatozoa to the zona pellucida of bovine oocytes and interrupted the achievement of meiosis II and pronuclear formation. The presence of anti-HSP70 in the culture medium on days 3–9 will increase the incidence of apoptosis and significantly reduce the number of embryos reaching the blastocyst stage. Thus, the reduction of embryo development is a response to HSP70 inhibition related to increased apoptosis because of temperature stress. The inhibitory effect of anti-HSP70 on sperm-oocyte interaction can occur at all stages of the fertilization process, from the initiation of sperm fusion with the oolemma. The mechanism of antibody inhibition against HSP70 has not been explained precisely, but it is suspected that anti-HSP70 receptors are present in the zona pellucida and plasma membrane of the embryo. Immunolabeling of the presence of anti-HSP70 shows that antibodies can penetrate the zona pellucida and the membrane of living embryo cells, thereby providing intracellular effects. Meanwhile, Child et al. (2006) conducted a study to prove that in several cases of birth defects in humans, there was an increase in anti-HSP70 in blood collected at 16 weeks of gestation. Embryonic organogenesis occurs in the first trimester of pregnancy. A cell can provide protection through repair mechanisms and apoptosis. Vitrified oocytes that cannot undergo repair will experience apoptosis or experience failure of fertilization and growth and development processes, whereas those that are able to survive will continue to grow and develop. According to Li et al. (2000), HSPs can also control the apoptosis mechanism that occurs in these cells. Unlike embryos, mature oocytes exposed to temperature stress conditions do not induce HSP70 synthesis. In oocytes, HSP70 is synthesized during germinal vesicle (GV) growth and maturation to metaphase II (Le Masson and Christians, 2011). Lánská et al. (2006) stated that during the growth period, oocytes with a size of 80–99 μm synthesize HSP70 even though it is not triggered by temperature stress conditions but has the ability to increase HSP70 synthesis when experiencing heat stress for 1 h. Likewise, during the oocyte growth period with a size of 100–115 μm, the oocyte continues to synthesize HSP70, but no longer synthesizes HSP70 when experiencing heat stress. Neuer (2000) also added that at the end of the growth period, in mature oocytes, HSP70 can be found in large amounts, but the content of HSP70 does not increase when temperature stress occurs. The ability to synthesize HSP70 decreases at the end of oocyte growth, and oocytes no longer induce HSP70 synthesis when temperature stress occurs. During oocyte growth, HSP70 is synthesized and stored. Lánská et al. (2006) concluded that HSP70 plays an important role in oocyte growth and maturation. According to Miętkiewska et al. (2022) HSP70, which is synthesized in the early stages of oocyte development, namely at the GV stage, is then accumulated and stored at the oocyte maturation stage, needed to protect oocytes from environmental influences that occur in the ovaries, and HSP70 is thought to be provided for the activation of zygote genes and the protective effect of inhibiting apoptosis. Figure 4 explains the HSP70 protection mechanism in vitrified oocytes.
Fig. 4. HSP70 protection mechanism in vitrified oocytes through HSP70 cell signaling by blocking cytochrome c and caspase 3 to inhibit apoptosis Figure 4 shows the molecular regulatory pathway of HSP70 in maintaining cell viability through inhibition of vitrification-induced apoptosis, especially in sensitive cells such as oocytes or embryos. During stress processes, such as vitrification, activation of heat shock factor 1 (HSF-1) occurs through phosphorylation by protein kinases, which causes trimerization of HSF-1 and its translocation to the nucleus to induce transcription of HSP70 mRNA (Jin et al., 2018). The newly synthesized HSP70 protein then enters the cytoplasm and functions as a molecular chaperone (Mayer and Bukau, 2005). HSP70 plays a crucial role in inhibiting the release of cytochrome C from mitochondria, thereby preventing the activation of the intrinsic apoptosis pathway, thereby inhibiting the formation of the apoptosome complex (APAF-1–Caspase 9) and the activation of Caspase 3, thereby blocking the apoptosis cascade, supporting DNA repair, and inhibiting cell death signals, which overall increases cell viability (Beere et al., 2000). However, if HSP70 is not available or is not successfully internalized, vitrification stress can lead to cytochrome C release, caspase activation, and ultimately apoptosis, which is characterized by decreased cell viability (Mosser et al., 2000). Figure 4 also shows that the HSP response (HSP-R) is insufficient to prevent cell damage under vitrification conditions without HSP70, resulting in increased apoptosis. In contrast, vitrification in the presence of HSP70 produces an effective protective response and increases post-vitrification cell survival. The mechanism of oocyte protection during the vitrification process played by HSP70 is also a new finding in this study. HSP70 supplemented into cryoprotectants can interact with oocytes through the presence of HSP70 receptors (HSP70-R) in the zona pellucida and cause an intracellular reaction in the form of cell signaling (Spinaci et al., 2005). Extreme temperature changes as stressors during the vitrification process will cause HSF-1 in the cytosol to bind to groups with HSP that were originally inactive to become active. HSF-1 is separated from HSP70 (Stamperna et al., 2021). Protein kinase phosphorylates HSF-1 and trimerizes it in the cytosol. This HSF-1 trimer complex enters the cell nucleus and binds to the heat shock element (HSE) in the promoter region of the HSP70 gene (Prince et al., 2020). The HSP70 mRNA undergoes transcription and enters the cytoplasm to be synthesized and translated into new HSP70 (Horrell et al., 1987). This cell protection mechanism includes the function of HSPs as chaperone molecules that help the translocation process of proteins synthesized in cells and help the repair and refolding process of damaged proteins, as well as inhibit apoptosis. HSP70 shows a cell protection mechanism by preventing the activation of proapoptotic caspases (Ran et al., 2004). Nevertheless, several limitations should be acknowledged regarding HSP70 supplementation in vitrification protocols. Excessive exogenous HSP70 may disrupt endogenous chaperone networks, alter protein homeostasis, or even trigger unintended immunogenic responses, thereby compromising developmental competence. In addition, species-specific differences in membrane composition, receptor distribution, and intracellular signaling pathways may limit the generalizability of results obtained from model organisms. The lack of standardized dosing regimens further complicates reproducibility and hinders translation into clinical or field applications. Therefore, while HSP70 supplementation offers promising cytoprotective effects, its application should be carefully optimized and validated through comprehensive long-term safety assessments. ConclusionHSP70 plays a central role in protecting oocytes from vitrification-induced cellular stress. Numerous experimental evidence suggests that HSP70 functions as a molecular chaperone, not only preventing protein denaturation but also inhibiting the apoptotic pathway through mitochondrial stabilization, cytochrome c release suppression, apoptosome formation inhibition, and caspase-3 activation. Adequate HSP70 during vitrification can significantly increase oocyte viability and reduce the extent of structural and functional damage. However, recent studies have also highlighted that the protective effects of HSP70 are highly dependent on the biological context and expression level of HSP70. Studies in various species indicate that the optimal levels of HSP70 supplementation are not fully agreed upon, and excessive expression can disrupt important physiological processes, such as oocyte maturation, cell division, and subsequent embryonic development. Therefore, balancing the protective benefits and potential biological disruptions of HSP70 is crucial. Further research is needed to determine the dose-response curve, the most effective timing of supplementation, and differences in sensitivity between species and stages of oocyte development in the context of clinical applications or reproductive technology. AcknowledgmentsThe authors would like to thank the Faculty of Veterinary Medicine, Universitas Airlangga. The author also thanks Win Darmanto, Aucky Hinting, and Aulanni’am Aulanni’am for their expertise. Author’s contributionsRR, SPM, ARK, and WW: conceived the idea and drafted the manuscript. PS, SRA, IM, and AOA: acquired, analyzed, and interpreted the data. BPP, FAR, SW, BWKW, and ATK: critically read and revised the manuscript for intellectual content. All authors have read and approved the final version of the manuscript. All authors have read, reviewed, and approved the final version of the manuscript. Conflict of interestThe authors declare no conflict of interest. FundingThis study was funded by the authors. Data availabilityAll data are available in the revised manuscript. ReferencesAlmanza, A., Carlesso, A., Chintha, C., Creedican, S., Doultsinos, D., Leuzzi, B., Luís, A., McCarthy, N., Montibeller, L., More, S., Papaioannou, A., Püschel, F., Sassano, M.L., Skoko, J., Agostinis, P., De Belleroche, J., Eriksson, L.A., Fulda, S., Gorman, A.M., Healy, S., Kozlov, A., Muñoz-Pinedo, C., Rehm, M., Chevet, E. and Samali, A. 2019. Endoplasmic reticulum stress signalling - from basic mechanisms to clinical applications. FEBS. J. 286(2), 241–278. Amini, J.D. and Benson. 2023. Technologies for vitrification based cryopreservation. Bioengineering (Basel). 10(5), 508. Araújo-Lemos, P.F., Neto, L.M.F., Moura, M.T., Melo, J.V., Lima, P.F. and Oliveira, M.A. 2015. Comparison of vitrification and conventional freezing for cryopreservation of caprine embryos. Zygote 23(4), 594–602. Arguello, O.A.D. and Haisma, H.J. 2021. Apoptosis-Inducing TNF Superfamily Ligands for Cancer Therapy. Cancers (Basel). 13(7), 1543. Argyle, C.E., Harper, J.C. and Davies, M.C. 2016. Oocyte cryopreservation: where are we now?. Hum. Reprod. Update 22(4), 440–449. Bankowski, B.J., Lyerly, A.D., Faden, R.R. and Wallach, E.E. 2005. The social implications of embryo cryopreservation. Fertil. Steril. 84(4), 823–832. Baust, J.G., Gao, D. and Baust, J.M. 2009. Cryopreservation: an emerging paradigm change. Organogenesis 5(3), 90–96. Beere, H.M., Wolf, B.B., Cain, K., Mosser, D.D., Mahboubi, A., Kuwana, T., Tailor, P., Morimoto, R.I., Cohen, G.M. and Green, D.R. 2000. Heat-shock protein 70 inhibits apoptosis by preventing recruitment of procaspase-9 to the Apaf-1 apoptosome. Nat. Cell Biol. 2(8), 469–475. Bhat, G.R. and Sofi, K.A. 2021. Oocyte and embryo preservation in wild animals: an update. CryoLetters 42(5), 251–260. Bissoyi, A., Tomás, R.M.F., Gao, Y., Guo, Q. and Gibson, M.I. 2023. Cryopreservation of Liver-Cell Spheroids with Macromolecular Cryoprotectants. ACS. Appl. Mater. Inter. 15(2), 2630–2638. Bojic, S., Murray, A., Bentley, B.L., Spindler, R., Pawlik, P., Cordeiro, J.L., Bauer, R. and De Magalhães, J.P. 2021. Winter is coming: the future of cryopreservation. BMC Biol. 19(1), 56. Boopathy, L.R.A., Jacob-Tomas, S., Alecki, C. and Vera, M. 2022. Mechanisms tailoring the expression of heat shock proteins to proteostasis challenges. J. Biol. Chem. 298(5), 101796. Braakman, I. and Hebert, D.N. 2013. Protein folding in the endoplasmic reticulum. Cold Spring Harb. Perspect. Biol. 5(5), 13201. Bratton, S.B. and Salvesen, G.S. 2010. Regulation of the Apaf-1-caspase-9 apoptosome. J. Cell Sci. 123(Pt 19), 3209–3214. Chang, C.C., Shapiro, D.B. and Nagy, Z.P. 2022. The effects of vitrification on oocyte quality. Biol. Reprod. 106(2), 316–327. Chang, T. and Zhao, G. 2021. Ice Inhibition for Cryopreservation: materials, Strategies, and Challenges. Adv. Sci. (Weinh). 8(6), 2002425. Chatterjee, N. and Walker, G.C. 2017. Mechanisms of DNA damage, repair, and mutagenesis. Environ. Mol. Mutagen. 58(5), 235–263. Chen, H., Zhang, L., Meng, L., Liang, L. and Zhang, C. 2022. Advantages of vitrification preservation in assisted reproduction and potential influences on imprinted genes. Clin. Epigenetics 14(1), 141. Chen, S. and Brown, I.R. 2007. Translocation of constitutively expressed heat shock protein Hsc70 to synapse-enriched areas of the cerebral cortex after hyperthermic stress. J. Neurosci. Res. 85(2), 402–409. Chi, H.J. 2002. Cryopreservation of human embryos using ethylene glycol in controlled slow freezing. Hum. Reprod. 17(8), 2146–2151. Child, D.F., Hudson, P.R., Hunter-Lavin, C., Mukhergee, S., China, S., Williams, C.P. and Williams, J.H. 2006. Birth defects and anti-heat shock protein 70 antibodies in early pregnancy. Cell Stress Chaperones 11(1), 101–105. Chronopoulou, E., Raperport, C., Sfakianakis, A., Srivastava, G. and Homburg, R. 2021. Elective oocyte cryopreservation for age-related fertility decline. J. Assist. Reprod. Genet. 38(5), 1177–1186. Comizzoli, P., Wildt, D.E. and Pukazhenthi, B.S. 2008. Impact of anisosmotic conditions on structural and functional integrity of cumulus-oocyte complexes at the germinal vesicle stage in the domestic cat. Mol. Reprod. Dev. 75(2), 345–354. Costa-Borges, N., Matia-Algué, Q., Coello, A., Mestres, E., Acacio, M., Flores-saiffe Farias, A., Castello, C., Gallardo, M., Chavez-Badiola, A., Marco-Jiménez, F., Cobo, A. and Cohen, J. 2025. Preclinical validation of fast oocyte vitrification and warming protocols with comparable efficiencies to a standard method. Hum. Reprod. 40(6), 1066–1076. Cottle, C., Porter, A.P., Lipat, A., Turner-Lyles, C., Nguyen, J., Moll, G. and Chinnadurai, R. 2022. Impact of cryopreservation and freeze-thawing on therapeutic properties of mesenchymal stromal/stem cells and other common cellular therapeutics. Curr. Stem Cell Rep. 8(1), 72–92. Das, M. and Son, W.Y. 2023. In vitro maturation (IVM) of human immature oocytes: is it still relevant?. Reprod. Biol. Endocrinol. 21(1), 110. De Leon, P.M., Campos, V.F., Corcini, C.D., Santos, E.C., Rambo, G., Lucia, T., Deschamps, J.C. and Collares, T. 2012. Cryopreservation of immature equine oocytes, comparing a solid surface vitrification process with open pulled straws and the use of a synthetic ice blocker. Theriogenology 77(1), 21–27. De Maio, A. and Hightower, L.E. 2021. Heat shock proteins and the biogenesis of cellular membranes. Cell Stress Chaperones 26(1), 15–18. Denton, D. and Kumar, S. 2015. Terminal Deoxynucleotidyl Transferase (TdT)-Mediated dUTP Nick-End Labeling (TUNEL) for Detection of Apoptotic Cells in Drosophila. Cold Spring Harb. Protoc. 6(6), 568–571. Dhali, A., Manik, R.S., Das, S.K., Singla, S.K. and Palta, P. 2000. Vitrification of buffalo (Bubalus bubalis) oocytes. Theriogenology 53(6), 1295–1303. Díez, C., Muñoz, M., Caamaño, J.N. and Gómez, E. 2012. Cryopreservation of the bovine oocyte: current status and perspectives. Reprod. Domest. Anim. 47(Suppl 3), 76–83. Doyle, J.O., Richter, K.S., Lim, J., Stillman, R.J., Graham, J.R. and Tucker, M.J. 2016. Successful elective and medically indicated oocyte vitrification and warming for autologous in vitro fertilization, with predicted birth probabilities for fertility preservation according to number of cryopreserved oocytes and age at retrieval. Fertil. Steril. 105(2), 459–466. Du, M., Li, X., Bayinnamula., Wang, N., Liu, Y., Zhang, L., Zhao, Y. and Dugarjaviin, M. 2024. Optimization of vitrification methods for equine oocytes. Tissue Cell 91(1), 102632. Duncan, F.E., Chiang, T., Schultz, R.M. and Lampson, M.A. 2009. Evidence that a defective spindle assembly checkpoint is not the primary cause of maternal age-associated aneuploidy in mouse eggs. Biol. Reprod. 81(4), 768–776. Elmore, S. 2007. Apoptosis: a review of programmed cell death. Toxicol. Pathol. 35(4), 495–516. Elnahas, A., Alcolak, E., Marar, E.A., Elnahas, T., Elnahas, K., Palapelas, V., Diedrich, K. and Al-Hasani, S. 2010. Vitrification of human oocytes and different development stages of embryos: an overview. Middle East Fertility Soc. J. 15(1), 2–9. Estudillo, E., Jiménez, A., Bustamante-Nieves, P.E., Palacios-Reyes, C., Velasco, I. and López-Ornelas, A. 2021. Cryopreservation of gametes and embryos and their molecular changes. Int. J. Mol. Sci. 22(19), 10864. Feng, Y., Zhang, M., Yuan, W., Zhao, D., Luo, Z., Tang, Z., Wang, Y. and Cang, M. 2025. Effects and mechanisms of imperatorin on vitrified mouse oocytes. Animals 15(5), 661. García-Martínez, T., Martínez-Rodero, I., Roncero-Carol, J., Yánez-Ortiz, I., Higgins, A.Z. and Mogas, T. 2022. Impact of equilibration duration combined with temperature on the outcome of bovine oocyte vitrification. Theriogenology 184(1), 110–123. García-Martínez, T., Mogas, T., Mullen, S.F., Martínez-Rodero, I., Gulieva, R.E. and Higgins, A.Z. 2021. Effect of cryoprotectant concentration on bovine oocyte permeability and comparison of two membrane permeability modelling approaches. Sci. Rep. 11(1), 15387. Gerasimavicius, L., Livesey, B.J. and Marsh, J.A. 2022. Loss-of-function, gain-of-function and dominant-negative mutations have profoundly different effects on protein structure. Nat. Commun. 13(1), 3895. Ghafari, F., Gutierrez, C.G. and Hartshorne, G.M. 2007. Apoptosis in mouse fetal and neonatal oocytes during meiotic prophase one. BMC Develop. Biol. 7(1), 87. Giffard, R.G., Han, R.Q., Emery, J.F., Duan, M. and Pittet, J.F. 2008. Regulation of apoptotic and inflammatory cell signaling in cerebral ischemia: the complex roles of heat shock protein 70. Anesthesiology 109(2), 339–348. Gil, L., Olaciregui, M., Luño, V., Malo, C., González, N. and Martínez, F. 2014. Current status of freeze-drying technology to preserve domestic animals sperm. Reprod. Domest. Anim. 49(Suppl 4), 72–81. Gomes, C.M., Silva, C.A., Acevedo, N., Baracat, E., Serafini, P. and Smith, G.D. 2008. Influence of vitrification on mouse metaphase II oocyte spindle dynamics and chromatin alignment. Fertil. Steril. 90(4 Suppl), 1396–1404. Gomez, M.A.R. and Ibba, M. 2020. Aminoacyl-tRNA synthetases. RNA 26(8), 910–936. Gomez-Pastor, R., Burchfiel, E.T. and Thiele, D.J. 2018. Regulation of heat shock transcription factors and their roles in physiology and disease. Nat. Rev. Mol. Cell Biol. 19(1), 4–19. Gutierrez-Castillo, E., Diaz, F.A., Talbot, S.A. and Bondioli, K.R. 2022. Recovery of spindle morphology and mitochondrial function through extended culture after vitrification-warming of bovine oocytes. Theriogenology 189(1), 192–198. Hai, E., Li, B., Zhang, J. and Zhang, J. 2024. Sperm freezing damage: the role of regulated cell death. Cell Death Discovery 10(1), 239. Hashikawa, N., Ido, M., Morita, Y. and Hashikawa-Hobara, N. 2021. Effects from the induction of heat shock proteins in a murine model due to progression of aortic atherosclerosis. Sci. Rep. 11(1), 7025. Helm, M. 2006. Post-transcriptional nucleotide modification and alternative folding of RNA. Nucleic Acids Res. 34(2), 721–733. Hochi, S., Fujimoto, T., Choi, Y.H., Braun, J. and Oguri, N. 1994. Cryopreservation of equine oocytes by 2-step freezing. Theriogenology 42(7), 1085–1094. Horrell, A., Shuttleworth, J. and Colman, A. 1987. Transcript levels and translational control of hsp70 synthesis in Xenopus oocytes. Genes Dev. 1(5), 433–444. Hu, C., Yang, J., Qi, Z., Wu, H., Wang, B., Zou, F., Mei, H., Liu, J., Wang, W. and Liu, Q. 2022. Heat shock proteins: biological functions, pathological roles, and therapeutic opportunities. MedComm. (2020). 3(3), e161. Huang, J., Li, Q., Zhao, R., Li, W., Han, Z., Chen, X., Xiao, B., Wu, S., Jiang, Z., Hu, J. and Liu, L. 2008. Effect of sugars on maturation rate of vitrified-thawed immature porcine oocytes. Anim. Reprod. Sci. 106(1–2), 25–35. Huang, Z., Gao, L., Hou, Y., Zhu, S. and Fu, X. 2018. Cryopreservation of farm animal gametes and embryos: recent updates and progress. Front. Agr. Sci. Eng. 6(1), 42–53. Isachenko, E. 2004. DNA integrity and motility of human spermatozoa after standard slow freezing versus cryoprotectant-free vitrification. Hum. Reprod. 19(4), 932–939. Jäättelä, M., Wissing, D., Kokholm, K., Kallunki, T. and Egeblad, M. 1998. Hsp70 exerts its anti-apoptotic function downstream of caspase-3-like proteases. EMBO. J. 17(21), 6124–6134. Jaiswal, A.N. and Vagga, A. 2022. Cryopreservation: a review article. Cureus 14(11), e31564. Jan, R. and Chaudhry, G.E. 2019. Understanding apoptosis and apoptotic pathways targeted cancer therapeutics. Adv. Pharm. Bull. 9(2), 205–218. Jin, X., Qiao, A., Moskophidis, D. and Mivechi, N.F. 2018. Modulation of Heat Shock Factor 1 Activity through Silencing of Ser303/Ser307 Phosphorylation Supports a Metabolic Program Leading to Age-Related Obesity and Insulin Resistance. Mol. Cell. Biol. 38(18), e00095. Jung, S.E. and Ryu, B.Y. 2023. New strategies for germ cell cryopreservation: cryoinjury modulation. Clin. Exp. Reprod. Med. 50(4), 213–222. Kalmar, B. and Greensmith, L. 2009. Induction of heat shock proteins for protection against oxidative stress. Adv. Drug Deliv. Rev. 61(4), 310–318. Kalogeris, T., Baines, C.P., Krenz, M. and Korthuis, R.J. 2012. Cell biology of ischemia/reperfusion injury. Int. Rev. Cell Mol. Biol. 298(1), 229–317. Kasai, M. and Mukaida, T. 2004. Cryopreservation of animal and human embryos by vitrification. Reprod. Biomed. Online 9(2), 164–170. Kmiecik, S.W., Le Breton, L. and Mayer, M.P. 2020. Feedback regulation of heat shock factor 1 (Hsf1) activity by Hsp70-mediated trimer unzipping and dissociation from DNA. EMBO J. 39(14), e104096. Konc, J., Kanyó, K., Kriston, R., Somoskői, B. and Cseh, S. 2014. Cryopreservation of embryos and oocytes in human assisted reproduction. Biomed. Res. Int. 1(1), 307268. Konc, J., Kanyo, K., Varga, E., Kriston, R. and Cseh, S. 2008. Births resulting from oocyte cryopreservation using a slow freezing protocol with propanediol and sucrose. Syst. Biol. Reprod. Med. 54(4–5), 205–210. Kyogoku, H. and Kitajima, T.S. 2023. The large cytoplasmic volume of oocyte. J. Reprod. Dev. 69(1), 1–9. Lang, L., Miskovic, D., Lo, M. and Heikkila, J.J. 2000. Stress-induced, tissue-specific enrichment of hsp70 mRNA accumulation in Xenopus laevis embryos. Cell Stress Chaperones 5(1), 36–44. Lanneau, D., Brunet, M., Frisan, E., Solary, E., Fontenay, M. and Garrido, C. 2008. Heat shock proteins: essential proteins for apoptosis regulation. J. Cell. Mol. Med. 12(3), 743–761. Lánská, V., Chmelíková, E., Sedmíková, M., Petr, J., Rajmon, R., Ješeta, M. and Rozinek, J. 2006. Expression of heat shock protein70 in pig oocytes: heat shock response during oocyte growth. Anim. Reprod. Sci. 96(1–2), 154–164. Latchman, D.S. 2001. Heat shock proteins and cardiac protection. Cardiovasc. Res. 51(4), 637–646. Le Masson, F. and Christians, E. 2011. HSFs and regulation of Hsp70.1 (Hspa1b) in oocytes and preimplantation embryos: new insights brought by transgenic and knockout mouse models. Cell Stress Chaperones 16(3), 275–285. Ledwaba, M.R., O’Neill, H.A., Thema, M.A., Maqhashu, A. and Mphaphathi, M.L. 2025. Techniques for in vitro fertilisation of vitrified cattle oocytes: challenges and new developments. Agriculture 15(4), 363. Li, C.Y., Lee, J.S., Ko, Y.G., Kim, J.I. and Seo, J.S. 2000. Heat shock protein 70 inhibits apoptosis downstream of cytochrome c release and upstream of caspase-3 activation. J. Biol. Chem. 275(33), 25665–25671. Li, L., Bi, X., Wu, X., Chen, Z., Cao, Y. and Zhao, G. 2024. Improving vitrification efficiency of human in vitro matured oocytes by the addition of LEA proteins. Hum. Reprod. 39(6), 1275–1290. Li, P., Nijhawan, D., Budihardjo, I., Srinivasula, S.M., Ahmad, M., Alnemri, E.S. and Wang, X. 1997. Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell 91(4), 479–489. Liebermann, J. and Tucker, M.J. 2002. Effect of carrier system on the yield of human oocytes and embryos as assessed by survival and developmental potential after vitrification. Reproduction. 124(4), 483–489. Litscher, E.S., Williams, Z. and Wassarman, P.M. 2009. Zona pellucida glycoprotein ZP3 and fertilization in mammals. Mol. Reprod. Dev. 76(10), 933–941. López, A., Ducolomb, Y., Casas, E., Retana-Márquez, S., Betancourt, M. and Casillas, F. 2021. Effects of Porcine Immature Oocyte Vitrification on Actin Microfilament Distribution and Chromatin Integrity During Early Embryo Development In Vitro. Front. Cell Develop. Biol. 9(1), 636765. Małecki, J.M., Davydova, E. and Falnes, P.Ø. 2022. Protein methylation in mitochondria. J. Biol. Chem. 298(4), 101791. Martínez-Burgos, M., Herrero, L., Megías, D., Salvanes, R., Montoya, M.C., Cobo, A.C. and Garcia-Velasco, J.A. 2011. Vitrification versus slow freezing of oocytes: effects on morphologic appearance, meiotic spindle configuration, and DNA damage. Fertil. Steril. 95(1), 374–377. Masser, A.E., Kang, W., Roy, J., Mohanakrishnan Kaimal, J., Quintana-Cordero, J., Friedländer, M.R. and Andréasson, C. 2019. Cytoplasmic protein misfolding titrates Hsp70 to activate nuclear Hsf1. Elife 8(1), e47791. Matwee, C. 2001. The effects of antibodies to heat shock protein 70 in fertilization and embryo development. Mol. Hum. Reprod. 7(9), 829–837. Mayer, M.P. and Bukau, B. 2005. Hsp70 chaperones: cellular functions and molecular mechanism. Cell. Mol. Life Sci. 62(6), 670–684. McWilliams, R.B., Gibbons, W.E. and Leibo, S.P. 1995. Osmotic and physiological responses of mouse zygotes and human oocytes to mono- and disaccharides. Hum. Reprod. 10(5), 1163–1171. Meneghel, J., Kilbride, P. and Morris, G.J. 2020. Cryopreservation as a Key Element in the Successful Delivery of Cell-Based Therapies-A Review. Front. Med. (Lausanne). 7(1), 592242. Miętkiewska, K., Kordowitzki, P. and Pareek, C.S. 2022. Effects of Heat Stress on Bovine Oocytes and Early Embryonic Development-An Update. Cells 11(24), 4073. Miller, M.A. and Zachary, J.F. 2017. Mechanisms and Morphology of Cellular Injury, Adaptation, and Death. Pathologic Basis Vet. Dis. 1(1), 2–43. Moore, L.D., Le, T. and Fan, G. 2013. DNA Methylation and Its Basic Function. Neuropsychopharmacol 38(1), 23–38. Mosser, D.D., Caron, A.W., Bourget, L., Meriin, A.B., Sherman, M.Y., Morimoto, R.I. and Massie, B. 2000. The chaperone function of hsp70 is required for protection against stress-induced apoptosis. Mol. Cellular Biol. 20(19), 7146–7159. Mustafa, M., Ahmad, R., Tantry, I.Q., Ahmad, W., Siddiqui, S., Alam, M., Abbas, K., Moinuddin., Hassan, M.I., Habib, S. and Islam, S. 2024. Apoptosis: a Comprehensive Overview of Signaling Pathways, Morphological Changes, and Physiological Significance and Therapeutic Implications. Cells 13(22), 1838. Neuer, A. 2000. The role of heat shock proteins in reproduction. Hum. Reprod. Update 6(2), 149–159. Otoi, T., Yamamoto, K., Koyama, N., Tachikawa, S. and Suzuki, T. 1998. Cryopreservation of mature bovine oocytes by vitrification in straws. Cryobiology 37(1), 77–85. Pai, H.D., Baid, R., Palshetkar, N.P., Pai, A., Pai, R.D. and Palshetkar, R. 2021. Oocyte cryopreservation - current scenario and future perspectives: a narrative review. J. Hum. Reprod. Sci. 14(4), 340 -349; doi:10.4103/jhrs.jhrs_173_21 Pardede, B.P., Kusumawati, A., Pangestu, M. and Purwantara, B. 2023. Bovine sperm HSP-70 molecules: a potential cryo-tolerance marker associated with semen quality and fertility rate. Front. Vet. Sci. 10(1), 1167594. Park, H.S., Lee, J.S., Huh, S.H., Seo, J.S. and Choi, E.J. 2001. Hsp72 functions as a natural inhibitory protein of c-Jun N-terminal kinase. EMBO J. 20(3), 446–456. Park, M.J., Lee, S.E., Yoon, J.W., Park, H.J., Kim, S.H., Oh, S.H., Lee, D.G., Pyeon, D.B., Kim, E.Y. and Park, S.P. 2022. Effect of Supplementation of Cryoprotectant Solution with Hydroxypropyl Cellulose for Vitrification of Bovine Oocytes. Animals 12(19), 2636. Pearson, W.R. 2013. An introduction to sequence similarity ('homology') searching. Curr. Protoc. Bioinf. 3(1), 3.1.1–3.1.8. Pegg. 2015. Principles of cryopreservation. Methods Mol. Biol. 1257(1), 3–19. Pezo, F., Zambrano, F., Uribe, P., De Andrade, A.F.C. and Sánchez, R. 2023. Slow Freezing of Preserved Boar Sperm: comparison of Conventional and Automated Techniques on Post-Thaw Functional Quality by a New Combination of Sperm Function Tests. Animals 13(18), 2826. Pirkkala, L., Nykänen, P. and Sistonen, L. 2001. Roles of the heat shock transcription factors in regulation of the heat shock response and beyond. FASEB. J. 15(7), 1118–1131. Prentice-Biensch, J.R., Singh, J., Mapletoft, R.J. and Anzar, M. 2012. Vitrification of immature bovine cumulus-oocyte complexes: effects of cryoprotectants, the vitrification procedure and warming time on cleavage and embryo development. Reprod. Biol. Endocrinol. 10(1), 73. Prince, T.L., Lang, B.J., Guerrero-Gimenez, M.E., Fernandez-Muñoz, J.M., Ackerman, A. and Calderwood, S.K. 2020. HSF1: primary Factor in Molecular Chaperone Expression and a Major Contributor to Cancer Morbidity. Cells 9(4), 1046. Prokhorova, E.A., Zamaraev, A.V., Kopeina, G.S., Zhivotovsky, B. and Lavrik, I.N. 2015. Role of the nucleus in apoptosis: signaling and execution. Cell. Mol. Life Sci. 72(23), 4593–4612. Ran, R., Lu, A., Zhang, L., Tang, Y., Zhu, H., Xu, H., Feng, Y., Han, C., Zhou, G., Rigby, A.C. and Sharp, F.R. 2004. Hsp70 promotes TNF-mediated apoptosis by binding IKK gamma and impairing NF-kappa B survival signaling. Genes Dev. 18(12), 1466–1481. Redza-Dutordoir, M. and Averill-Bates, D.A. 2016. Activation of apoptosis signalling pathways by reactive oxygen species. Biochimica Et Biophysica Acta (BBA). -. Mol. Cell. Res. 1863(12), 2977–2992. Rienzi, L., Gracia, C., Maggiulli, R., Labarbera, A.R., Kaser, D.J., Ubaldi, F.M., Vanderpoel, S. and Racowsky, C. 2017. Oocyte, embryo and blastocyst cryopreservation in ART: systematic review and meta-analysis comparing slow-freezing versus vitrification to produce evidence for the development of global guidance. Hum. Reprod. Update 23(2), 139–155. Rienzi, L., Romano, S., Albricci, L., Maggiulli, R., Capalbo, A., Baroni, E., Colamaria, S., Sapienza, F. and Ubaldi, F. 2010. Embryo development of fresh 'versus' vitrified metaphase II oocytes after ICSI: a prospective randomized sibling-oocyte study. Hum. Reprod. 25(1), 66–73. Rodriguez-Villamil, P., Ongaratto, F.L., Moreira, G., Taranco, M.F. and Bó, G.A. 2016. Vitrification of immature and matured bovine oocytes: effect of brilliant cresyl blue selection and hyaluronan addition. Anim. Reprod. 13(1), 42–49. Roque-Borda, C.A., Kulus, D., Vacaro De Souza, A., Kaviani, B. and Vicente, E.F. 2021. Cryopreservation of Agronomic Plant Germplasm Using Vitrification-Based Methods: an Overview of Selected Case Studies. Int. J. Mol. Sci. 22(11), 6157. Roth, Z. and Hansen, P.J. 2004. Involvement of apoptosis in disruption of developmental competence of bovine oocytes by heat shock during maturation. Biol. Reprod. 71(6), 1898–1906. Saibil, H. 2013. Chaperone machines for protein folding, unfolding and disaggregation. Nat. Rev. Mol. Cell Biol. 14(10), 630–642. Sanchez-Osorio, J., Cuello, C., Gil, M.A., Parrilla, I., Maside, C., Almiñana, C., Lucas, X., Roca, J., Vazquez, J.M. and Martinez, E.A. 2010. Vitrification and warming of in vivo-derived porcine embryos in a chemically defined medium. Theriogenology 73(3), 300–308. Schulze-Osthoff, K., Walczak, H., Dröge, W. and Krammer, P.H. 1994. Cell nucleus and DNA fragmentation are not required for apoptosis. J. Cell Biol. 127(1), 15–20. Scieglinska, D., Krawczyk, Z., Sojka, D.R. and Gogler-Pigłowska, A. 2019. Heat shock proteins in the physiology and pathophysiology of epidermal keratinocytes. Cell Stress Chaperones 24(6), 1027–1044. Shin, Y.O., Oh, J.K., Bae, J.S., Lee, M.Y., Lee, J.B., Yang, H.M., Min, Y.K. and Matsumoto, T. 2004. Progressive strenuous exercise induces the expression of HSP70 in rat skeletal muscles and myocardium. J. Therm. Biol. 29(7–8), 765–768. Singh, M.K., Han, S., Ju, S., Ranbhise, J.S., Ha, J., Yeo, S.G., Kim, S.S. and Kang, I. 2025. Hsp70: a Multifunctional Chaperone in Maintaining Proteostasis and Its Implications in Human Disease. Cells 14(7), 509. Singh, M.K., Shin, Y., Ju, S., Han, S., Choe, W., Yoon, K.S., Kim, S.S. and Kang, I. 2024. Heat Shock Response and Heat Shock Proteins: current Understanding and Future Opportunities in Human Diseases. Int. J. Mol. Sci. 25(8), 4209. Sonna, L.A., Fujita, J., Gaffin, S.L. and Lilly, C.M. 2002. Invited review: effects of heat and cold stress on mammalian gene expression. J. Appl. Physiol. (1985). 92(4), 1725–1742. Souza-Cácares, M.B., Fialho, A.L.L., Silva, W.A.L., Cardoso, C.J.T., Pöhland, R., Martins, M.I.M. and Melo-Sterza, F.A. 2019. Oocyte quality and heat shock proteins in oocytes from bovine breeds adapted to the tropics under different conditions of environmental thermal stress. Theriogenology 130(1), 103–110. Spinaci, M., Volpe, S., Bernardini, C., De Ambrogi, M., Tamanini, C., Seren, E. and Galeati, G. 2005. Immunolocalization of heat shock protein 70 (Hsp 70) in boar spermatozoa and its role during fertilization. Mol. Reprod. Dev. 72(4), 534–541. Stamperna, K., Giannoulis, T., Dovolou, E., Kalemkeridou, M., Nanas, I., Dadouli, K., Moutou, K., Mamuris, Z. and Amiridis, G.S. 2021. Heat Shock Protein 70 Improves In Vitro Embryo Yield and Quality from Heat Stressed Bovine Oocytes. Animals (Basel). 11(6), 1794. Stankiewicz, A.R., Lachapelle, G., Foo, C.P.Z., Radicioni, S.M. and Mosser, D.D. 2005. Hsp70 inhibits heat-induced apoptosis upstream of mitochondria by preventing Bax translocation. J. Biol. Chem. 280(46), 38729–38739. Szabo, A., Langer, T., Schröder, H., Flanagan, J., Bukau, B. and Hartl, F.U. 1994. The ATP hydrolysis-dependent reaction cycle of the Escherichia coli Hsp70 system DnaK, DnaJ, and GrpE. Proc. Natl. Acad. Sci. U. S. A. 91(22), 10345–10349. Szyller, J. and Bil-Lula, I. 2021. Heat shock proteins in oxidative stress and ischemia/reperfusion injury and benefits from physical exercises: a review to the current knowledge. Oxid. Med. Cell. Longev. 2021(1), 6678457. Tamura. and K. 2015. Origins and Early Evolution of the tRNA Molecule. Life (Basel). 5(4), 1687–1699. Tharasanit, T. and Thuwanut, P. 2021. Oocyte Cryopreservation in Domestic Animals and Humans: principles, Techniques and Updated Outcomes. Animals (Basel). 11(10), 2949. Tomanek, L. 2010. Variation in the heat shock response and its implication for predicting the effect of global climate change on species' biogeographical distribution ranges and metabolic costs. J. Exp. Biol. 213(6), 971–979. Tytell, M. and Hooper, P.L. 2001. Heat shock proteins: new keys to the development of cytoprotective therapies. Expert Opin. Therap. Targets 5(2), 267 -287. doi:10.1517/14728222.5.2.267 Vajta, G., Holm, P., Kuwayama, M., Booth, P.J., Jacobsen, H., Greve, T. and Callesen, H. 1998. Open Pulled Straw (OPS) vitrification: a new way to reduce cryoinjuries of bovine ova and embryos. Mol. Reprod. Dev. 51(1), 53–58. Valcarce, D.G., Cartón-García, F., Herráez, M.P. and Robles, V. 2013. Effect of cryopreservation on human sperm messenger RNAs crucial for fertilization and early embryo development. Cryobiology 67(1), 84–90. Vendrell-Flotats, M., Arcarons, N., Barau, E., López-Béjar, M. and Mogas, T. 2017. Effect of heat stress during in vitro maturation on developmental competence of vitrified bovine oocytes. Reprod. Domest. Anim. 52(Suppl 4), 48–51. Verma, P., Pfister, J.A., Mallick, S. and D'Mello, S.R. 2014. HSF1 protects neurons through a novel trimerization- and HSP-independent mechanism. J. Neurosci. 34(5), 1599–1612. Vihinen, M. 2021. Functional effects of protein variants. Biochimie 180(1), 104–120. Volloch, V., Gabai, V.L., Rits, S., Force, T. and Sherman, M.Y. 2000. HSP72 can protect cells from heat-induced apoptosis by accelerating the inactivation of stress kinase JNK. Cell Stress Chaperones 5(2), 139–147. Walker, B.N. and Biase, F.H. 2020. The blueprint of RNA storages relative to oocyte developmental competence in cattle (Bos taurus). Biol. Reprod. 102(4), 784–794. Wang, L., Schumann, U., Liu, Y., Prokopchuk, O. and Steinacker, J.M. 2012. Heat shock protein 70 (Hsp70) inhibits oxidative phosphorylation and compensates ATP balance through enhanced glycolytic activity. J. Appl. Physiol. 113(11), 1669–1676. Wang, M., Todorov, P., Wang, W., Isachenko, E., Rahimi, G., Mallmann, P. and Isachenko, V. 2022. Cryoprotectants-Free Vitrification and Conventional Freezing of Human Spermatozoa: a Comparative Transcript Profiling. Int. J. Mol. Sci. 23(6), 3047. Waudby, C.A., Dobson, C.M. and Christodoulou, J. 2019. Nature and Regulation of Protein Folding on the Ribosome. Trends Biochem. Sci. 44(11), 914–926. Wen, H., Deng, H., Li, B., Chen, J., Zhu, J., Zhang, X., Yoshida, S. and Zhou, Y. 2025. Mitochondrial diseases: from molecular mechanisms to therapeutic advances. Signal Transduct. Target. Ther. 10(1), 9. Wu, C.C. and Bratton, S.B. 2013. Regulation of the intrinsic apoptosis pathway by reactive oxygen species. Antioxid. Redox Signal. 19(6), 546–558. Wu, Z., Zhou, R., Li, B., Cao, M., Wang, W. and Li, X. 2024. Methylation modifications in tRNA and associated disorders: current research and potential therapeutic targets. Cell Prolif. 57(9), e13692. Xu, L., Gao, J., Guo, L. and Yu, H. 2020. Heat shock protein 70 (HmHsp70) from Hypsizygus marmoreus confers thermotolerance to tobacco. AMB. Expr. 10(1), 12. Xu, L., Xiong, X., Ouyang, Y., Barreto, G. and Giffard, R. 2011. Heat shock protein 72 (Hsp72) improves long term recovery after focal cerebral ischemia in mice. Neurosci. Lett. 488(3), 279–282. Xu, X., Hao, T., Komba, E., Yang, B., Hao, H., Du, W., Zhu, H., Zhang, H. and Zhao, X. 2022. Improvement of Fertilization Capacity and Developmental Ability of Vitrified Bovine Oocytes by JUNO mRNA Microinjection and Cholesterol-Loaded Methyl-β-Cyclodextrin Treatment. Int. J. Mol. Sci. 24(1), 590. Yacoub, A.N.A., Gauly, M. and Holtz, W. 2010. Open pulled straw vitrification of goat embryos at various stages of development. Theriogenology 73(8), 1018–1023. Yamanaka, K.I., Aono, N., Yoshida, H. and Sato, E. 2007. Cryopreservation and in vitro maturation of germinal vesicle stage oocytes of animals for application in assisted reproductive technology. Reprod. Med. Biol. 6(2), 61–68. Yang, Y., Jiang, G., Zhang, P. and Fan, J. 2015. Programmed cell death and its role in inflammation. Mil. Med. Res. 2(1), 12. Yapryntseva, M.A., Zhivotovsky, B. and Gogvadze, V. 2024. Permeabilization of the outer mitochondrial membrane: mechanisms and consequences. Biochimica. Et. Biophysica. Acta. (BBA). -. Mol. Basis. Dis. 1870(7), 167317. Yassin, H.B., Abu-Elnaga, N.A., Farrag, B. and El-Bahrawyb, K.A. 2022. Effect of cryoprotectants on camel oocytes vitrification. Al-Azhar Bull. Sci. Sect. C 33(1), 8. Zhou, C.J., Wang, D.H., Niu, X.X., Kong, X.W., Li, Y.J., Ren, J., Zhou, H.X., Lu, A., Zhao, Y.F. and Liang, C.G. 2016. High survival of mouse oocytes using an optimized vitrification protocol. Sci. Rep. 6(1), 19465. Zhou, M., Li, Y., Hu, Q., Bai, X.C., Huang, W., Yan, C., Scheres, S.H. and Shi, Y. 2015. Atomic structure of the apoptosome: mechanism of cytochrome c- and dATP-mediated activation of Apaf-1. Genes Dev. 29(22), 2349–2361. Zhu, S., Yoshizawa, M. and Muramatsu, S. 1998. Analysis of Fertilizability of Bovine Oocytes Cryopreserved in Various Cryoprotectants after In-Vitro Maturation and their Chromosomes at the First Cleavage Division. J. Mamm. Ova. Res. 15(1), 37–42. | ||
| How to Cite this Article |
| Pubmed Style Rimayanti R, Khairullah AR, Madyawati SP, Rantam FA, Mustofa I, Widjiati W, Srianto P, Akintunde AO, Pratama BP, Ahmad RZ, Wardhani BWK, Khalisa AT, Wibowo S. The role of heat shock protein 70 on oocyte apoptosis during vitrification. Open Vet. J.. 2025; 15(10): 4847-4864. doi:10.5455/OVJ.2025.v15.i10.5 Web Style Rimayanti R, Khairullah AR, Madyawati SP, Rantam FA, Mustofa I, Widjiati W, Srianto P, Akintunde AO, Pratama BP, Ahmad RZ, Wardhani BWK, Khalisa AT, Wibowo S. The role of heat shock protein 70 on oocyte apoptosis during vitrification. https://www.openveterinaryjournal.com/?mno=252630 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.5 AMA (American Medical Association) Style Rimayanti R, Khairullah AR, Madyawati SP, Rantam FA, Mustofa I, Widjiati W, Srianto P, Akintunde AO, Pratama BP, Ahmad RZ, Wardhani BWK, Khalisa AT, Wibowo S. The role of heat shock protein 70 on oocyte apoptosis during vitrification. Open Vet. J.. 2025; 15(10): 4847-4864. doi:10.5455/OVJ.2025.v15.i10.5 Vancouver/ICMJE Style Rimayanti R, Khairullah AR, Madyawati SP, Rantam FA, Mustofa I, Widjiati W, Srianto P, Akintunde AO, Pratama BP, Ahmad RZ, Wardhani BWK, Khalisa AT, Wibowo S. The role of heat shock protein 70 on oocyte apoptosis during vitrification. Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 4847-4864. doi:10.5455/OVJ.2025.v15.i10.5 Harvard Style Rimayanti, R., Khairullah, . A. R., Madyawati, . S. P., Rantam, . F. A., Mustofa, . I., Widjiati, . W., Srianto, . P., Akintunde, . A. O., Pratama, . B. P., Ahmad, . R. Z., Wardhani, . B. W. K., Khalisa, . A. T. & Wibowo, . S. (2025) The role of heat shock protein 70 on oocyte apoptosis during vitrification. Open Vet. J., 15 (10), 4847-4864. doi:10.5455/OVJ.2025.v15.i10.5 Turabian Style Rimayanti, Rimayanti, Aswin Rafif Khairullah, Sri Pantja Madyawati, Fedik Abdul Rantam, Imam Mustofa, Widjiati Widjiati, Pudji Srianto, Adeyinka Oye Akintunde, Bima Putra Pratama, Riza Zainuddin Ahmad, Bantari Wisynu Kusuma Wardhani, Andi Thafida Khalisa, and Syahputra Wibowo. 2025. The role of heat shock protein 70 on oocyte apoptosis during vitrification. Open Veterinary Journal, 15 (10), 4847-4864. doi:10.5455/OVJ.2025.v15.i10.5 Chicago Style Rimayanti, Rimayanti, Aswin Rafif Khairullah, Sri Pantja Madyawati, Fedik Abdul Rantam, Imam Mustofa, Widjiati Widjiati, Pudji Srianto, Adeyinka Oye Akintunde, Bima Putra Pratama, Riza Zainuddin Ahmad, Bantari Wisynu Kusuma Wardhani, Andi Thafida Khalisa, and Syahputra Wibowo. "The role of heat shock protein 70 on oocyte apoptosis during vitrification." Open Veterinary Journal 15 (2025), 4847-4864. doi:10.5455/OVJ.2025.v15.i10.5 MLA (The Modern Language Association) Style Rimayanti, Rimayanti, Aswin Rafif Khairullah, Sri Pantja Madyawati, Fedik Abdul Rantam, Imam Mustofa, Widjiati Widjiati, Pudji Srianto, Adeyinka Oye Akintunde, Bima Putra Pratama, Riza Zainuddin Ahmad, Bantari Wisynu Kusuma Wardhani, Andi Thafida Khalisa, and Syahputra Wibowo. "The role of heat shock protein 70 on oocyte apoptosis during vitrification." Open Veterinary Journal 15.10 (2025), 4847-4864. Print. doi:10.5455/OVJ.2025.v15.i10.5 APA (American Psychological Association) Style Rimayanti, R., Khairullah, . A. R., Madyawati, . S. P., Rantam, . F. A., Mustofa, . I., Widjiati, . W., Srianto, . P., Akintunde, . A. O., Pratama, . B. P., Ahmad, . R. Z., Wardhani, . B. W. K., Khalisa, . A. T. & Wibowo, . S. (2025) The role of heat shock protein 70 on oocyte apoptosis during vitrification. Open Veterinary Journal, 15 (10), 4847-4864. doi:10.5455/OVJ.2025.v15.i10.5 |