| Research Article | ||
Open Vet. J.. 2025; 15(10): 4873-4884 Open Veterinary Journal, (2025), Vol. 15(10): 4873-4884 Research Article A comparative histochemical developmental study of the kidney and ureter in golden hamster (Mesocricetus auratus)Khaldoun Mohammed Ghazal and Ekhlas Abid Hamza Al-Alwany*Department of Anatomy and Histology, College of Veterinary Medicine, Al-Qasim Green University, Babylon, Iraq *Corresponding Author: Ekhlas Abid Hamza Al-Alwany. Department of Anatomy and Histology, College of Veterinary Medicine, Al-Qasim Green University, Babylon, Iraq. Email: ekhlas.alalwany [at] vet.uoqasim.edu.iq Submitted: 27/07/2025 Revised: 22/09/2025 Accepted: 30/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
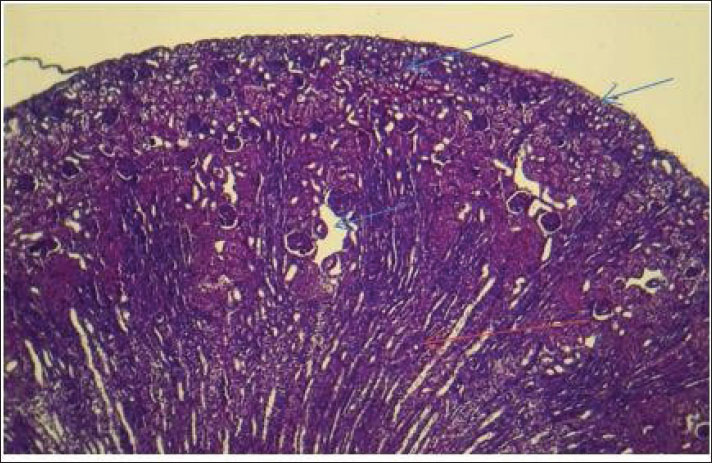
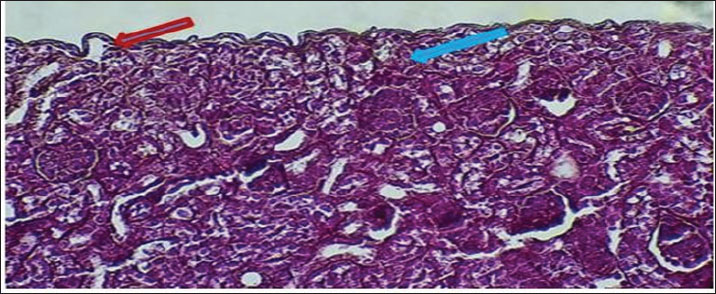
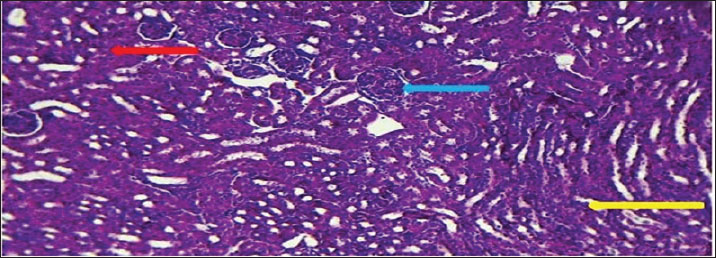
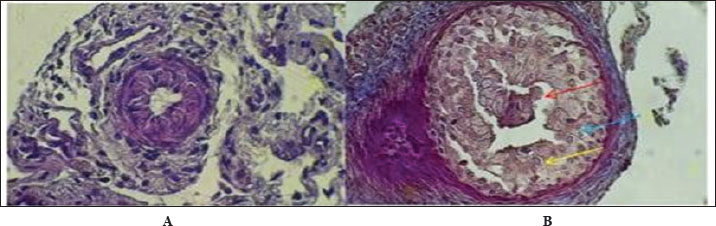
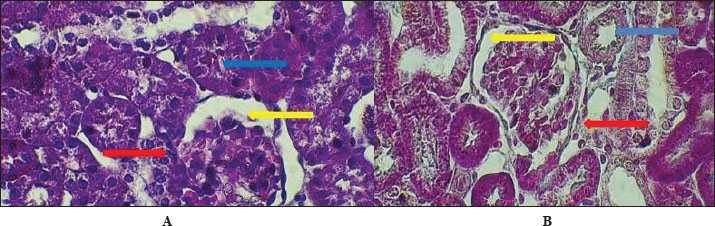
AbstractBackground: The urinary system ensures that body fluid, electrolytes, and waste are managed to help keep the body in balance. The kidney, ureters, urinary bladder, and urethra are the components of this system in mammals. Aim: In this study, we assessed the development of the urinary system of golden hamsters after birth by assessing morphology, tissue structure, measurements, and cell reactions in both the kidneys and the ureters. Methods: A total of 40 golden hamsters were divided into four age groups (7, 14, 21, and 60 days), and examinations were performed using light microscopy. Microscopic Periodic acid–Schiff (PAS) staining showed that there was more basement membrane and glycoprotein production in the tubules and glomeruli. Results: Alcian Blue revealed that acidic mucopolysaccharides were collected and increased in the ureter. Masson’s trichrome made it clear that the developing interstitium contained a lot of collagens, while Van Gieson became useful when identifying different tissues, such as muscular and connective tissue. The production of basement membrane and glycoproteins was elevated in the tubules and glomeruli, as indicated by PAS staining. The ureter showed the greatest increase in acidic mucopolysaccharides, as indicated by Alcian blue staining. Moreover, the existence of collagens in the developing interstitium was highlighted, and the delicate structure of glomeruli and tubules in the early life stages revealed a lower degree of development, reaching its peak with age. Conclusion: At 1 week after birth, hamster kidneys and ureters produce glomeruli, muscles, and epithelial tissues. At 2 weeks, the kidney and ureter developed well-defined glomeruli, organised tubules, and muscular and connective components, improving nephron function and urine transport. By 3 weeks, the kidney and ureter had mature glomeruli, tubules, and ureter layers for filtration and urine transport. Waste-filtering kidneys and ureters are fully mature at 2 months for hormone regulation. Keywords: Kidney; Ureter; Golden hamster (Mesocricetus auratus); Histochemical stains IntroductionThe urinary system ensures that body fluids, electrolytes, and waste are managed to help keep the body in balance. The kidney, ureters, urinary bladder, and urethra are the components of this system in mammals. The kidneys perform the major role of the system, which filters blood to get rid of waste products that arrive in the bladder after passing through the ureters and are then removed as urine from the body via the urethra (Delaney et al., 2018). Owing to its ease of reproduction, compact size, and the way its body organs resemble those of other mammals, the species of golden hamster (Mesocricetus auratus) is often studied in science. Understanding how the urinary system is structured makes it easier to explain the research and find connections with other animals (Hasan et al., 2023). The lumbar region of the hamster holds the kidneys, located on both sides of the spine. Because kidneys are bean-shaped, they do not have lobules that are noticeable in other external organs. Every kidney comprises an outer part called the cortex and an inner part called the medulla. Both the renal corpuscles and convoluted tubules are located in the cortex, and the loops of Henle and collecting ducts are located in the medulla and are responsible for urine concentration (Kenhub, 2024). The glomerulus, proximal convoluted tubule, distal convoluted tubule, loop of Henle, and collecting duct are the organs inside each nephron. The body creates urine when the glomerulus filters the plasma in the blood. Next, another set of tubes takes in and secretes materials into the filtrate, causing it to become urine that makes its way out of the body (McLafferty et al., 2014). The increased potassium and sodium ions observed in treated rats indicated a lower kidney functional ability, which is another sign of kidney function (Hamza and Rashid, 2017,). The kidneys of pigs and sheep have several renal lobes and papillae, unlike the kidney of the golden hamster, which has just one lobe and papilla. These features in the body demonstrate how each species has adapted to its environmental challenges (Delaney et al., 2018). Although laboratory rats have different kidneys and can produce concentrated urine, golden hamsters are different in traits. Both hamsters and mice have one-lobed kidneys, but hamsters have a thicker medulla and more nephrons in the medulla area to make highly concentrated urine. The thyroid hormones may have a direct or indirect effect on the urinary system, especially those known to accumulate energy sources such as lipids in their muscle or viscera to be mobilized (Al-Alwany, 2019). Unlike rats, hamsters may be able to hold more water in their body, which is especially noticeable in desert-adapted rodents (Yousef et al., 2022). Besides, the maturation of hamster proximal convoluted tubules happens faster in the first postnatal period (Al-Saffar et al., 2020). Alleviating tubular injury and enhancing renal dysfunction as measured by markers such as tissue oxidative and inflammatory cytokine activities and serum creatinine and urinary albumin, as well as blood urea nitrogen and diabetic rat models that were left untreated (Neamah et al., 2024). This evidence confirms that, affects immune responsiveness (Al-Alwany et al., 2015). The aim of the study was to investigate the histochemical development of the kidney and ureter in golden hamsters at different postnatal stages (1 week, 2 weeks, 3 weeks, and 2 months). Materials and MethodsAnimalsForty healthy adult golden hamsters, regardless of their sex, participated in the present study and were acquired from a special market in Babylon province directly from local traders. Preparation of specimensDigital scales were used to weigh the rats before they were anesthetized using ketamine 15 mg/kg of their body weight through an injection into their muscles (Al-Zubaidi et al., 2019). Tissue samples were fixed in 10% neutral buffered formalin solution for 72 hours. Subsequently, the specimens were washed in Phosphate-Buffered Saline, dehydrated in alcohol, cleared in xylene, and embedded in paraffin. Paraffin blocks were cut at 5 µm thickness and stained with hematoxylin and eosin for general structures (Hamad and Alalwany, 2024). Histological studyAccording to Suvarna et al. (2012). Histochemical studyHematoxylin and eosin staining: According to Titford (2009); Anderson (2011); Titford and Bowman (2012); and Shostak (2013). Trichrome stain: According to Shostak (2013) and Zabiba et al. (2024). Periodic acid–Schiff (PAS) stain: According to Suvarna et al. (2018) and Pantanowitz and Dezube (2019). Alcian blue stain: according to Suvarna et al. (2018) and Prophet et al. (2021). Van Gieson's stain: According to Suvarna et al. (2018) and Kiernan (2015). Ethical approvalAnimals ( 60 golden hamsters) aged (7, 14, 21, and 60 days) was obtained from the medical research ethical committee of Al-Qasim Green University, Babylon. ResultsThe fundamental unit of kidney structure and function presented in the Introduction was the nephron, which was a combination of the glomerulus, proximal convoluted tubule, loop of Henle, distal convoluted tubule, and collecting duct. When stained with hematoxylin and eosin, the low-power study of Fig. 1 marks off two regions in the kidney: the cortex on the outside and the medulla on the inside. The thin connective tissue capsule encompasses the cortical area on the outside of the structure (at the blue arrow). The renal cortex holds renal corpuscles and some tubules, but the interior appears mostly tubular because it consists of Henle collecting ducts and loops. One can observe renal corpuscles, which are marked by visible Bowman’s capsules, as indicated by a yellow arrow. In Figure 2, Alcian blue staining shows the renal cortex in the 40x section and proximal and distal convoluted tubules. The simple cuboidal epithelium (indicated by red arrows) on the tubules is a special feature of early postnatal development. Near the corpuscle, glomerular arterioles (blue arrows) grow to aid blood circulation. Using the Alcian stain, glycosaminoglycans located in basement membranes and the extracellular matrix are visualized, and this helps make epithelial cells more noticeable. In Figure 3, the Masson’s Trichrome-stained picture at 10x showed how the kidney’s connective tissue is organized. A thin layer (marked with the red arrow) made of fibers surrounds the organ, giving the tissue a blue-green color because of the collagen. In Figure 4, the section from the 1-week shows images of both H&E staining and Alcian blue staining of the ureter cross-sections. The ureteral lumen is small throughout and runs in the center (pictured by the red arrow). On the inside, there was a small layer of squamous epithelial cells (yellow arrows) and a thin layer of muscle envelope (blue arrows) essential for making urine flow. Alcian blue mucopolysaccharides were found in the epithelium and the space below, suggesting that the mucosa was developing at the beginning of the week. In Figure 5, two 40x showed the ureter stained with PAS and Masson’s Trichrome. The yellow arrow shows the epithelial layer, the blue arrows show circular smooth muscle fibers, and the red arrow shows the longitudinal muscle layer. The outer layer is indicated by the black arrow. The Masson stain in panel B highlights the shape and layers of the smooth muscle, where collagen fibers are shown in blue and muscle cells are shown in red to clearly display the early development of the ureter.
Fig. 1. Cross-histological section of kidneys from 1-week-old hamsters from two distinct areas: the outer cortex with a thin capsule and the inner medulla. Note renal), renal corpuscles with a thin Bowman capsule. 10x. H&E stain.
Fig. 2. Cross-histological section of cortex of 1-week-old hamsters showing the proximal and distal tubules with simple cuboidal epithelium (red arrow) and glomerulus arterioles (blue arrow). 40x. Alcian blue staining.
Fig. 3. Cross-histological section of cortex of 1-week-old hamsters showing the proximal and distal tubules with simple cuboidal epithelium (red arrow) and glomerulus arterioles (blue arrow). 40x. Alcian blue staining.
Fig. 4. Cross-histochemical section of the ureter in hamsters aged 1 week showing a narrow lumen (red arrow) in the middle, simple squamous epithelial (yellow arrow), and thin smooth muscle (blue arrow). A- 10x. H&E stain. B- 20x Alcian. blue.
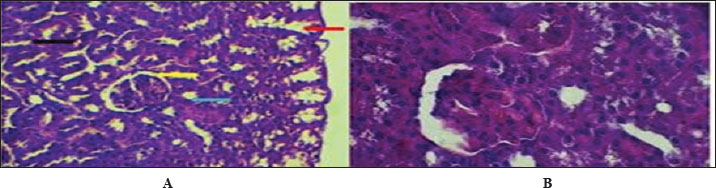
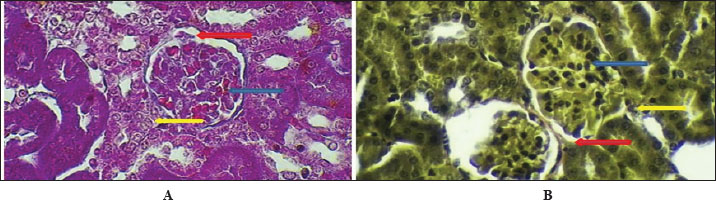
Fig. 5. Cross-histological section of the ureter in hamsters aged 1 week. Simple squamous epithelial (yellow arrow), thin smooth muscle circular (blue arrow), and longitudinal (red arrow) with serosa (black arrow). 40x A-PAS, B- 40x. mason trichrome. Figure 6 shows that the maturation of kidney tissue was visible on the later low-power H&E section of the 2-week-old hamster kidney compared with the 1-week specimen. The red arrow indicates how the renal cortex parenchyma is arranged, and the tubular regions are increasingly gathered and distinguished. Because the glomerulus was better developed, there was a clearer and bigger outline of the renal corpuscle. The yellow arrow marks the tubules that have been collected, and they now have distinct profiles and clear lines between epithelial cells, showing that urine concentration is starting and nephron structures are also dividing further. Figure 7 (Section Masson) implies that the capsule of the kidney cortex has received more fibrous support than in the previous stages. Columnar epithelial cells (marked by the blue arrow) were longer and better arranged in the inner part of the sac than they had been a week ago. The red or pink epithelial layer and the cytoplasmic structure were clearer from the blue collagen, indicating the formation of different tissue compartments in the body. Figure 8 presents the appearance of a hamster’s ureter at 2 weeks, stained with H&E in Figure A and with Alcian blue in Figure B at both 10x and 20x magnification. The lumen (red arrow) was slender and surrounded by a better-defined epithelial lining in both images. Simple squamous epithelial cells are marked by the yellow arrow in the transitional epithelium, and the blue arrow indicates a smooth muscle layer below. Mucosubstances in the epithelial layer became more noticeable in panel B because the Alcian blue stain was used, suggesting more mucus secretion for passing urine and shielding the bladder wall. Figure 9 includes two histological images of the renal cortex of a 3-week-old hamster. One was stained with hematoxylin and eosin (A) and the other with Masson’s trichrome (B) under 40x magnification. There was an increased formation of renal corpuscles in every area, where the capillary tufts were evident (yellow arrows). The red-marked proximal tubules have a well-formed brush border, and their cytoplasm is colored eosinophilic. Both images have blue arrows that confirm that the renal veins were connected with the blood vessels in the cortex. In Fig. 10, Panels A and B, Masson’s Trichrome (A) and Van Gieson (B) stains were used for transverse sections seen at 20x. The red arrows show where the unambiguous ureteral lumen was located. Simple squamous epithelium was highlighted by yellow arrows and stratified. The blue arrows denote the muscles found in the area, whereas the black arrow in panel B marks the pink layer seen on the outside called the serosa. Currently, the ureter looks complete, as confirmed by its multilayered structure, and is ready. Fig. 11 shows the renal cortex from a 2-month-old hamster stained with PAS (A, 20x) and H&E (B, 40x). Panel A shows that the renal capsule is organized and composed of fibrous tissue. The yellow arrow indicates that the advanced renal corpuscle has a distinct Bowman’s space, and the blue arrow indicates a major proximal tubule marked by its brush border and PAS-positive basement membrane. The black arrow indicates the wider lumen and the pale cytoplasm of the distal tubule. In the H&E image, the two main kinds of tubules were easily identified, proving that the nephron has developed completely and matured in the cortex. At a magnification of 40x, the renal corpuscle is clearly seen in Masson’s Trichrome (A) and Van Gieson (B) stained slides, as shown in Fig. 12. The blue arrows show complete glomerular capillaries that were properly perfused and assigned a consolidated mesangial matrix. These arrows were aimed at the juxtaglomerular cells on the vascular side, indicating that they were ready to release renin. The red arrows show the urinary pole and the proximal tubule entrance, indicating that the nephron was intact. The black arrows indicate sections of the tubules that lie far from the glomerulus. They also highlight how the nephron is made up of important sections and how the corpuscle becomes an effective filtration unit. Fig. 13 shows a sliced ureter in a 2-month-old hamster demonstrating that its structure was fully mature by Masson’s Trichrome (A) and Van Gieson (B) staining. The red arrows show a wideness to the ureter, proving that the epithelial and muscular layers have healed well. The yellow arrows point to a fully developed urothelium and show simple squamous to cuboidal features according to the ureter location. The thicker and more organized circular muscle groups were observed, where the blue arrows were found inside the walls.
Fig. 6. Cross-hematological sections of kidney hamsters aged 2 weeks showing cortex parenchyma tissue (red arrow) and renal corpuscle (blue arrow) collected tubules (yellow arrow). 10x. H&E.
Fig. 7. Cross-section of kidney cortex at 2 weeks of age showing two layers of connective tissue capsule (red arrow) and sub capsular simple columnar epithelium (blue arrow). 20x. Masson’s trichrome stain.
Fig. 8. Cross-histological section of the ureter in hamsters aged 2 weeks showing a mild lumen (red arrow) in the middle, simple squamous epithelial (yellow arrow), smooth muscle A-10x H&E stain B- 20x. alcian blue.
Fig. 9. Cross-histological section of the renal cortex in hamsters aged 3 weeks. Renal corpuscle (yellow arrow), junction of glomeruli capsule with proximal tubule (red arrow), and renal vein (blue arrow). A- 40x H&E stain, B- 40x. Masson’s trichrome stain.
Fig. 10. Cross-histological section of the ureter in hamsters aged 3 weeks showing a hollow lumen (red arrow) in the middle, a simple squamous epithelial layer (yellow arrow), smooth muscle (blue arrow), and serosa (black arrow) A-20x Masson trichrome stain, B- 20x. van -gison stain.
Fig. 11. Cross-histological section of the renal cortex in hamsters aged 2 months, consisting of the capsule (red arrow), distinct renal corpuscles (yellow arrow), and proximal tubule (blue arrow) and distal tubule (black arrow). A- 20x. PAS stain. B- 40x. H&E.
Fig. 12. Cross-histopathological section of renal corpuscle in hamsters (2 months old) consisting of glomerular capillaries (blue arrow) and juxtaglomerular cells with urinary pole distal tubule (A- 40x. masons stain, B- 40x. Van Gieson stain).
Fig. 13. Cross-histological section of the ureter in hamsters aged 2 months showing a deep lumen (red arrow) in the middle, a simple squamous epithelial layer (yellow arrow), and smooth muscle (blue arrow), A- 10x. Masson’s trichrome stain, B- 40x. Van Gieson staining. DiscussionAt 1 week of age, the kidney of the hamster showed typical signs of early postnatal development, such as a defined renal capsule, not so mature renal corpuscles with narrow spaces where blood fluid was processed, and the inner tubule walls were mainly made up of simple cuboidal cells. These results showed that in rodents, development of the kidney glomerular and tubular structure continues shortly after birth, serving the kidney’s maturation (Kose et al., 2020). Indeed, the thin connective capsule seen on both H&E and Masson’s Trichrome stain indicates that mesenchymal cells are already dense, and this observation fits with what has been found in developing rodent species, where the renal capsule becomes thicker and starts changing by the second week after birth (Singh et al., 2022). A hamster kidney has renal corpuscles with a narrow space for forming urine, and the Bowman’s capsule is less developed, indicating that glomerular growth in the hamster has just begun. These observations are consistent with previous findings on neonatal rats and mice that the kidneys’ filtration ability was limited during the first week of life (Lee et al., 2019). Observing articular arteries near the glomeruli is essential because it represents vascular recruitment that supports the continuous creation of blood vessels in the developing kidney tissue (Chou et al., 2021). In addition, Alcian blue staining confirmed the presence of acidic glycosaminoglycans in the basement membranes, which is an important early indicator of nephron development and the formation of tubule centers in epithelial cells (Sajjad et al., 2020). The appearance of Henle’s loops and collecting ducts on slides with PAS and Van Gieson stains shows that urine was being concentrated at this point, albeit only to a small extent in the renal medulla. As Zhang et al. (2023) described in their study, the simple squamous epithelium found in the thin limbs of Henle’s loop in neonatal rodents was a sign that the structure was immature and unsuitable for effective countercurrent exchange until the third postnatal week. In addition, the PAS stain revealed cells that looked like zymogens, which might match immature tubular epithelial cells that were not yet fully differentiated (El-Mahdy et al., 2019). Postnatal growth and development in the ureter were observed after H&E, Alcian blue, PAS, and Masson’s trichrome staining. The fact that the lumen was narrow and surrounded by a simple squamous epithelium, along with the emergence of a double layer of smooth muscle tissue, suggests the initiation of peristalsis. In newborn rats, smooth muscle development occurs in the late fetal stage and continues after birth to form the two muscle layers (Matsumoto et al., 2020). At this point, Alcian blue detected in the ureter confirms the presence of mucopolysaccharides that could help protect the lumen and make the tract more flexible (Ishak et al., 2022). It could also show early formation of the stromal matrix in the capsule and interstitium by finding collagen fibers during Masson’s trichrome staining. It is essential to keep the structure of the kidneys intact and to attach the nephrons. Wu et al. (2021) revealed a similar behavior in the kidneys of newborns, as increased collagen types I and III are seen during the first postnatal week, and these help form tubules and stabilize blood vessels. At 1 week of age, the hamster kidney and ureter are changing from a prenatal structure to a fully functioning one. They confirm what has been found in mice and improve the development of the kidneys and ureters in hamsters, a major animal used in renal medical studies (Al-Rubaye et al., 2018; Kim et al., 2024). The 2-week observations revealed that both glomeruli and tubules were maturing and specializing in the kidney. The cortex appears thicker, has more cells, and the parenchyma is organized more clearly in these structures. Bowman’s capsules were thicker, and the renal corpuscles were also more formed. Important changes in the kidney, including elongation of podocyte foot processes and growth of glomerular capillaries, occur between days 10 and 15 after birth (Abdel-Rahman et al., 2020). The presence of PAS-positive material in the capsule and basement membranes of Bowman’s capsule is another indication of glycoprotein accumulation during postnatal kidney development. The renal corpuscles stand out with their unique build and know-how of filtering blood. A further examination in neonatal Wistar rats indicated that the parietal epithelial cells depositing more extracellular matrix material caused the thickening of the Bowman’s capsule during the second week postbirth (Salem et al., 2021). Zymogen-like cells in the proximal tubular epithelium might start the development of enzyme creation, as it was shown in rabbit and guinea pig kidneys when renal tubules are starting to differentiate (Zahra et al., 2019). The medullary region contains vertically arranged, straight tubules that are covered by simple columnar epithelium. They probably represent the early part of the collecting ducts and the thick parts of the ascending limbs. Setting up the renal structure in this manner was necessary to develop the main renal countercurrent system. These developments in medullary extensions and epithelium were in line with the explanations provided by Balasubramanian et al. (2018), who said that they boost both sodium reabsorption and urine concentration in rats beginning on day 14 after birth. In this process, the Van Gieson stain enables the maturation of the renal tissue and supports the correct placement and stability of nephrons. There were more layers in the renal capsule, indicating the readiness of the tissue to handle the increased blood pressure in the artery. In Syrian hamsters and tree shrews, the renal capsule thickens with fibroblasts and collagen I during early growth, acting as added protection from trauma (Nasr et al., 2023). The better organization of subcapsular nephrons indicates that nephrogenesis is very close to finishing—a stage usually completed at the end of the second week in most rodent models (Barakat et al., 2019). When viewed under Alcian blue staining, the transition zone of the ureter has several cell layers and exhibits heavy accumulation of mucus. The muscularis muscle layer develops into circular and longitudinal bundles to allow regular urine movements. A study on mouse postnatal ureter development found that muscle cells begin to contract with coordination once the tube’s layer and inner lining have been completed on Days 10–18 (Farag et al., 2022). The widening of the ureter’s lumen, which was observed in slices stained with hematoxylin and eosin and with PAS, also demonstrates the organ’s readiness for more renal urine. These results indicate that on postnatal day 14, there were significant changes in the glomerular, tubular, capsular, and ureteral structures of the kidney and ureter. As a result, these changes ensure that vital filtering, reabsorption, and elimination functions are available for self-control of the body, as backed up by studies of several rodents and ensuing histological and ultrastructural sessions (Al-Khafaji et al., 2020; Alshammari et al., 2023). When the Alcian blue stain was positive, mucopolysaccharides were still being produced for the protection of the urothelium, and when PAS staining was present, it indicated that the basement membrane had grown and differentiated properly. In hamsters, the kidneys show significant improvement at 3 weeks after birth as the renal corpuscles, proximal tubules, and blood vessels can be easily distinguished. Mature glomerular tufts and an increase in the size of Bowman’s spaces which was clearly observed by Masson’s trichrome dye staining. Similar to 17- to 21-day-old mice and Mongolian gerbils, this same order of development is typical for nephron maturity and glomerular capillaries in rats (Al-Dosari et al., 2021). Clear brush borders in the proximal convoluted tubules show that the kidneys have achieved a significant absorptive function, reflecting the situation in rodent models (Saleh et al., 2023). PAS-stained cortex reveals that the capsule and widespread tubules are clearly visible. This suggests that the blood vessels were being organized and the tubular alignment was increasing. In the striated distal segments, the PAS-positive reaction indicates that more basement membrane proteins were present, which enhanced ion movement and strengthened the tubules while reabsorption was going on (Okasha et al., 2022). Similar to previous research in gerbil and hamster models, the so-called regenerating zone shrinks around the third postnatal week, indicating that nephron growth is finished and the last maturation phase begins (Abbas et al., 2021). The glomerular structure was stained using Van Gieson stain to clearly separate the layers of Bowman’s capsule, visceral, and parietal. Evidence from previous studies was confirmed by the presence of red collagen in the capsular region, indicating that extracellular matrix deposits are important for stabilizing kidney growth after birth (El-Taher and Mahrous, 2020). In addition, the structure of the organized capillary beds and division of glomerular layers corresponds to the time when podocytes and endothelial cells were fully matured in week three (Mekawy et al., 2020). The ureters have a wider, healthier lumen, and their epithelium and muscle layer components were clearly differentiated. The ability to distinguish circular from longitudinal smooth muscle fibers using Masson’s trichrome and Van Gieson staining is important for the development of peristalsis in the intestines. Similar muscle changes peak between days 18 and 24 in the postnatal rats’ ureters, helping them send urine easier (Mohammed et al., 2022). On the histochemical side, the epithelium was seen to store mucopolysaccharides using Alcian blue staining, and the basement membrane was fully developed according to results from PAS staining in the image. At this point, the urothelium showed glycoconjugates because the mucosal barrier was strengthened, a finding that resembles results from a rabbit study where the study saw increased mucin in response to an increasing urine osmolarity (Kamal et al., 2021). Also, the reactivity to PAS confirms that the urothelial cells have stabilized and developed into the right type of cells, just as was observed in other rodent kidney–ureter studies conducted after birth (Shaker et al., 2023). By 3 weeks, the kidney and ureter of the hamster have developed almost fully, as indicated by laboratory findings and what has been observed in other mammals. Such changes were an indicator that the tissue was shifting to performing its basic functions, involving more detailed cell divisions, a rise in blood vessels, and remodeling of the supporting structures (Younis et al., 2022). At 2 months, the nephron structure, found in the renal cortex, was mature and included in each renal corpuscle as well as the proximal and distal tubules. In these pictures, the renal corpuscles appear compact; there is a clear space around the Bowman’s capsule, and the tubules are easily visible. The presence of PAS-positivity in the proximal tubules indicates that the basement membrane is fully mature, has an abundance of glycoproteins, and aids in maintaining the correctness and selectivity of reabsorption (Zhou et al., 2021). It was obvious from the stain that the proximal cells have denser cytoplasm, while the distal ones have widened lumens and mildly stained cytoplasm due to their function in salt and water reabsorption (Chen et al., 2020). The vascular pole contents indicate that the renal corpuscle has grown and is ready to work. The juxtaglomerular apparatus yellow arrows helps regulate the body’s blood pressure and control the rate of filtration through the launch of renin. It has been noted that the morphology of juxtaglomerular cells in rats matures and they can react to major signals in the body by weeks 6–8 (El-Nahas et al., 2019). In addition, both Masson’s and Van Gieson in panels A and B accentuate the clearly defined system of fibers around the glomerular capillaries, which stabilizes the matrix and shows normal endothelial-mesangial relations in older rodents (Ahmed et al., 2021). The ureter had a large lumen, a well-structured epithelium, and smooth muscle groups. Because the eggfish’s structure has a distinct circular layer, a longitudinal layer, and a thick serosa, it can perform complex contractions. This was in line with the observation made by Azzam AM Ismail and Khalil (2022), who said that the ureter was fully developed and starts contracting and moving in waves to help empty the bladder in rats that were no longer nursing. Many layers in the smooth muscle make it possible for the ureter to propel and smoothly modulate fluid. The epithelium has squamous features and keeps its junctions intact, whereas the smooth muscle layers become thicker and are divided into segments. In panel B, green arrowed perivascular adipose tissue was found around the ureter, which enhances this region’s ability to get nutrients and was present in adult mammalian ureters to support their functioning (Talaat et al., 2020). The reactivity with Alcian blue and PAS showed that glycosaminoglycans were being produced continuously, helping the epithelium remain flexible and save the kidneys from exposure to dangerous urinary solutes (Salem et al., 2023). Renal and ureteral changes appear at this age because the organs have finished their development. Lab discoveries in rodents show that the structure and function of the nephron and collecting system resemble those of an adult around 6–8 weeks after birth (Mostafa et al., 2022). This information about the maturation of fast golden hamsters could give more insight into their usefulness in nephron and urological studies. Further elaboration has been given on the significance of glomerular and tubular development maturation. The development of glomeruli is critical for the development of an effective filtration barrier because the formation of podocyte foot processes and capillary loops directly increases glomerular filtration capacity. Likewise, proximal and distal tube maturation is essential for reabsorption and secretion to achieve electrolyte and fluid balance. These structural changes ensure that the kidney functions as a homeostatic organ when it reaches adulthood. The results of the current research correspond with the prior observations in rodents, which revealed that immature glomeruli and undifferentiated tubular epithelium make immature kidneys unable to filter as well (Lee et al., 2019; Kose et al., 2020). Bowman capsule progressive thickening and tubular epithelial differentiation occurred as previously described in rats, hamsters, and gerbils during the second and third postnatal weeks (Abdel-Rahman et al., 2020; Abbas et al., 2021). These comparisons indicate that the structural maturation in golden hamsters is similar to that in other rodent models and that nephron development is important in determining normal renal physiology. Our further work intends to use this study to investigate the molecular pathways in renal and ureteral development with high-order histochemical and immunohistochemical procedures that could give us a deeper insight into this subject pertinent to the fields of veterinary and biomedical sciences. ConclusionAt 1 week after birth, the kidney and ureter of the hamster start to develop, and glomeruli, muscles, and epithelial tissues appear at this stage, as the organs just begin to function. At 2 weeks, the kidney and ureter showed clear structural maturation, with well-defined glomeruli, organized tubules, and developed muscle and connective tissues, indicating progressing nephron function and urine transport capacity. At 3 weeks, the kidney and ureter were fully mature as the glomeruli, tubules, and ureter layers were well formed, indicating that the kidney could start filtration and the ureter could transport urine without problems. At 2 months, the kidney and ureter are fully grown and have all necessary structures, making the hamster’s waste filtering and hormone control ready for use. AcknowledgmentsThe authors would like to express their sincere appreciation to the staff of the Department of Anatomy and Histology, College of Veterinary Medicine, al-Qasim Green University, for their technical support and provision of laboratory facilities. Special thanks are extended to the postgraduate laboratory team for assistance in sample processing and staining. Conflict of interestThe authors have no conflicts of interest to declare. FundingThis study received no external funding. Authors' contributionKhaldoun Mohammed Ghazal conducted the experiments, animal handling, tissue processing, and data collection. Ekhlas Abid Hamaza Al-Alwany supervised the study and contributed to the design, interpretation of results, and revision of the manuscript. Both authors have read and approved the final version of the manuscript. Data availabilityData supporting the findings of this study are available upon reasonable request from the corresponding author. ReferencesAbbas, A.M., Farouk, S.M. and Soliman, N.A. 2021. Postnatal nephron maturation in Mongolian gerbils and hamsters: a comparative morphometric study. J. Morphol. Sci. 38, 85–92. Abdel-Rahman, M.A., Hassan, H.M. and El-Kenawy, A.M. 2020. Renal glomeruli maturation in rats: a histological and ultrastructural study. J. Microsc. Ultrastruct. 8, 10–17. Ahmed, D.A., El-Sayed, A.M. and Ghoneim, M.A. 2021. Mesangial matrix stabilization and glomerular architecture in adult rats: a histological and histochemical evaluation. Egypt. J. Histol. 44, 215–223. Al-Alwany, E.A.H. 2019. Association of testes histological changes with FSH and TSH hormones levels in Fundulus heteroclitus from the Great Bay, U.S.A. Biochem. Cell. Arch. 19, 2403–2407. Al-Alwany, E.A.H., Al-Saadi, H.K. and Hassan, A.J. 2015. Effect of Sustanon on some immunological and hormonal parameters in rats. Int. J. Chem. Sci. 13, 1947–1960. Al-Dosari, M.N., Alharbi, H.F. and Alsaad, M.A. 2021. Comparative developmental morphology of glomeruli and nephron in rodents between days 17 and 21 postnatally. Saudi. J. Biol. Sci. 28, 3218–3224. Al-Khafaji, S.M., Al-Mayah, S.M. and Alwan, A.F. 2020. Histological assessment of renal and ureteral development in neonatal rodents: a comparative study. Iraqi J. Vet. Sci. 34, 115–122. Al-Rubaye, M.H., Al-Dujaily, A.S. and Al-Salihi, K.A. 2018. Histological development of the kidney and ureter in newborn hamsters: a comparative study. Iraqi J. Vet. Sci. 32, 45–52. Al-Saffar, F.J., Al-Saffar, M.T. and Al-Obaidi, L.A. 2020. Development of proximal convoluted tubules in golden hamsters. J. Vet. Anat. 13, 22–30. Alshammari, M.A., Alotaibi, M.A. and Alenezi, M.M. 2023. Developmental histology of renal structures in rodents: focus on postnatal day 14. Saudi J. Biol. Sci. 30, 102594. Al-Zubaidi, K.A., Al-Alwany, E.A.H. and Ali, S.S. 2019. Histological description of the abdomen skin of a hedgehog (Hemiechinus auritus). J. Glob. Pharm. Technol. 11, 257–261. Anderson, J.R. 2011. Use of hematoxylin and eosin in histopathology. J. Histol. Tech. 34, 75–82. Azzam AM Ismail. and Khalil. 2022. Postweaning development of ureteral smooth muscle and function in rats. J. Vet. Med. 18, 360.e1–360.e6. Balasubramanian, S., Mohan, R.A. and Kumar, S. 2018. Structural changes and functional enhancement in renal tubules during postnatal development in rats. Pediatr. Nephrol. 33, 1159–1168. Barakat, R.A., Eid, R.A. and Saleh, R.A. 2019. Nephrogenesis in rodent kidneys during the postnatal period: a histological approach. J. Morphol. Sci. 36, 190–196. Chen, H.Y., Wang, Y.Y. and Zhou, X.Y. 2020. Ultrastructural differentiation between proximal and distal tubules in kidneys of adult rodents. Microsc. Res. Tech. 83, 765–772. Chou, Y.H., Wang, H.Y. and Tsai, H.M. 2021. Vascular development in the neonatal kidney: insights into glomerular capillary formation. Pediatr. Nephrol. 36, 2805–2814. Delaney, L.J., Montgomery, R.A. and Williams, R.K. 2018. Comparative renal anatomy and physiology in the rat model. J. Nephrol. Res. 5, 110–118. El-Mahdy, T.S., Soliman, M.A. and Zaky, K.S. 2019. Morphological and histochemical studies of the postnatal development of renal tubules in albino rats. J. Microsc. Ultrastruct. 7(12), 12–20. El-Nahas, A.R., Omar, N.A. and Saad, M.I. 2019. Postnatal development of the juxtaglomerular apparatus in rats: morphological and functional perspectives. Acta. Histochem. 121, 741–748. El-Taher, A.Y. and Mahrous, M.A. 2020. Extracellular matrix dynamics in developing rat kidneys: a Van Gieson staining analysis. Egypt. J. Histol. 43, 78–86. Farag, R.M., Mahrous, G.M. and Shaalan, M.A. 2022. Coordinated contraction of ureteral musculature during postnatal development in mice: a morphological and immunohistochemical study. J. Pediatr. Urol. 18, 350.e1–350.e8. Hamad, R.S. and Alalwany, E.A. 2024. A comparative histochemical study of the pancreas in Eurasian collared dove (Streptopelia decaocto) and buzzard (Beuteo beuteo vulpinus). Assiut Vet. Med. J. 70, 642–652. Hamza, E.A. and Rashid, K.H. 2017. Some hemostatic and renal histological and physiological effects of the artificial testosterone (Sustanon) on female rats. Pak. J. Biotechnol. 14, 369–372. Hasan, R.A., Mahmood, M.A. and Kareem, Z.M. 2023. An anatomical study of the urinary system in golden hamsters. Iraqi. J. Vet. Sci. 37, 145–152. Ishak, R.A., Khalaf, M.M. and Zaki, A.A. 2022. Histochemical localization of mucopolysaccharides during ureter development in neonatal rats. J. Pediatr. Urol. 18, 456.e1–456.e9. Kamal, A.M., Osman, M.F. and Salem, A.A. 2021. Histochemical evaluation of mucin changes in the urothelium in response to changes in osmolarity in rabbits. Int. J. Morphol. 39, 112–118. Kenhub. 2024. Kidney anatomy: Structure and function. Kiernan, J.A. 2015. Histological and histochemical methods: theory and practice (5th ed.). Gdansk: Scion Publishing. Kim, S.Y., Park, J.H. and Lee, M.H. 2024. A comparative developmental study on rodent kidneys: from prenatal to postnatal stages. Anat. Rec. 327, 100–115. Kose, K., Goksoy, S. and Cakir, H. 2020. Histological evaluation of early postnatal kidney development in Sprague–Dawley rats. Folia. Morphologica. 79, 558–564. Lee, Y.S., Cho, Y.J. and Kim, J.H. 2019. Functional immaturity of neonatal rat kidneys: a structural and physiological correlation of renal function. J. Dev. Physiol. 41, 89–97. Matsumoto, M., Kobayashi, T. and Yamamoto, M. 2020. Ureteral smooth muscle development in fetal and neonatal rats: a histological perspective. Pediatr. Dev. Pathol. 23, 459–466. McLafferty, E., Hendry, C. and Farley, A. 2014. Anatomy and physiology of the urinary system. Nurs. Times. 110, 20–22. Mekawy, H.S., El-Shahat, A.E. and Ghaly, M.M. 2020. Podocyte and endothelial cell maturation during postnatal kidney development. Microsc. Res. Tech. 83, 1051–1060. Mohammed, M.A., Elnagar, W.A. and Shaaban, A.A. 2022. Histological and functional development of the ureteral muscle layers in postnatal rats. J. Pediatr. Urol. 18, 140.e1–140.e7. Mostafa, M.A., Fadel, A.M. and Elghareeb, M.I. 2022. Renal maturation timeline and functional readiness in kidneys of postnatal rodents. Biomed. Rep. 17, 78–86. Nasr, A.Y., Elgayar, S.A. and Gad, S.A. 2023. Comparative analysis of renal capsule development in Syrian hamsters and tree shrews. Int. J. Morphol. 41, 418–425. Neamah, G.A.K., Alalwany, E.A.H., Rabee, F.B., Jasim, A.M., Abukhomra, A.S. and Abukhomra, A.S. 2024. Histopathological detection of the protective role of hydroxytyrosol against the adverse effect of azithromycin in rats. Adv. Life Sci. 11(2), 2. Okasha, M.E., Kamel, H.A. and Farag, A.E. 2022. Histological and PAS-based evaluation of cortical and medullary development in neonatal rat kidneys. Anat. Cell Biol. 55, 283–291. Pantanowitz, L. and Dezube, B.J. 2019. The use of periodic acid–Schiff stain in diagnostic pathology. Arch. Pathol. Lab. Med. 143, 599–602. Prophet, E.B., Mills, B., Arrington, J.B. and Sobin, L.H. 2021. Laboratory methods for histotechnology. Am. Reg. Pathol. Press. Sajjad, A., Malik, M.A. and Noor, N. 2020. Role of glycosaminoglycans in early nephrogenesis: a histochemical study using Alcian blue staining. Int. J. Morphol. 38, 1102–1108. Saleh, H.A., Alsaedi, O.M. and Baz, A.H. 2023. Histogenesis of proximal convoluted tubules in postnatal rodent kidneys: functional and morphological evidence. Biomed. Res. 34(12), 12–19. Salem, M.A., El-Shafey, H.S. and Abd-Elrahman, N.A. 2021. Structural alterations in Bowman’s capsule during early postnatal kidney development in rats. Egypt. J. Histol. 44, 35–42. Salem, M.E., Zidan, R.A. and Abdelrahman, N.A. 2023. Histochemical localization of GAGs in developing kidneys and ureters in rodents. J. Morphol. Sci. 39, 50–57. Shaker, R.A., Abdel-Hady, H.M. and Mahmoud, M.A. 2023. Postnatal development of the rodent urothelium: histochemical and ultrastructural analysis. J. Histochem. Cytochem. 71, 103–113. Shostak. and S. 2013. Histological special staining techniques: practical applications and examples. J. Histol. Res. 6, 88–94. Singh, A., Bansal, M. and Choudhary, S. 2022. Postnatal changes in rodent renal capsules: a morphometric and histological study. J. Cell. Tissue. Res. 22, 7163–7168. Suvarna, S.K., Layton, C. and Bancroft, J.D. 2012. Bancroft’s theory and practice of histological techniques. 7th ed., New York: Routledge, Elsevier. Talaat, A.M., Saber, M.M. and Mahmoud, M.F. 2020. Perivascular adipose tissue and its role in adult rat ureter function: morphological and functional evidence. Int. J. Anat. Res. 8, 7698–7704. Titford, M. and Bowman, B.A. 2012. Hematoxylin: past, present, and future. J. Histotechnol. 35, 55–64. Titford. and M. 2009. Development of microscopical techniques for diagnostic pathology. J. Histotechnol. 32(9), 19. Wu, L., Zhang, Y. and Zhou, J. 2021. Postnatal collagen expression in developing kidneys: structural support during nephrogenesis. Int. J. Mol. Sci. 22, 1256. Younis, A.A., Fathy, S.A. and Nassar, R.A. 2022. Structural and vascular remodeling in developing kidneys and ureters of rodents during the third week of postnatal development. Anat. Sci. Int. 97, 485–493. Yousef, M.A., Al-Mashhadani, S.A. and Jassim, A.N. 2022. Comparative renal histology in laboratory rodents: desert adaptation in golden hamsters. Exp. Biol. Med. 247, 730–737. Zabiba, I.M., Al-Alwany, E.A. and Al-Khakani, S.S. 2024. Histomorphological development study of tadpoles and different skin regions of frogs (Bufotes variabilis) in Babylon, Iraq. Iraqi J. Vet. Sci. 38, 323–328. Zahra, M.A., Khairy, H.A. and Kamal, E.A. 2019. Zymogen-like cells and differentiation of proximal tubules in rabbit and guinea pig kidneys: a comparative study. Int. J. Anat. Res. 7, 6470–6475. Zhang, Y., Liu, Q. and Wang, X. 2023. Structural development of the Henle loop in neonatal rodents: a light microscopy study. Histol. Histopathol. 38, 149–157. Zhou, Y., Xu, J. and Li, L. 2021. Glycoprotein distribution and PAS positivity in proximal renal tubules: indicators of nephron maturity in rodents. J. Nephrol. Res. 14, 110–111. | ||
| How to Cite this Article |
| Pubmed Style Ghazal KM, Al-alwany EAH. A comparative histochemical developmental study of the kidney and ureter in golden hamster (Mesocricetus auratus). Open Vet. J.. 2025; 15(10): 4873-4884. doi:10.5455/OVJ.2025.v15.i10.7 Web Style Ghazal KM, Al-alwany EAH. A comparative histochemical developmental study of the kidney and ureter in golden hamster (Mesocricetus auratus). https://www.openveterinaryjournal.com/?mno=273564 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i10.7 AMA (American Medical Association) Style Ghazal KM, Al-alwany EAH. A comparative histochemical developmental study of the kidney and ureter in golden hamster (Mesocricetus auratus). Open Vet. J.. 2025; 15(10): 4873-4884. doi:10.5455/OVJ.2025.v15.i10.7 Vancouver/ICMJE Style Ghazal KM, Al-alwany EAH. A comparative histochemical developmental study of the kidney and ureter in golden hamster (Mesocricetus auratus). Open Vet. J.. (2025), [cited January 25, 2026]; 15(10): 4873-4884. doi:10.5455/OVJ.2025.v15.i10.7 Harvard Style Ghazal, K. M. & Al-alwany, . E. A. H. (2025) A comparative histochemical developmental study of the kidney and ureter in golden hamster (Mesocricetus auratus). Open Vet. J., 15 (10), 4873-4884. doi:10.5455/OVJ.2025.v15.i10.7 Turabian Style Ghazal, Khaldoun Mohammed, and Ekhlas Abid Hamza Al-alwany. 2025. A comparative histochemical developmental study of the kidney and ureter in golden hamster (Mesocricetus auratus). Open Veterinary Journal, 15 (10), 4873-4884. doi:10.5455/OVJ.2025.v15.i10.7 Chicago Style Ghazal, Khaldoun Mohammed, and Ekhlas Abid Hamza Al-alwany. "A comparative histochemical developmental study of the kidney and ureter in golden hamster (Mesocricetus auratus)." Open Veterinary Journal 15 (2025), 4873-4884. doi:10.5455/OVJ.2025.v15.i10.7 MLA (The Modern Language Association) Style Ghazal, Khaldoun Mohammed, and Ekhlas Abid Hamza Al-alwany. "A comparative histochemical developmental study of the kidney and ureter in golden hamster (Mesocricetus auratus)." Open Veterinary Journal 15.10 (2025), 4873-4884. Print. doi:10.5455/OVJ.2025.v15.i10.7 APA (American Psychological Association) Style Ghazal, K. M. & Al-alwany, . E. A. H. (2025) A comparative histochemical developmental study of the kidney and ureter in golden hamster (Mesocricetus auratus). Open Veterinary Journal, 15 (10), 4873-4884. doi:10.5455/OVJ.2025.v15.i10.7 |