| Research Article | ||
Open Vet. J.. 2026; 16(3): 1743-1756 Open Veterinary Journal, (2026), Vol. 16(3): 1743-1756 Research Article Seasonal adaptations in the ultrastructural and immunohistochemical characterization of the epididymal duct in Meriz bucksBayan S. Saadi, Yahya A. Mohammed and Zeravan A. Mohammed*Department of Physiology and Anatomy, College of Veterinary Medicine, University of Duhok, Duhok, Iraq *Corresponding Author: Zeravan Abdularazaq Mohammed. Department of Physiology and Anatomy, College of Veterinary Medicine, University of Duhok, Duhok, Iraq. Email: zeravan.mohammed [at] uod.ac Submitted: 10/09/2025 Revised: 15/02/2026 Accepted: 28/02/2026 Published: 31/03/2026 © 2026 Open Veterinary Journal
ABSTRACTBackground: Although the epididymal duct (ED) supports sperm maturation and storage, its ultrastructural and immunohistochemical adaptations in seasonal breeders, such as the Meriz buck, remain unclear. Aim: This study aimed to investigate seasonal adaptations in the ultrastructure and immunohistochemical profile of the ED in Meriz buck during mating and non-mating seasons. Methods: Twenty-four Meriz bucks from Duhok Province, Iraq, at the College of Veterinary Medicine were restudied. Epididymal tissues were examined using Transmission Electron Microscopy and immunohistochemistry α-smooth muscle actin (α-SMA), S-100, and von Willebrand factor (VWF) to characterize cellular localization during the mating and non-mating seasons. Results: The epithelium of the ED comprised principal cells (PCs), basal cells, apical cells, narrow cells (NCs), and clear cells. Ultrastructurally, PCs displayed Abundant microvilli, apical blebs, and well-developed endocytotic apparatuses (vesicles and multivesicular bodies), which were more prominent in autumn (October) than in other seasons. Adjacent PCs were joined by occluding junctions, forming the Blood-Epididymal Barrier. During autumn, the supranuclear region of PCs exhibited a larger Golgi apparatus, extensive rough endoplasmic reticulum, and numerous mitochondria, whereas other epithelial cells exhibited notable seasonal variations. Immunohistochemical, α-SMA expression intensified in the peritubular and vascular smooth muscle layers during autumn (9.3 ± 0.01, p < 0.01), suggesting enhanced contractile activity or smooth muscle remodeling associated with reproductive seasonality. S-100 immunoreactivity was strongest in the PCs and NCs (7.5 ± 0.002, p < 0.01), whereas VWF expression in vascular endothelial cells increased significantly in autumn compared to other seasons (0.99 ± 0.01, p < 0.01). Conclusion: The study concludes that in October, increased ultrastructural activity and elevated expression of α- SMA, S-100, and VWF in the ED indicate increased reproductive function. Suggesting that this period corresponds to the Meriz bucks' mating season. Keywords: Epididymal duct, Ultrastructure, Immunohistochemistry, Seasonal variation, Meriz Buck. IntroductionThe Meriz is a unique breed of goat native to the mountainous areas of Iraq (Alkass and Merkhan, 2013; Mustafa et al., 2022). It is valued for producing cashmere, a luxurious and expensive natural fiber renowned for its exceptional softness and warmth (Aziz, 2009). It is widely used in the manufacture of special types of popular Kurdish clothing called mariz. Recently, the demand for this breed has increased, especially among younger generations who favor traditional clothing (Aziz, 2009). Unfortunately, the increase in demand has led to the slaughter of these animals during their peak productive and reproductive years. Consequently, the survival of Meriz goats is crucial for local livelihoods, biodiversity, and cultural heritage and is under serious threat. Addressing this requires controlled breeding. Strategies and a deeper understanding of the reproductive biology of the breed (Aziz, 2009). Reproductive efficiency is the foundation of sustainable livestock management. The fertility of male animals is affected by the functional integrity of the epididymis, a highly specialized part of the male reproductive tract (James et al., 2020). Epididymis plays a crucial role in sperm formation, concentration, and maturation, including the acquisition of motility and fertilization capability protection, and storage (James et al., 2020). Histomorphometrical and histochemical study by Saadi et al. (2024) on the epididymal duct (ED) of Meriz bucks showed that it consists of six distinct parts: the initial segment (IS), proximal segment (PS), and middle segment and distal segment (DS) of the corpus, and the PS and DS of the cauda. The lining epithelium of this duct comprises five types of cells: principal cells (PCs), basal cells (BCs), apical cells (ACs), narrow cells (NCs), and clear cells (CCs). Several protein markers serve as indicators of the functional state of the ED, including α-smooth muscle actin (α-SMA), S-100 protein, and von Willebrand factor (VWF). α-SMA is related to smooth muscle cell activity and contractility in the peritubular region for sperm transport. Calcium-binding S-10 participates in cellular processes, such as differentiation and signal transduction, and is expressed in the supporting cells of the reproductive tract (Alkafafy, 2024). VWF, an endothelial marker, is used for vascularization and epididymal tissue integrity (Mohammed, 2016). An immunohistochemical evaluation of these proteins across different seasons could provide valuable insight into reproductive readiness and optimal mating periods of Meriz bucks. Therefore, this study aimed to investigate the seasonal changes in ultrastructural and immunohistochemical parameters in the epididymis of the Meriz buck, as reported by Alkass and Merkhan (2013) and supported by recent molecular findings (Mustafa et al., 2022). The study sought to characterize key epithelial cell types, particularly clear cells, and to examine the seasonal expression patterns of α-SMA, S-100, and VWF. These findings are expected to provide a deeper understanding of the perm maturation and transport, as well as the adaptive mechanisms of reproductive physiology in response to seasonal and environmental variations. Materials and MethodsAnimals and housing conditionsTwenty-four clinically healthy 2-year-old Meriz bucks (Capra hircus kurdistanca) were sourced from Duhok Province, Iraq. Animals were maintained in semi-closed barns all year round and year-round in semi-closed barns and fed a mixed diet of grass and corn silage, and supplemented with concentrate and mineral additives (Mustafa and Yateem, 2019). The study region experiences a Mediterranean climate with high temperatures ranging from 3°C to 45°C and variable photoperiods: 9.5–10.5. In winter, 11–13 in spring, 14–14.5 in summer, and 10–12 in autumn. Animals were housed at the College of Veterinary Medicine, University of Tokyo. of Duhok, under controlled environmental conditions, and provided with water. To examine seasonal changes in the epididymis, two bucks were randomly selected each month over a year period (December 2020; November 2021) for ultrastructural Transmission Electron Microscopy (TEM) and Immunohistochemical analyses. Age was estimated using dental examination (Eubanks, 2012), and all animals were clinically healthy and free of blood and fecal-borne disease by the Department of Medicine and Surgery, College of Veterinary Medicine, University of Duhok, to ensure that all animals were disease-free. Preparation of tissue specimensTissue specimens were collected following surgical castration. Performed under local anesthesia. A 2% lidocaine solution was administered at a dose of 4–6 mg/kg to achieve analgesia through a combination of spermatic cord block and scrotal infiltration. The animals were restrained in lateral recumbency, and xylazine (0.05–0.1 mg/kg IM) was administered when necessary to ensure safe handling and to mitigate stress during the procedure (DeRossi et al., 2005). After castration, the testicular tissues were exteriorized through a scrotal incision, and the epididymides were carefully excised from the testes body, trimmed, and separated into their anatomical regions, and immediately processed for subsequent ultrastructural TEM and Immunohistochemical analyses. Transmission Electronmicroscopy studyFor ultrastructural analysis, approximately 0.5–1 mm3 fragments of the were excised and prefixed with 2.5% Glutaraldehyde (5 ml of 50% glutaraldehyde with 95 ml of phosphate-buffered saline) (Morris, 1965). In 0.1 M PBS at pH 7.2 for 48 hours at 4°C. After the primary fixation, the samples were transferred into labeled airtight tubes, packed in dry ice containers, and transported to Deypetronic Laboratory (Tehran, Iran) for further processing. Samples were processed following the TEM protocol of the Deypetronic Laboratory, Tehran, Iran. In the laboratory, secondary fixation was performed in 1% osmium tetroxide (Os O₄) for 1–2 hour, followed by dehydration through a graded ethanol series (30%, 50%, 70%, 90%, and 100%). Dehydrated tissues were cleared in propylene oxide and embedded in epoxy resin (2:1). The ultrathin sections (60–90 nm) were cut using an ultramicrotome, mounted on copper grids, and stained with uranyl acetate and lead citrate. To enhance the contrast. Sections were examined using a high-powered transmission electron microscope. microscope (EM208S, Philips, Nederland) operating at 80–120 kV. Digital micrographs captured at various magnifications for evaluating epithelial cell morphology, stereocilia, and basal lamina characteristics, and seasonal ultrastructural changes. Immunohistochemical studyFor the immunohistochemical analysis, the epididymal tissue samples (0.5–1 mm3) were fixed in Blouin’s solution, dehydrated through graded Ethanol, cleared with xylene, and embedded in paraffin. Sections of 4 µm thickness were cut using a rotary microtome and mounted on poly-L-lysine-coated slides. Antigen retrieval was performed by boiling sections in sodium citrate buffer (pH 6.0) at 800 W Microwave for 10 minutes, and the endogenous peroxidase activity was quenched using 3% hydrogen peroxide (v/v) H2O2 in methanol for 10 minutes at room temperature (Mohammed, 2016). Sections were blocked with 20% normal goat serum for 30 minutes and incubated at 4°C with the following primary antibodies: rabbit polyclonal anti-human VWF (Code No. A0082, 1:400 dilution (Dako, Glostrup, Denmark), rabbit polyclonal anti-S-100 (Code No. Z0311, 1:400 dilution, Dako, Glostrup, Denmark), and mouse monoclonal anti-human α-SMA (Code No. M0851, 1:100 dilution (Dako, Glostrup, Denmark). Endothelial cells were identified using the VWF, and α-SMA was used for peritubular and vascular Smooth muscle cells and epithelial cells with S-100 immunostaining. Negative controls were processed with rabbit or mouse Immunoglobulin G (IgG). After washing, the slides were incubated with biotinylated secondary antibodies and Peroxidase complex. Immunoreactivity was visualized using DAB as chromogen, counterstained with hematoxylin, dehydrated, cleared, and mounted in medium. Slides were coded to ensure unbiased analysis. Images were randomly acquired from 10 fields of view per coverslip per animal under 10 × magnification. The staining area was quantified as follows: using ImageJ software (National Institutes of Health, NIH, Bethesda, MD, USA). Captured images were arranged into a composite grid for analysis, and any artifacts or imperfections in the staining were excluded. Only the brown areas that exhibited positive staining for VWF, α-SMA, and S-100 staining were used in this study. Statistical analysisData were checked for normality and homogeneity of variances before analysis. Performing statistical analysis using residual plots and Bartlett’s test. Group comparisons were performed using one-way analysis of variance followed by Tukey’s post hoc test using the GenStat software (16th edition). Results were considered statistically significant at p < 0.05. All values are expressed as the mean ± SEM (standard error of the mean). Differences between seasons are indicated using superscript letters (a,b,c,d). Ethical approvalAll animal procedures were conducted by the scientific committee of the College of Veterinary Medicine. Medicine at the University of Duhok, issue date 1-10-2019, reference no. CVM20190110UoD. All animal experiments complied with the Animal Research: Reporting of in vivo (ARRIVE) guidelines. Experiments) guidelines (Kilkenny et al., 2010) for reporting animal research and adhering to the principle outlined in the Guide for the Care and Use of Laboratory Animals, 8th edition, 2011. All efforts were made to minimize animal suffering and reduce the number of animals used. ResultsUltrastructural findingsThe PCs were tall, columnar cells extending from the basal lamina to the lumen. In all parts of the ED, the PCs’ free surface was characterized by microvilli. During the mating season, the number of microvilli became fewer and shorter microvilli became fewer in number and shorter in the initial part of the caput (Fig. 1 A and B) and in all parts of the corpus (Fig. 1C), whereas in segments of the cauda, epithelial cells demonstrated increased height, along with significant Microvilli, resulting in densely packed microvillous formations (Fig. 1D). Branched microvilli were observe the first part of the caput and all segments of the corpus (Fig. 1B and C). Several Pinocytotic pits originate from the PCs’ free surface (Fig. 1B). Moreover, many apical blebs (Fig. 1D) project from the same surface. These blebs sometimes separated from the cells and released their contents (epididymsomes) into the ED lumen (Fig. 1E). Smooth-walled and coated-walled vehicles occupied the PCs’ supranuclear cytoplasm (Fig.1B and C). Vacuoles of different sizes were found in the supranuclear (Fig. 1A and C) and intranuclear cytoplasm of PCs (Fig. 1F). These vacuoles were either empty or contained electron-dense materials. In addition to the vacuoles, the supranuclear cytoplasm of the PCs contained large multivesicular bodies containing electron-dense granular material (Fig. 1 C and D). The lateral cell membranes of adjacent PCs were locked with the occluding junction. This junction was present along all ED segments (Fig. 1A, B, D). A large Golgi apparatus was present in the supranuclear region of PCs in all ED segments (Fig. 2A). Abundant rER with parallel, ribosome-studded cisternae throughout the cytoplasm (Fig. 2B). Mitochondria, varying in shape (spherical, oval, and elongated) and size (Fig. 1F), were distributed across the cytoplasm, with a higher concentration in the intranuclear region.
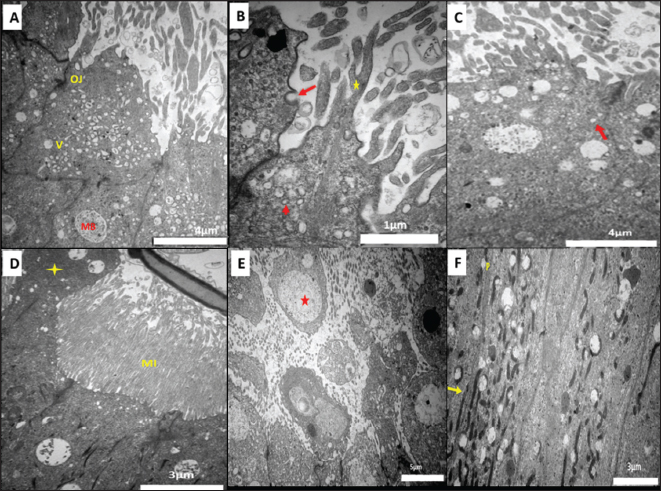
Fig. 1. Transmission electron micrograph of the epididymal duct of Meriz Bucks (autumn, October). (A)Caput (initial): principal cell (P), basal cells (BC), scale bar=5 µm. (B) Corpus (distal): principal cells (P) Rough ER (RER), scale bar=2 µm. (C) Caput (initial): Golgi apparatus (GA), scale bar=5 µm. (D) Caput (initial): large Golgi apparatus (GA), scale bar=4 µm.(E) Cauda (proximal): principal cells (P), Intraepithelial lymphocytes (IEL), scale bar=5µm. (F) Corpus (middle): principal (P) and basal cells (BC), scale bar=5µm. In the caput and corpus, the PCs had oval nuclei with slight indentations (Fig. 2C) or deep notches (Fig. 2D), whereas in the cauda, the nuclei were large and lobed (Fig. 2E). The BCs were small, pyramidal, with bilobed or irregularly oval nuclei in the caput and corpus (Fig. 2D, F) and rounded, notched Nuclei in the cauda (Fig. 3A). Some cauda BCs exhibited basal plasma membrane folding (Fig. 3A). ACs, located between PCs, had round or oval nuclei, prominent nucleoli, and a microvilli-free luminal surface, with mitochondria, vacuoles, rER, and Golgi apparatus cytoplasm (Fig. 3B). The NCs spanned the epithelium height, from the basal lamina to the lumen, with Spindle-shaped, dense nuclei, abundant vacuoles, and organelle-scarce cytoplasm (Fig. 3D). CCs had light-staining nucleoplasm and cytoplasm, with C-shaped vesicles, dense bodies, lysosomes and mitochondria. Intraepithelial Lymphocytes (ILEs)were irregularly rounded with indented nuclei and peripheral chromatin. Large and irregular IEMs with pseudopodia, triangular nuclei, and cytoplasm dense bodies, lysosomes, and organelles. IELs and IEMs were distributed throughout the epididymal epithelium (Fig. 3D). Ultrastructural seasonal changes: All BCs, ACs, NCs, CCs, and immune cells, lining all segments of the ED, showed no noticeable changes in their cellular ultrastructural characteristics. During all seasons of the year, except for the PCs. During the non-mating season, scarce microvilli and apical blebs were projected from the free surface of the PCs in all epididymal segments of the duct. Additionally, the number of vesicles, vacuoles, and multi-vesicular bodies was reduced (Fig . 4A). Furthermore, the PCs were characterized by small size, slightly heterochromatic nuclei, a relatively small Golgi apparatus (Fig. 4B), and less abundant rER and mitochondria (Fig. 4C).
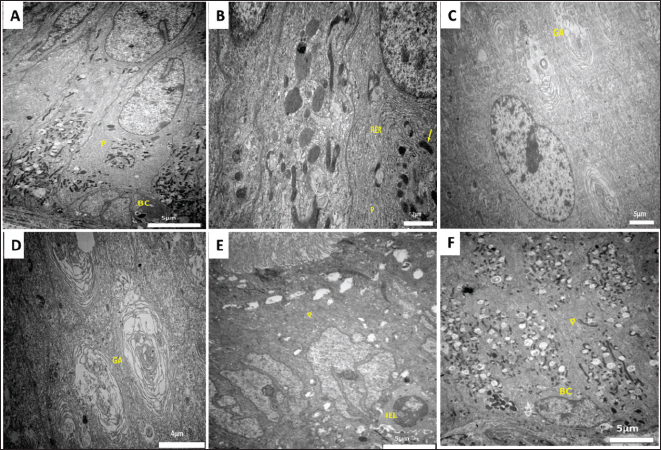
Fig. 2. Transmission electron micrograph of the epididymal duct of Meriz Bucks (autumn, October). (A) Caput (initial): principal cell (P), basal cells (BC), scale bar=5 µm. (B) Corpus (distal): principal cells (P) Rough ER (RER), scale bar=2 µm. (C) Caput (initial): Golgi apparatus (GA), scale bar=5 µm. (D) Caput (initial): Large Golgi apparatus (GA), scale bar=4 µm. (E) Cauda (proximal):Principal cells (P), intraepithelial lymphocytes (IEL), scale bar=5µm. (F) Corpus (middle): Principal cells (P) and basal cells (BC), scale bar=5µm.
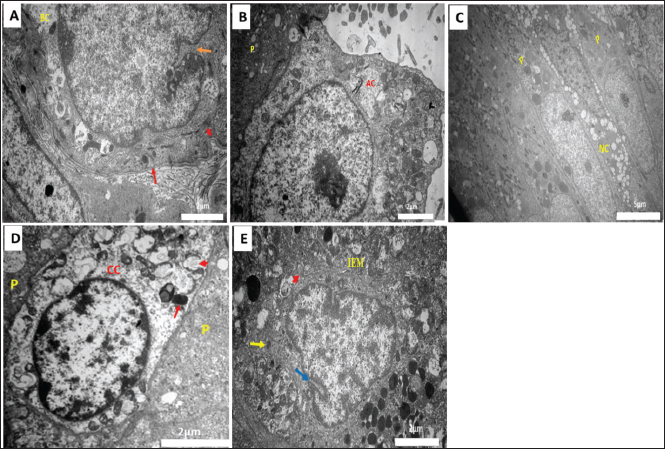
Fig. 3. Transmission electron micrographs of the epididymal duct in Meriz bucks during the autumn season (October). (A) Cauda (distal): Basal cell (BC), notch (orange arrow), and basal plasma membrane Folding (red arrowhead), and basal lamina (red arrow); scale bar=2 µm. (B) Proximal corpus: Apical cell (AC) and principal cell (P); scale bar=2 µm. (C) Caput (initial): Narrow cell (NC) and principal cells (P). Scale bar=5 µm. (D) Cauda (proximal): CC, principal cells (P), C-shaped vesicles (red arrowhead), and dense bodies (red arrow). Scale bar=2 µm. (E) Corpus (distal): IEM, notches (blue arrows), pseudopodia (red arrowhead) and lysosomes (yellow arrow); scale bar=2 µm.
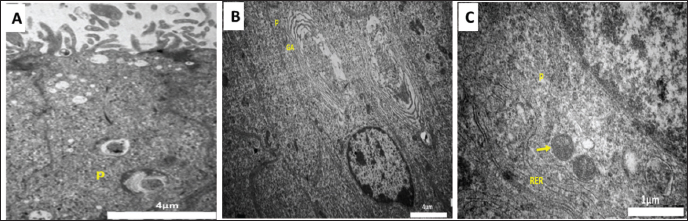
Fig. 4. Transmission electron micrograph of Meriz bucks epididymal duct (A) Winter—Middle corpus: principal cells (P); scale bar=4 µm. (B) Spring—Initial caput: principal cells (P) and Golgi apparatus (GA); scale bar=4 µm. ( C) Summer Distal corpus: a principal cell (P), mitochondria (yellow arrow), and rough endoplasmic reticulum (RER); scale bar=1 µm. Immunohistochemical findingsA-Alpha-smooth muscle actin (α-SMA)Throughout the mating and non-mating seasons. The peritubular muscular layer and smooth muscle cells surrounding blood vessels had α-SMA positivity throughout the ED, including the first segment of the caput (Fig. 5A–D). corpus segments (Fig. 6A–D) and both cauda segments (Fig. 6 E–H). During the breeding season, α-SMA expression was higher than that in the previous seasons. Nevertheless, throughout the length of the ED in every season, negative expression was observed in the epithelial cells, including PCs, BCs, ACS, CCs, IELs, and IEMs (Figs. 5 and 6).
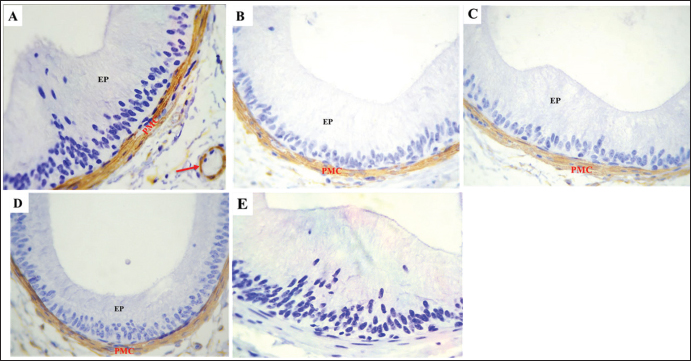
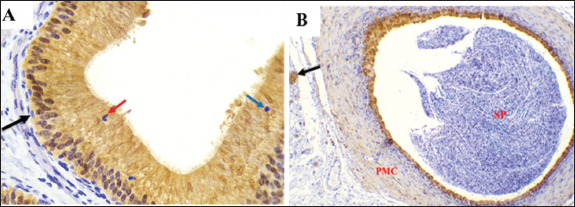
Fig .5. The expression of α-SMA in the initial segment of the Meriz Buck epididymal caput. A. Strong immunoreactivity observed in the vascular smooth cells (red arrow) peritubular muscular coat (PMC) during the mating season and in the epididymal epithelium (EP). Negative control IgG for α-SMA (E) exhibits diminished α-SMA staining in non-mating mice seasons (B–D). Magnification: 40 ×. scale bar=20 µm.
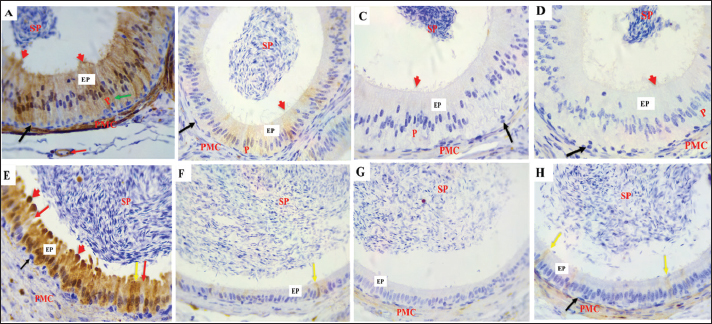
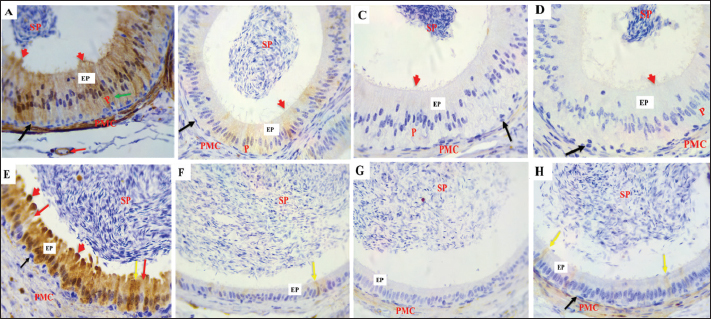
Fig .6. α-SMA expression in the epididymal intermediate (A–D) and distal (E–H) segments duct. (A) During the mating season, α- SMA is strongly expressed in VSMcells (red arrows) and the peritubular muscular coat (PMC), with a negative reaction The epididymal epithelium (EP). (B–D) α-SMA staining is less intense in the non-mating season. (E) In the DS during the mating season, strong α-SMA expression is observed in the PMC and vascular smooth muscle cells (red arrows), EP and spermatozoa (SP) show negative reactions. (F–H) The staining intensity is reduced in the non-mating season. Magnification: 40 ×; scale bar=20 µm. B- S-100The findings demonstrated that the epithelium in every ED segment showed strong immunoreactivity with S-100 during mating season (autumn) (Figs. 7A, 8A and E). All the ED segments, including the cytoplasmic and nuclear areas, exhibited high PC reactivity (Figs. 7A, 8A, and E). On the other hand, BCs continuously showed no reaction with S-100 (Figs. 7A, 8A, and E). While ACs and CCs exhibited negative reactions (Figs. 7A and 8E), NCsdisplayed a positive response (Fig. 8A). Moreover, there was a negative immune reaction in cells, such as IELs and IEMs. During non-mating seasons, staining exhibited variability ranging from mild to negative throughout all corpus segments and both cauda segments (Fig. 7B–D and 8 B–D, F–H). Moreover, differing levels of S-100, with positivity detected in the initial section of the caput. Except for PCs, all epithelial cells exhibited negative responses across the ED segments. Decreased immunological reaction of PCs in the cytoplasm and nucleus, the initial caput section was particularly noteworthy (Fig. 7 B–D). Although some mild reactions in the cytoplasm and nuclei, most PCs in the corpus exhibited a negative reaction. (Fig. 8 B–D). Similarly, most PCs in the cauda had a negative response to S-100 (Fig. 8 F–H). The IELs and IEMs also exhibited a negative reaction (Fig. 9). Stereocilia (Fig. 8A) and apical blebs (Fig. 8E) on PCs exhibited positive S-100 reactions, with a marginal increase in intensity during the mating season relative to the non-mating seasons (Fig. 8B–D). peritubular muscular coat (PMC) (Fig. 7A, 8A, 8E, 9B), the nerve trunk (Fig. 9B), and the vascular endothelium (Fig. 8A) exhibited positive S-100 with heightened intensity throughout the mating season (Fig. 7 B–D, 8B–D, F–H). Conversely, luminal spermatozoa in every segment of the epididymis exhibited negative S-100 antibodies during all seasons (Figs. 8 and 9B).
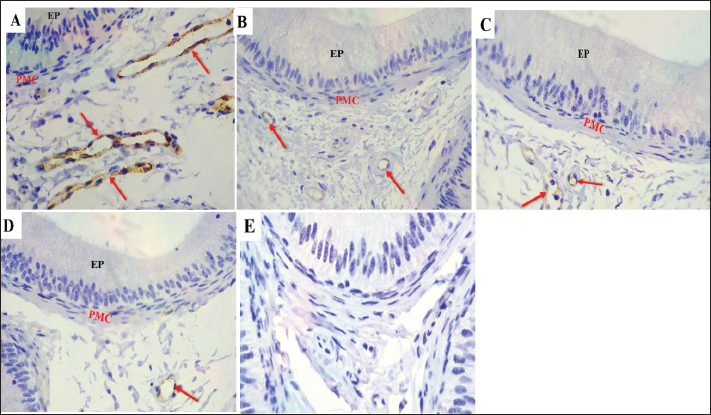
Fig. 7. S-100 expression in the first part of the caput showing A: Strong immunoreactivity in the epithelium (EP), principal cells (P), cytoplasm (red arrows), and nucleus (yellow arrows) Apical cells (green arrows), basal cells (black arrows), and the PMC During the mating season. However, weak S-100 expression was observed during non-mating seasons (B–D), Negative controls IgG (E). Magnification:40 ×, scale bar=20 µm.
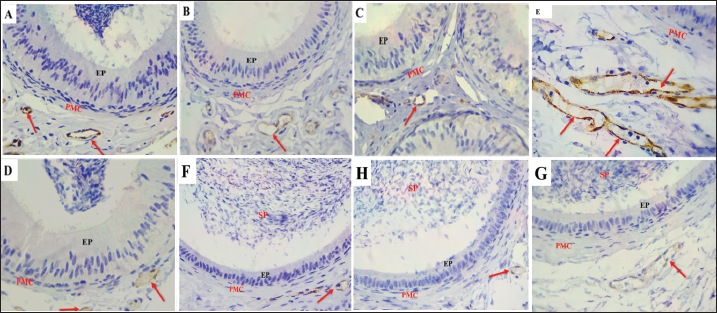
Fig. 8. S-100 protein expression in the epididymal duct of Meriz buck differs between the proximal corpus (A–D) and distal cauda (E–H) segments during mating and non-mating A: Strong immunoreactivity in principal cells (P), basal cells (black arrows), and thin cells (green arrow), the endothelium (red arrow), and the peritubular muscle coat are labelled during mating. season. The stereocilia (red arrowheads) and spermatozoa also stained positively. During non-mating Season (B–D), immunoreactivity is reduced, with only weak staining in the principal yellow arrows). Basal cells (black arrows) and transparent cells (red arrows). Distal cauda (E–H). Intense S-100 Expression in epithelial and supporting cells, including apical blebs (red arrowheads) During the mating season (E). Minimal or no staining observed in samples from the non-mating season (F–H). Spermatozoa (SP), peritubular muscle coat (PMC), and apical blebs are shown in red labelled where present. Magnification: 40x.
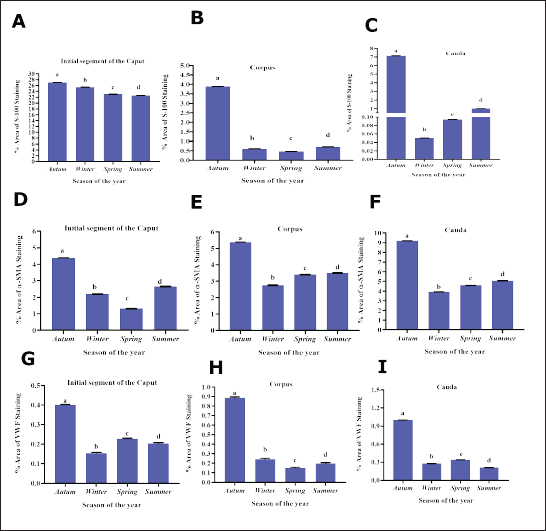
Fig. 9. S-100 expression in the epididymal duct of Meriz bucks. A: Preliminary portion of the caput during the non-mating season, exhibiting a negative reaction lymphocyte inside the epithelial cells (blue arrow), intraepithelial macrophages (red arrow), and basal cells (black arrow) Magnification: (40x). B: Distal portion of the cauda during mating season, focusing on the nerve trunk (black arrow), peritubular muscle coat (PMC), and Spermatozoa (SP)(10x). C-von willebrand factorThe results of the present investigation demonstrated that VWF immunoreactivity was confined to VE cells throughout all seasons in the Epididymal parts (Figs. 10 and 11). The staining intensity was strongest during the mating season (Figs. 10A, 11 A and D) relative to other seasons (Figs. 10 B–D, 11B–D, F–H). However, a negative expression in the epithelial cells of the ED or the peritubular muscular coat in all Epididymal segments throughout the seasons. Control IgG demonstrated negative staining for VWF (Fig. 10E). This study found that the α-SMA area was higher in autumn. In comparison to other seasons (p < 0.05, Fig. 12D). The quantification of α-SMA in the ISs of the caput was higher in autumn than in spring, summer, and winter (4.44 ± 0.02; 1.34 ± 0.02; 2.6 ± 0.02; 2.20 ± 0.02 p=0.03), respectively (Fig. 12 D–F). Measurement of the S-100 ISs of the caput demonstrated that its levels were elevated in autumn relative to those in spring, summer, and winter (27.1 ± 0.01; 23.01 ± 0.01;22.6 ± 0.01; 25.4 ± 0.01 p=0.002), respectively (Fig. 12A–C). VWF measurement of the ISs of the caput indicated that its Area was larger in autumn than in spring, summer, and winter (0.33 ± 0.09; 0.24 ± 0.09; 0.20 ± 0.09; 0.23 ± 0.09, p=0.001) (Fig. 12G–I). The quantification of α-SMA in the corpus was as follows: Higher in autumn than in spring, summer, and winter (5.50 ± 0.02; 3.45 ± 0.02; 3.51 ± 0.02; 2.76 ± 0.01, p=0.005) (Fig. 12D–F). Measurement of the S-100 ISs of the caput demonstrated that its levels were elevated in autumn relative to those in spring, summer, and Winter (3.9 ± 0.01; 0.45 ± 0.01; 0.7 ± 0.01; 0.6 ± 0.01, p=0.004) (Fig. 12A–C). The VWF measurement of the ISs of the caput indicated that its area was larger in Autumn than spring, summer, and winter (0.89 ± 0.09; 0.15 ± 0.09; 0.196 ± 0.09; 0.24 ± 0.09, p=0.007), respectively (Fig. 12G–I). The quantification of α-SMA in the cauda was higher in autumn compared to spring, summer, and winter (9.3 ± 0.01; 4.6 ± 0.01; 5.06 ± 0.01; 3.93 ± 0.01, p=0.001) (Fig. 12D–F). Measurement of the S-100 ISs of the caput demonstrated that its levels were elevated in autumn relative to those in spring, summer, and winter (7.5 ± 0.002; 0.09 ± 0.002; 1.01 ± 0.002; 0.05 ± 0.002, p=0.004), respectively (Fig. 12A–C). The VWF measurement of the ISs of the caput indicated that its area was larger in Autumn than in spring, summer, and winter (0.99 ± 0.01; 0.0.34 ± 0.01; 0.21 ± 0.01; 0.27 ± 0.01, p=0.001) (Fig. 12G–I).
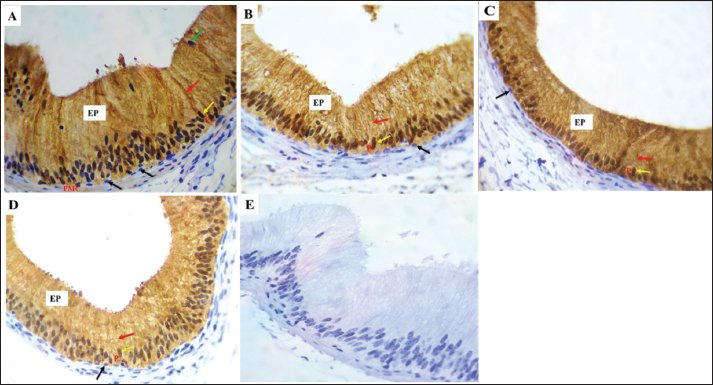
Fig. 10. VWF expression in the initial portion of caput A: Strong immunoreactivity was identified in vascular endothelial cells (red arrows) throughout the mating season staining was noted in the epithelium (EP) and peritubular muscular coat (PMC). (B–D): Weak VWF expression was detected during non-mating seasons. E: Negative control IgG. Magnification: 40X.
Fig. 11. VWF expression in the distal corpus (A–D) and distal cauda (E–H) of the epididymal duct showing: A: Strong immunoreactivity in VECs (red arrows) during mating season, the epithelium (EP), peritubular muscle coat (PMC), and spermatozoa displayed no staining (SP). VWF expression was weak during non-seasonal breeding (B–D) Strong immunoreactivity was identified in endothelial cells (red arrows) during mating. season, but no reaction was noted in the epithelium (EP) and peritubular muscle coat (PMC). Spermatozoa (SP) displayed no staining. Reduced VWF expression intensity during non-mating seasons (F–G). Magnification: 40X. Note E before D in the figures above.
Fig. 12. Quantification of S-100, α-SMA, and VWF throughout the study period. A–C Note The quantification of S-100 in the early segments of the caput, corpus, and cauda exhibited substantial differences (p < 0.05) between seasons. D–F: The quantification of α-SMA in the early section of the caput, corpus, and cauda varied significantly (p < 0.05) across the seasons. G–I: VWF expression in the early section of the caput, corpus, and cauda (p < 0.05) between seasons. Different superscript letters (a, b, c, and d) indicate statistically significant differences. Differences between group means, as determined by one-way analysis of variance followed by Tukey’s test. DiscussionThis study demonstrates that seasonal changes markedly influence the structural organization and immuno-expression of α-SMA, S-100, and VWF in the ED of Meriz buck. In seasonal breeders, such as the Meriz buck, the epididymis—responsible for sperm maturation and transport, underwent morphological and functional changes. Despite its importance, the ultrastructural and molecular characteristics of the Meriz buck epididymis remain poorly understood. Biomarkers such as α-SMA, S-100 protein, and VWF are critical for smooth muscle contractility, neural signalling, and vascular function; their seasonal expression in this species has not yet been studied. Understanding these variations is essential to reveal how reproductive efficiency adapts to environmental and seasonal changes (Tanuwiria et al., 2022; 2023). This study revealed that the PCs of the ED of Meriz bucks were the only cell type that showed considerable cytological variations among different segments. The lateral cell membranes of Adjacent PCs were sealed, forming occluding junctions throughout the year. These junctions serve as a barrier to limit the passage of molecules from the IC into the lumen of the ED (França et al., 2014), thereby maintaining an appropriate sperm-protecting milieu from the immune system (França et al., 2014; Akbarsha et al., 2015). The present results showed that the cytoplasm of PCs contained an endocytic apparatus consisting of numerous small smooth-walled and coated vesicles, variously sized vacuoles, and prominent multivesicular bodies. Comparable results were observed in the PCs of the ED in bovines (Özbek et al., 2025). These components signify a distinctive morphological characteristic of a cell type involved in activity (Schimming and Vicentini, 2008). During the non-breeding season, the number of vesicles, vacuoles, and multivesicular structures in PCs decreased. The analysis also revealed that the luminal borders of the PCs exhibited apical cytoplasmic blebs involved in the excretion of substances into the duct lumen in an apocrine mode of secretion (Abdel-Maksoud et al.,2019). The current findings showed that, during the mating season, the cytoplasm of PCs exhibited prominent organelles, including an extensive Golgi apparatus, abundant rough endoplasmic reticulum, and numerous mitochondria, in contrast to the non-mating seasons. This observation is consistent with previous reports by Ibrahim and Abdel-Maksoud (2019) and Ibrahim et al. (2021). In donkeys, Abdel-Maksoud et al. (2024) indicated that the abundance of rER and Golgi apparatus reflects active involvement in protein synthesis and secretion. Conversely, BCs of Meriz bucks exhibited a paucity of cellular organelles, which differed from the findings of Alkafafy (2005) who reported the year-round presence of organelles in bovine basal cells. Additionally, the study showed that the free border of clear cells ACs lacked microvilli, which is consistent with observations in bovines (Alkafafy, 2005) buffaloes (Alkafafy et al., 2011), but contrasting with goats (Hermo and Robaire, 2002) and camels (Ibrahim and Abdel-Maksoud, 2019), that ACs displayed a limited number of microvilli near their luminal borders. Furthermore, these cells contained numerous mitochondria surrounding the nucleus, likely to support ATP synthesis, although no seasonal ultrastructural changes. NCs were observed in the ED of Meriz buck but were conspicuously lacking in many mammalian species, including bovine (Alkafafy, 2005), buffalo (Alkafafy et al., 2011), and camel (Ibrahim and Abdel-Maksoud, 2019). This disparity may be ascribed to interspecies dissimilarity. To the best of our knowledge, this is the first study to report the presence of clear CCs in the ED of Meriz bucks, predominantly located in the proximal region of the cauda. CCs are endocytic cells that mediate the removal of proteins from the ED lumen and absorb materials derived from spermatozoa-extracted cytoplasmic droplets during transit through the duct (Hermo and Robaire, 2002; Robaire et al., 2006). Previous studies have reported the expression of S-100 and α-SMA in the epididymis of various species, highlighting their roles in sperm physiology (Czykier et al., 2010; Ijiri et al., 2011). However, seasonal variations in these proteins and VWF have not been investigated in the Meriz buck, representing a gap in the current study. Therefore, this study provides the first year-round Immunohistochemical evaluation of α-SMA, S-100, and VWF in Meriz bucks epididymis. The research identified α-SMA positivity in the peritubular muscular layer and endothelial cells across all ED segments, whereas epithelial cells exhibited α-SMA negativity. This aligns with observations from other species, such as bovines and camels. Significantly, α-SMA staining was increased during the mating season in the Meriz buck relative to the non-mating season, aligning with findings in camels (Ibrahim et al., 2017). The elevated α-SMA expression observed during the mating season indicates enhanced epididymal muscle contraction, facilitating sperm transport via the duct. The association between α-SMA and sperm structural alterations suggests its potential function in facilitating sperm maturation and transport within the epididymis, particularly during periods of increased reproductive activity. Positive S-100 immunoreactivity throughout all ED segments during the mating season, consistent with the findings in bovines, camels, and donkeys. Conversely, varying reactions were observed in the non-mating season: pronounced staining in the initial caput segment and minimal to absent staining in the posterior segment of the corpus and cauda. This pattern is consistent with observations in buffalo (Alkafafy et al., 2011), where the caput showed positive immunoreactivity, although the corpus and cauda were negative. This study identified a persistent positive expression of S-100 in the epithelium of the IS of the caput in the epididymis of Meriz bucks across all seasons. It has been reported that S-100 levels in the caput epididymis decline during the non-mating season due to decreased androgen production and decreased epididymal epithelial activity, resulting in reduced synthesis of Ca²⁺-binding proteins. Seasonal retraction of epididymal tissue and diminished sperm maturation further inhibit S-100 expression (Ibrahim et al., 2017). This aligns with the finding of Axnér (2006) who demonstrated significant absorptive activity in this area, where most proteins from the rete testis are reabsorbed (Gatti et al., 2004), leading to the epididymal fluid, predominantly including secretory products. The research identified significant S-100 protein immunoreactivity in the early segment of the caput cells of the epididymis of Meriz bucks, aligning with bovine and buffalo observations. Basal cells exhibited a negative response across the epididymis during both mating and non-mating seasons, consistent with previous studies (Alkafafy, 2005). Nonetheless, apical cells demonstrated a negative response, in contrast to previous studies in the same species. The research identified a favourable the S-100 response in the PMC exhibited, exhibiting a modest increase during the mating season, similar to observations in camels (Ibrahim et al., 2017). These alterations indicate that the S-100 and α-SMA may play a role in preserving epididymal architecture and functionality. Facilitating seasonal reproductive activity. Nonetheless, direct reproductive evaluation efficiency was not conducted in this study. Furthermore, the vascular endothelium exhibited a positive S-100 reaction, which is consistent with findings in bovines (Czykier et al., 1999; Alkafafy, 2005) and mice (Czykier et al., 2000), suggesting possible involvement in transcytosis and cellular contractility. The findings indicated a favorable S-100 response in the Meriz buck epididymal interstitium epididymal interstitium of the Meriz buck, especially within the nerve trunk. This finding is consistent with previous observations by Slater et al. (2000) who reported that S100 serves as an axon marker. Investigation revealed uniform VWF immunostaining of the endothelial cells of the Meriz buck epididymis throughout all seasons, with a marked increase in intensity during autumn. To the best of our knowledge, this is the first study to characterize VWF expression in the epididymal vasculature of Meriz bucks, suggesting active Endothelial modulation during the mating season. The findings enhance the comprehension of angiogenesis in epididymal tissue and may assist studies on reproductive physiology. In the absence of prior investigations, the research predominantly cited Vascular Endothelial Growth Factor (VEGF) as an angiogenic agent. VEGF is essential for testicular neovascularization and male germ cell production angiogenesis, and vasculogenesis to satisfy the growing germ cell metabolic requirements (Baltes et al., 2006; Reddy et al., 2012). It is expressed in Sertoli, endothelial, and Leydig cells across various species, including blackbuck and bovine, are crucial for controlling spermatogenesis and microvascular permeability in both healthy and pathological contexts. Bucks exhibited Seasonal mating patterns, with differing sexual activity according to breed and geographic area. The ideal Breeding seasons were often late summer and autumn (Delgadillo et al., 2004). Research on the Meriz deer indicated that peak reproduction rates were observed in August, followed by September (Alkass et al., 2021). Ultrastructural and immunohistochemical findings suggest that the reproductive season for Meriz bucks occurs in autumn, specifically in October. This contrasts with previous findings, perhaps attributable to the effects of global warming on mating seasonality in nations such as Iraq (Sissakian et al., 2023). Historically, mating occurred around the conclusion of summer and the onset of autumn; however, the goats can adjust to fluctuating conditions, and they prolong the mating season into October in response to these environmental alterations. ConclusionThis study demonstrates seasonal changes in the ED of Meriz bucks, which is characterized by increased cell activity and heightened expression of α-SMA, S-100, and VWF throughout the Mating season, particularly in October. These proteins likely contribute to the maintenance of Epididymal architecture, function, and sperm transport. Observed alterations in clear cells and vascular profiles provide new insights into this breed’s reproductive physiology. The findings identifying October as the peak mating season for Meriz goats, information that can guide breeding programs and support conservation. Farmers in the Kurdistan region were advised to use this breeding period to enhance progeny and mitigate the risk of extinction of this distinctive goat breed. AcknowledgmentsThe authors express sincere gratitude to the Dean and staff of the College of Veterinary Medicine, University of Duhok, and the Duhok Medical Research Center staff (DMRS) and the Animal House at the College of Veterinary Medicine, University of Duhok. The authors are grateful for the kind assistance of the Vin lab staff. The authors thank Duhok City for their kind cooperation and assistance. Special thanks are extended to their supervisors: Dr. Yahay M. Ahmed, our supervisor. and Dr. Zeravan A. Mohammed for their continuous guidance, encouragement, and invaluable advice throughout the course of this study. Conflict of interestThere are no conflicts of interest to declare. FundingThis study was funded by the authors only. Authors' contributionsYahaya A. Mohammed and Zeravan A. Mohammed contributed to the conception and study design. and the methodology of the study. Bayan S. Saady performed the sampling and laboratory tests. Both authors interpreted the statistical analyses and read the histological and immunohistochemical data. Microscopic slides plus electron microscopic results. Both authors drafted the main text. Yahya Mohammed A and Zeravan A. Mohammed supervised the study and revised the text. The authors read and approved the final version of the manuscript for publication. Data availabilityThe data supporting this study’s findings are available within the manuscript. ReferencesAbdel Maksoud, F.M., Zayed, A.E., Abdelhafez, E.A. and Hussein, M.T. 2024. Seasonal epididymis variations in donkeys (Equus asinus) with special reference to bloodEpididymal barrier. Microsc. Res. Tech. 87(2), 326–338; doi:10.1002/jemt.24436 Abdel-Maksoud, F.M., Hussein, M.T. and Attaai, A. 2019. Seasonal variation in the Intraepithelial gland in camel epididymis with special reference to autophagosomes. Microsc. Microanal. 25(4), 1052–1060; doi:10.1017/S1431927619014557 Akbarsha, M.A., Faisal, K. and Radha, A. 2015. The epididymis: structure and function. In mammalian endocrinology and male reproductive biology. Boca Raton, FL: CRC Press, p: 115; doi: 10.1201/b18900-7 Alkafafy, M. 2005. Glycohistochemical, immunohistochemical, and ultrastructural analyses of the Bovine epididymis. Ph. D. Dissertation, Faculty of Veterinary Medicine, Munich, Germany: LMU München; doi: 10.5282/edoc.3153 Alkafafy, M., Elnasharty, M., Sayed-Ahmed, A. and Abdrabou, M. 2011. Immunohistochemical studies of the epididymal duct in Egyptian water buffalo (Bubalus bubalis). Histochemical 113(2), 96–102. Alkafafy, M.E. 2024. Effects of sexual maturity and seasonality on immunolocalization of S100 and alpha smooth muscle actin in the efferent ductules of the dromadary camel (Camelus dromedarius). Int. J. Morphol. 42(5), 1347–1354. Alkass, J.E. and Merkhan, K.Y. 2013. Meriz goats in the Kurdistan region/Iraq: a review. Adv. J. Agricult. Res. 1(7), 105–111. Alkass, J.E., Hermiz, H.N., Baper, M.I., Akreyi, I.A. and Rashid, F.M. 2021. Productivity traits of local mountain goats and the factors affecting them. Iraqi J. Agricult. Sci. 52(4), 913–917; doi:10.36103/ijas.v52i4.1399 Axnér, E. 2006. Sperm maturation in domestic cats. Theriogenology 66, 14–24; doi:10.1016/j.theriogenology.2006.03.022 Aziz, K.O. 2009. Cashmere production from Maraz goats. J. Zankoy Sulaimani 12(1), 13–21; doi:10.17656/jzs.10191 Baltes, M.M., Ten Broeck, R.A., Bott, R.C., Clopton, D.T. and Cupp, A.S. 2006. Vascular endothelial growth factor (VEGF) 165 tends to increase vascular density during testis morphogenesis and may be regulated by the inhibitory isoform VEGF164b. Biol. Reprod. 69(1), 228; doi:10.1530/JOE-15-0342 Czykier, E., Sawicki, B. and Zabel, M. 1999. Immunocytochemical localization of S-100 protein in the European bison testis and epididymis. Folia Histochem. Cytobiol. 37(2), 83–84. Czykier, E., Sawicki, B. and Zabel, M. 2000. S-100 protein immunoreactivity in mammalian testis and epididymis. Folia Histochem. Cytobiol. 38(4), 163–166. Czykier, E., Zabel, M., Surdyk-Zasada, J., Lebelt, A. and Klim, B. 2010. Assessment of the S100 Protein expression in the epididymis of juvenile and adult European bison. Folia Histochem. Cytobiol. 48(3), 333–338; doi:10.2478/v10042-10-0020-4 Delgadillo, J.A., Fitz-Rodríguez, G., Duarte, G., Veliz, F.G., Carrillo, E., Flores, J.A., Vielma, J., Hernández, H. and Chemineau, P. 2004. Management of photoperiod to control reproduction in goats. Theriogenology 62(1–2), 185–197; doi: 10.1016/j.theriogenology.2003.08.005 DeRossi, R., Junqueira, A.L. and Beretta, M.P. 2005. Analgesic and systemic effects of Xylazine, lidocaine, or their combination after subarachnoid administration in goats. J. South Afr. Vet. Assoc. 76(2), a402; doi:10.4102/jsava.v76i2.402 Eubanks, D.L. 2012. Dental considerations in cows and goats. J. Vet. Dent. 29(3), 200–203. França, L.R., Auharek, S.A., Hess, R.A., Dufour, J.M. and Hinton, B.T. 2014. Blood-tissue barriers: morphofunctional and immunological aspects of the blood-testis and blood Epididymal barriers. In Biology and regulation of blood-tissue barriers. New York, NY: Springer New York, pp: 237–59; doi: 10.1007/978-1-4614-4711-5_12 Gatti, J.L., Castella, S., Dacheux, F., Ecroyd, H., Métayer, S., Thimon, V. and Dacheux, J.L. 2004. Post-testicular sperm environment and fertility. Anim. Reprod. Sci. 82, 321–339; doi:10.1016/j.anireprosci.2004.05.011 Hermo, L. and Robaire, B. 2002. Epididymal cell types and their functions. In The epididymis: from molecules to clinical practice. New York, NY: Kluwer Academic/Plenum Publishers, pp: 81–102. Ibrahim, D. and Abdel-Maksoud, F.M. 2019. Immunohistochemical and ultrastructural analysis seasonal changes in the epididymal epithelium of the camel (Camelus dromedarius). Microsc. Microanal. 25(5), 1273–1282. Ibrahim, Z., Joshi, D. and Singh, S. 2017. Seasonal immunohistochemical reactivity of S-100 and S-200 cells α-smooth muscle actin protein expression in the epididymis of dromedary camel (Camelus dromedarius). Andrologia 49(6), e12667. Ibrahim, Z.H., Al-Kheraije, K.A. and Singh, S.K. 2021. Morphological and histochemical analyses changes in the dromedary camel epididymis with respect to reproductive activity. Histol. Histopathol. 36(5), 485–504; doi:10.14670/HH-18-303 Ijiri, T.W., Merdiushev, T., Cao, W. and Gerton, G.L. 2011. Identification and validation of the Mouse sperm proteins correlated with EDM. Proteomics 11(20), 4047–4062. James, E.R., Carrell, D.T., Aston, K.I., Jenkins, T.G., Yeste, M. and Salas-Huetos, A. 2020. The role of the epididymis and the contribution of epididymosomes to the development of mammalian reproduction. Int. J. Mol. Sci. 21(15), 5377; doi:10.3390/ijms21155377 Kilkenny, C., Browne, W.J., Cuthill, I.C., Emerson, M. and Altman, D.G. 2010. Improving bioscience research reporting: the ARRIVE guidelines for reporting animal research. PLos Biol. 8(6), e1000412; doi:10.1371/journal.pbio.10004 Mohammed, Z.A. 2016. Mechanisms of fertility failure in high-yielding dairy cows thesis). University of Nottingham, Nottingham, UK. Morris, J.K. 1965. High-osmolality formaldehyde glutaraldehyde fixative for use in electron microscopy. J. Cell. Biol. 27(2), 1A–149A. Mustafa, K.N.S. and Yateem, C.A.M. 2019. Effect of supplementation at different levels effects of Fenugreek Seeds (Trigonella Foenum Graceum) on milk yield, composition, and blood parameters in the Meriz goats. Int. J. Adv. Sci. Eng. Technol. 7(4), 66–71. Mustafa, S., Heslop-Harrison, J. and Schwarzacher, T. 2022. Complete mitochondrial genome from Iraqi Meriz goats and the maternal lineage using whole-genome sequencing data. Iraqi. J. Appl. Anim. Sci. 12(2), 321–328. Özbek, M., Beyaz, F., Hitit, M., Öztop, M., Karaca, H., Ergün, E., Cabir, A. and Ergün, L. 2025. Spatiotemporal expression patterns of vascular endothelial growth factor and receptors in rat testis and epididymis throughout postnatal development. Bratislava Med. J. 126, 2203–2219; doi: 10.1007/s44411-025-00230-5 Reddy, N., Kasukurthi, K.B., Mahla, R.S., Pawar, R.M. and Goel, S. 2012. Expression of VEGF transcript and protein in the testis of several Vertebrates, including endangered species. Theriogenology 77(3), 608–614. Robaire, B., Hinton, B.T. and Orgebin-Crist, M.C. 2006. The epididymis.In Knobil and Neill’s Physiology of Reproduction Academic Press. San Diego, CA: Academic Press, pp: 1071–148. Saadi, B.S., Mohammed, Y.A. and Mohammed, Z.A. 2024. Histomorphometrical and Histochemical seasonal variations in the epididymis of Meriz in Iraq’s Kurdistan Region Egypt. J. Vet. Sci. 55(7), 1837–1855; doi:10.21608/EJVS.2024.261149.1772 Schimming, B.C. and Vicentini, C.A. 2008. Morphological features of the apical region of the principal cells of mongrel dog epididymis. Int. J. Morphol. 26, 149–153. Sissakian, V.K., Adamo, N. and Al-Ansari, N. 2023. Severe consequences of climate change in Iraq: a case study. Eng 15(4), 242–260; doi:10.4236/eng.2023.154019 Slater, M., Barden, J.A. and Murphy, C.R. 2000. Autonomic and transmitter tyrosine kinase A Receptors, but not innervation, are upregulated in the prostate of aging rats. Acta. Histochem. 102(4), 427–438. Tanuwiria, U.H., Mushawwir, A., Zain, M. and Despal, D. 2023. Impact of the protein source from legume forages on lipid regulation and growth in native ram lambs reared extensively in the south coast of West Java, Indonesia. Biodiversitas 24(7), 4183–4192; doi:10.13057/biodiv/d240763 Tanuwiria, U.H., Susilawati, I., Tasripin, D., Salman, L.B. and Mushawwir, A. 2022. Evaluation of cardiovascular biomarkers and lipid regulation in lactation Friesian Holstein at different altitude in West Java, Indonesia. HAYATI J. Biosci. 29(4), 428–434; doi: 10.4308/hjb.29.4.428-434 | ||
| How to Cite this Article |
| Pubmed Style Saadi BS, Mohammed YA, Mohammed ZA. Seasonal adaptations in the ultrastructural and immunohistochemical characterization of the epididymal duct in Meriz bucks. Open Vet. J.. 2026; 16(3): 1743-1756. doi:10.5455/OVJ.2026.v16.i3.32 Web Style Saadi BS, Mohammed YA, Mohammed ZA. Seasonal adaptations in the ultrastructural and immunohistochemical characterization of the epididymal duct in Meriz bucks. https://www.openveterinaryjournal.com/?mno=282926 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.32 AMA (American Medical Association) Style Saadi BS, Mohammed YA, Mohammed ZA. Seasonal adaptations in the ultrastructural and immunohistochemical characterization of the epididymal duct in Meriz bucks. Open Vet. J.. 2026; 16(3): 1743-1756. doi:10.5455/OVJ.2026.v16.i3.32 Vancouver/ICMJE Style Saadi BS, Mohammed YA, Mohammed ZA. Seasonal adaptations in the ultrastructural and immunohistochemical characterization of the epididymal duct in Meriz bucks. Open Vet. J.. (2026), [cited March 31, 2026]; 16(3): 1743-1756. doi:10.5455/OVJ.2026.v16.i3.32 Harvard Style Saadi, B. S., Mohammed, . Y. A. & Mohammed, . Z. A. (2026) Seasonal adaptations in the ultrastructural and immunohistochemical characterization of the epididymal duct in Meriz bucks. Open Vet. J., 16 (3), 1743-1756. doi:10.5455/OVJ.2026.v16.i3.32 Turabian Style Saadi, Bayan S., Yahya A. Mohammed, and Zeravan A. Mohammed. 2026. Seasonal adaptations in the ultrastructural and immunohistochemical characterization of the epididymal duct in Meriz bucks. Open Veterinary Journal, 16 (3), 1743-1756. doi:10.5455/OVJ.2026.v16.i3.32 Chicago Style Saadi, Bayan S., Yahya A. Mohammed, and Zeravan A. Mohammed. "Seasonal adaptations in the ultrastructural and immunohistochemical characterization of the epididymal duct in Meriz bucks." Open Veterinary Journal 16 (2026), 1743-1756. doi:10.5455/OVJ.2026.v16.i3.32 MLA (The Modern Language Association) Style Saadi, Bayan S., Yahya A. Mohammed, and Zeravan A. Mohammed. "Seasonal adaptations in the ultrastructural and immunohistochemical characterization of the epididymal duct in Meriz bucks." Open Veterinary Journal 16.3 (2026), 1743-1756. Print. doi:10.5455/OVJ.2026.v16.i3.32 APA (American Psychological Association) Style Saadi, B. S., Mohammed, . Y. A. & Mohammed, . Z. A. (2026) Seasonal adaptations in the ultrastructural and immunohistochemical characterization of the epididymal duct in Meriz bucks. Open Veterinary Journal, 16 (3), 1743-1756. doi:10.5455/OVJ.2026.v16.i3.32 |