| Research Article | ||
Open Vet. J.. 2026; 16(1): 503-509 Open Veterinary Journal, (2026), Vol. 16(1): 503-509 Research Article Investigation of the oculo-head form of malignant catarrhal fever in Basrah Buffaloes in IraqAli Yousif Atshan and Kamal M. Alsaad*Department of Internal and Preventive Medicine, College of Veterinary Medicine, University of Basrah, Basrah, Iraq *Corresponding Author: Kamal Mahalhul Alsaad. Department of Internal and Preventive Medicine, College of Veterinary Medicine, University of Basrah, Basrah, Iraq. Email: kamalsad58 [at] yahoo.com Submitted: 11/09/2024 Revised: 02/12/2025 Accepted: 16/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
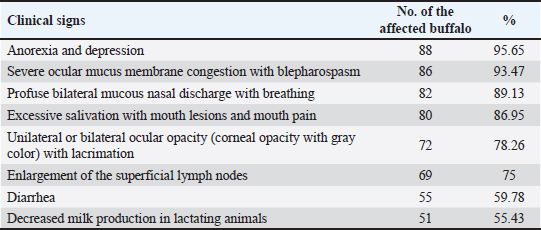
AbstractBackground: Malignant catarrhal fever (MCF) is a severe infectious disease that can be fatal and infects both domestic and wild animals, including cows, buffalo, deer, bison, exotic ruminants, and even pigs. Aim: MCF is suspected in Buffalo, Basrah, Iraq. Therefore, a clinical, hematological, biochemical, and diagnostic study was conducted. Methods: The study was conducted on 163 local Iraqi buffalo breeds, aged 2–6 years, of both sexes, showing signs suggestive of MCF. The control group comprised 25 clinically healthy local buffalo breeds. The disease was diagnosed based on clinical manifestations and confirmed by serodiagnosis using an indirect ELISA test. Results: The results indicated that 92% (56.44%) of the animals were positive for MCF. Moreover, the infected buffalo showed an acute form of the disease with anorexia and depression, severe congestion of ocular mucus membranes with blepharospasm, profuse bilateral mucus nasal discharge with mouth breathing, excessive rope salivation with mouth lesions and mouth pain, unilateral or bilateral ocular opacity with lacrimation, enlargement of superficial lymph nodes, diarrhea, and low milk yield, in addition to a significant increase in vital signs. Moreover, leukocytosis due to lymphocytosis, along with a significant difference in clotting factor indices, was encountered. In addition, hypoglycemia, decreased total protein count, and increased blood urea nitrogen, creatine kinase, and Alkaline phosphatase were encountered in MCF animals compared with controls. Conclusion: MCF is endemic and continues to spread among Iraqi animal fields, where the seroprevalence is high (56.44%). Therefore, strict control measures, including regular annual vaccination, are highly recommended. Keywords: Malignant catarrhal fever, Buffalo, ELISA, Iraq. IntroductionMalignant catarrhal fever (MCF) is a severe infectious disease that can be fatal, infecting both domestic and wild animals, including cows, buffalo, deer, bison, exotic ruminants, and even pigs (Constable et al., 2017; Orono et al., 2019). The disease is caused by 10 different viruses belonging to the Rhadinovirus genus (of the family Herpesviridae). Two of these viruses are carried by sheep, whereas the other five have been found in sheep (Dettwiler et al., 2011; Headley et al., 2015). The disease exists on most continents, including America, Africa, and Asia, affecting both domestic and zoo animals with various synonyms, such as Malignant Head Catarrh, Malignant Catarrh, Catarrhal Fever, Snotsiekt, and Gangrenous coryza (Russell et al., 2009; Constable et al., 2017). Two important and different groups of causative viruses were identified: the Alcelaphinae–Hippotraginae group and the Caprinae group. Alcelaphine herpesvirus 1 is endemic globally in most wild animals and is responsible for disease in these species. In addition, the ovine type of herpesvirus 2 was endemic in sheep. The caprine type of herpesvirus 2 infects goats and is responsible for in utero infection (Dewals and Vanderplasschen, 2011; Li et al., 2014; Cunha et al., 2019). The disease can be transmitted directly between animals. Close contact is important, in addition to in utero type transmission (Li et al., 2004; van Wuijckhuise-Sjouke and Knibbe, 2007). (Constable et al., 2017) added the possibility of transmission through semen in rams. Furthermore, more prevalent transmission can occur when sheep and cattle are cohoused or mixed, indicating the presence of both susceptible and carrier animals. Cattle of all ages were susceptible and could become infected with the disease. However, a high morbidity rate was registered in older adults (Sharma et al., 2019). The disease characterized by different clinical manifestations, where the peracute form are important and could be prevalent and common form manifested with high fever, rapid death mostly with (12–24 hours) which might preceded sometimes with, weakness and depression as well as diarrhea which always mixed with blood (Constable et al., 2017), Nevertheless, when the animal survive and pass the peracute form was show acute form with a clinical signs including, anorexia and the animal will show mostly bilateral corneal problems (corneal opacity) which started from corneal-scleral junction and developed more to be in side and affected all ocular tissues, diseased animal will have ocular mucus discharge which could developed to muco-purulent then purulent, Moreover, the muzzle of diseased animal and its nares tissue will become encrusted, However, diseased animal will show, open mouth breathing with dyspnea as well as salivation due to stomatitis characterized by hyperemic oral mucosa with necrotic foci (Sood et al., 2017). MCF has been reported in Iraq (Khudhair et al., 2019). However, it is suspected that the disease still exists and is spreading in this Iraqi environment and was suspected in Buffalo of Basrah, Iraq. Therefore, a clinical, hematological, biochemical, and diagnostic evaluation was conducted. Materials and MethodsArea of study and diagnosis by ELISA testThe study was conducted in the northern part of Basrah, Iraq, on (163) local Iraqi buffalo breeds (2–6 years old), both sexes, males, and females. Suspected animals showing unilateral and/or bilateral ocular opacity, salivation, anorexia, and high fever. Moreover, 25 clinically normal native breeds of buffalo are considered as a control group. The study was conducted from January to May 2025. Complete clinical examinations were performed on suspected and control buffalo. Blood smears and fecal samples were examined to rule out blood and endoparasitic infections. A serodiagnosis using the Bovine Anti-malignant Catarrhal Fever Ab (MCF-Ab) ELISA Kit from Sulong Biotech Company Ltd., China, was employed to confirm the presence of the qualitative MCF-Ab in buffalo, according to the manufacturer’s instructions. (Batch number SL0202Bo. Lot number is 20230328). Samples for hematological and biochemical analysis10 ml of blood was drained from the jugular vein of each diseased and control healthy buffalo, and 2.5 ml of blood was mixed with ethylenediaminetetraacetic acid for a complete blood picture and total platelet count using a GENX blood analyzer (USA). Differential leukocyte count was performed according to Weiss and Wardrop (2011) using blood smears stained with Giemsa. Furthermore, blood clotting factors were calculated according to Harvey (2012) using plasma mixed with Trisodium citrate. Kits from BIOLABO (FRANCE) are used for spectrophotometric evaluation of glucose, total protein, blood urea nitrogen (BUN), creatine kinase (CK), and alkaline phosphatase (ALP). Statistical analysisA student’s t-test of SPSS (version 16) was used to compare the diseased buffalo and the healthy control animals. Furthermore, the percentage values were also extracted, according to Leech et al. (2013). Moreover, the significance level was set at (p < 0.05). Ethical approvalAll ethical principles are considered in this article. However, the animal ethics committee provided their approval and permission to conduct this research, as stated in the official document by The College of Veterinary Medicine. VET 93/37/2025, issued by the College of Veterinary Medicine, University of Basrah, Iraq. ResultsIn the current study out of 163 examined animals (using serum) by ELISA, (92) (56.44%) were found positive for MCF (Table 1), Moreover, all those infected buffalo showed an acute form of the disease with clinical signs (Some of them were very prominent and indicative of the disease(, such as anorexia and depression (95.65%), severe congestion of ocular mucus membranes with blepharospasm (93.47%), profuse bilateral mucous nasal discharge with mouth breathing which seen in (89.13%), excessive rope salivation with mouth lesions and mouth pain (86.95%), unilateral or bilateral ocular opacity (corneal opacity with gray color, Fig. 1), with lacrimation (78.26). Moreover, superficial lymph nodes were seen enlarged in (75%) of diseased buffalo. In addition, (59.78%) of diseased buffalo were suffering from diarrhea mixed with mucus sometimes. Furthermore, milk production was decreased in 55.43% of diseased lactating animals (Table 2).
Fig . 1. Unilateral ocular opacity (grayish corneal opacity) in a buffalo infected with MCF. Table 1. Detection of specific antibodies against MCF using indirect enzyme-linked immunosorbent assay.
Table 2. Clinical signs of MCF-affected buffalo.
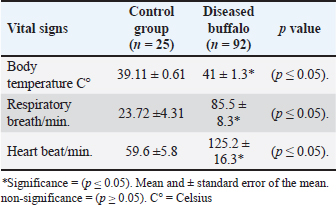
Moreover, diseased buffalo showed a significant rise (p < 0.05) in vital signs, as body temperature (41 ± 1.3), respiratory rate (85.5 ± 8.3), and heart rate (125.2 ± 16.3) rose significantly in infected animals compared to the control group (39.11 ± 0.61; 23.72 ± 4.31; 59.6 ± 5.8), respectively (Table 3). Table 3. The vital signs of MCF-infected buffalo and control group.
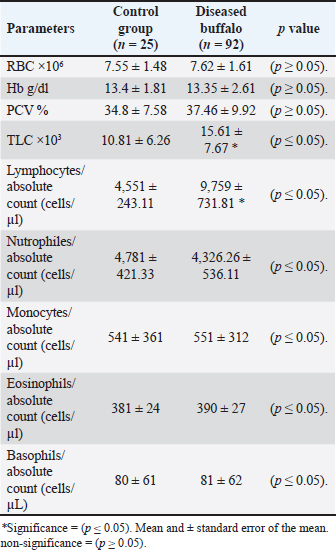
Concerning the hematological analysis of diseased buffalo compared with the control healthy buffalo, results indicate a significant increase in total leukocyte count (TLC) due to a significant rise in lymphocyte number in diseased buffalo than in controls (Table 4). Table 4. Hematological parameters of buffaloes infected with MCF compared with the control group.
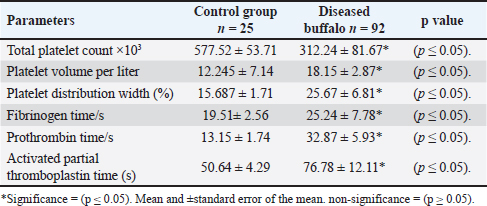
Furthermore, the indices of clotting factors of diseased buffalo indicate decreased values of the total platelet count compared with the healthy control buffalo. On the other hand, high values are indicated in the volume of platelets, distribution width of platelets, both fibrinogen and prothrombin time, as well as the activated partial thromboplastin time in diseased buffalo than in control animals (Table 5). Table 5. The indices of clotting factors of buffaloes with MCF and the control group.
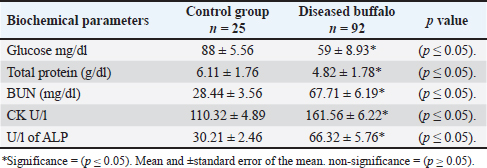
On the other hand, biochemical analysis of diseased buffalo with MCF and control healthy animals showed a decrease in the amount of glucose and total protein in diseased buffalo with MCF than in controls. Furthermore, BUN, CK, and ALP levels were higher in diseased buffalo than in healthy control buffalo (Table 6). Table 6. Biochemical parameters of buffaloes infected with MCF compared with the control group.
DiscussionTo differentiate MCF from rinderpest and FMD, it was previously called Kopfkarankheit, especially in Europe (Zaki et al., 2016). MCF is a sporadic and non-contagious illness that affects most domestic and wild animals. Furthermore, the presence of a reservoir for the disease, such as sheep and goats, makes the disease more dangerous, virulent, and widespread (Khudhair et al., 2019; Kumar et al., 2021). The disease can be diagnosed first based on the characteristic clinical manifestations mentioned in the current study, and also compatible with others (Smith, 2004; Swai et al., 2013; Headley et al., 2015; Constable et al., 2017), who indicated the disease in Iraq and other places with different infection rates. Moreover, the diagnosis was confirmed by ELISA (The indirect type of ELISA), where the test used a monoclonal antibody that largely preserved the epitope of the MCF causative agent applied to the explored of antibodies, which considered as an available technique for epidemiological investigations. The appearance of the specific antibodies could be deleted in some diseased animals. Therefore, occasionally, serological detection of the specific antibodies related to MCF could be uncertain for determining the infection when the suspected animal is less than 1 year old. However, non-infected healthy lambs and also kids could test positive because of their maternal antibodies (Li et al., 2001; Fraser et al., 2006; Sood et al., 2017). MCF is a hard, fatal disease with adverse pathogenic effects (multisystem disease) on all body tissues of the infected animal, characterized by lymphoid proliferation with obvious vascular epithelial lesions and keratoconjunctivitis. However, the enlargement of lymph nodes in the diseased animals might have resulted from the clear proliferation of sinusoidal cells (Constable et al., 2017). Furthermore, vasculitis was also registered, especially in cerebromeningeal changes, reflecting encephalitis. Furthermore, O’Toole and Li (2014)added that tibio-tarsal joint involvement was also present in rare cases, resulting in synovitis. However, in general, it was thought that all those pathological changes reflected in the animal body were a result of the direct effect of the virus interactions or could be an immunological response (type of immune-mediated response) targeted against the infected cells (Constable et al., 2017; Sood et al., 2017). It was mentioned that in MCF, necrosis of the anterior nasal mucosa was evident, especially on buccal mucus membranes. In addition, different degrees of necrosis could also be obvious on the gums, palate, and gingival membranes, reflecting a painful mouth with a careful movement of the jaws and smacking sometimes, which is the main reason for off food. Moreover, erosive stomatitis was diffuse, leading to ropy salivation (Russell et al., 2009; Li et al., 2014). In this study, the acute head and eye form was diagnosed based on the predominant clinical manifestations of diseased buffalo, which helps in differentiating this disease from others, such as bluetongue, mucosal disease, and Rinderpest. Moreover, the mentioned clinical signs in the current study, such as severe congestion of mucus membranes with blepharospasm and unilateral or bilateral ocular opacity (corneal opacity with gray color) with lacrimation, refer to the head and eye form of MCF, as also mentioned by (Constable et al., 2017; Orono et al., 2019). Results of hematological analysis indicate significant leucocytosis due to lymphocytosis in infected buffalo compared with healthy control buffalo. Some viral diseases can affect (directly or indirectly) clotting factor indices. Nevertheless, in the present study, an obvious change has been reported in those values. However, this effect could mostly end with disseminated intravascular coagulopathy agreed results as indicated also by (Erkılıç et al. (2017) and Uzlu et al. (2023). Increased leukocyte count (leucocytosis) due to significant lymphocytosis in infected buffalo might reflect the high activity and stimulation of the immune system of diseased animals, as well as increase the capabilities and the efficacy of the bone marrow for the production of regenerated cells to activate the phagocytosis mechanism (O’Toole and Li, 2014; Constable et al., 2017). In addition, the MCF virus could affect the white blood system, where lymphocytosis in MCF in cattle is caused by viral proteins that induce the survival and multiplication of latently infected lymphocytes, primarily CD8+ T cells, and potentially reprogram them into activated, proliferating cells. The results concerning the disturbance of clotting factor indices agreed with the results obtained by (Bick, 2003; Rebar et al., 2005; Muszbek et al., 2008), where this mechanism is related to either increased coagulation (Hyper-coagulation) or decreased coagulation (Hypo-coagulation). This mechanism depends on the rate of platelet aggregation. Moreover, the disturbance of coagulopathy related to viral infection might be related to abnormal blood viscosity and the possibility of causing vascular inflammation due to the causative virus (Constable et al., 2017). The biochemical results of the current study were also consistent with the results established by Smith (2004), Constable et al. (2017), and Sood et al. (2017), who concluded that MCF has different effects on body tissues, which could alter most biochemical profiles of diseased animals. Hypoglycemia, which is indicated in diseased buffalo with MCF, could be seen when low food consumption is indicated as a reflection of low animal energy, resulting in inadequate ruminal production of digestive acids (mainly propionate), which is considered the main source of glucose precursor in ruminants, causing hypoglycemia (Uzlu et al., 2023). Moreover, anorexia and/or dehydration due to loss of body fluids and electrolytes play a good role (Smith, 2004). As indicated in the current study, decreased total protein values could occur due to pyrexia, which results in protein desolation combined with limited protein intake because of anorexia and intestinal absorption defects. However, decreased hepatic protein synthesis, such as albumin, also plays a role (Sood et al., 2017). On the other hand, dehydration, increased body temperature, and viral multiplication might predispose to circulatory irregularities, which could contribute to inducing renal ischemia with insufficient perfusion of renal tissues, reflected by an increase in BUN values in diseased buffalo (Constable et al., 2017). The ALP level could reflect hepatic damage. Nonetheless, it might also be considered a useful marker for bone diseases and renal tissue damage, where it increases when those tissues are affected (Kaneko, 2014). On the other hand, increased values of CK indicate heart, skeletal, and brain tissue damage, as well as dehydration, which was observed in the present study due to the multiple adverse effects of the MCF virus on body tissues (Constable et al., 2017; Uzlu et al., 2023). MCF is a systemic condition that may impair the functioning of all body organs, reflecting obvious alterations in biochemical profile. AcknowledgmentsThe authors extend their gratitude to the College of Veterinary Medicine of the University of Basrah, Iraq. Conflict of interestAll authors agree and confirm that there are no conflicts of interest in this study. FundingThe research expenses were fully funded by the College of Veterinary Medicine, University of Basrah, Iraq. Authors’ contributionsThe authors are reviewed, discussed, and agree on their individual contributions before and during the submission of their article. The first author, Ali Y. Atshan, was responsible for collecting and analyzing the laboratory samples. Alsaad K.M., the second author, conducted the statistical analysis and wrote the entire article. Data availabilityAll the data supporting the findings of this study are available within the manuscript. ReferencesBick, R.L. 2003. Disseminated intravascular coagulation: current etiology, pathophysiology, diagnosis, and treatment concepts. Hematol. Oncol. Clin. North. Am. 17, 149–176. Constable, P.D., Hinchcliff, K.W., Done, S.H. and Grunberg, W. 2017. Veterinary medicine. a textbook of the diseases of cattle, sheep, goats and horses, 11th ed. Louise, Missouri, USA: W.B. Saunders Co. Cunha, C.W., Slater, O.M., Macbeth, B., Duignan, P.J., Warren, A., Highland, M.A. and Li, H. 2019. Domestic and bighorn sheep carry distinct gamma herpes viruses belonging to the genus Macavirus. Virus. Res. 2007, 197729. Dettwiler, M., Stahel, A., Krüger, S., Gerspach, C., Braun, U., Engels, M. and Hilbe, M. 2011. A possible case of caprine-associated malignant catarrhal fever in a domestic water buffalo (Bubalus bubalis) in Switzerland. BMC. Vet. Res. 7(78), 1–6. Dewals, B.G. and Vanderplasschen, A. 2011. Malignant catarrhal fever, induced by alcelaphine herpesvirus 1, is characterized by an expansion of activated CD3+CD8+CD4+ T cells expressing a cytotoxic phenotype in both lymphoid and non-lymphoid tissues. Res. 42(1): 95. Erkılıç, E.E., Öğün, M., Kırmızıgül, A.H., Adalı, Y., Ermutlu, C., Eroğlu, H.A., Kükürt, A., Çitil, M. and Uzlu, E. 2017. Determination of oxidative stress and inflammation markers in serum, blood, and cerebrospinal fluid in cattle with head-eye malignant catarrhal fever. Kafkas. Univ. Vet. Fak. Derg. 23(4), 515–519. Fraser, S.J., Nettleton, P.F., Dutia, B.M., Haig, D.M. and Russell, G.C. 2006. “Development of an enzyme-linked immunosorbent assay for the detection of antibodies against malignant catarrhal fever viruses in cattle serum”. Vet. Microbiol. 116(1–3), 21. Harvey, J.W. 2012. Veterinary hematology: a diagnostic guide and color Atlas Elsevier Inc. USA: Elsevier Inc.. Headley, S.A., Pimentel, L.A., Oliveira, V.H., Toma, H.S., Alfieri, A.F., Carvalho, A.M., Dos Santos, M.D. and Alfieri, A.A. 2015. Transplacental transmission of ovine herpesvirus 2 in cattle with sheep-associated malignant catarrhal fever. Pathol 153(4), 206. Kaneko, J.J. 2014. Clinical biochemistry of domestic animals, 3rd ed. New york, USA: Academic Press. Khudhair, Y.I., Ayyez, H.N. and Hussain, M.H. 2019. Phylogenetic analysis of ovine herpes virus-2 (OHV-2) in cattle infected with malignant catarrhal fever in the governorate of Al-Qadisiyah, Iraq. Iraqi. J. Vet. Sci. 33, 51–58. Kumar, N., Sood, R., Pateriya, A.K., Venkatesakumar, E., Ramprabhu, R., Dixit, R., Bhatia, S. and Singh, V.P. 2021. First molecular evidence and genetic characterization of ovine herpesvirus 2 in multiple animal species in India. J. Biol. Chem. Chem. 8, 1–8. Leech, N., Barrett, K. and Morgan, G.A. 2013. Statistical Package for the Social Sciences for intermediate statistics: use and interpretation. Routledge. Li, H., Cunha, C.W., Taus, N.S. and Knowles, D.P. 2014. Malignant Catarrhal fever: ‘inching toward understanding’. Ann. Rev. Anim. Biosci. 2(1), 209. Li, H., Mcguire, T.C., Müller-Doblies, U.U. and Crawford, T.B. 2001. A simpler, more sensitive competitive inhibition enzyme-linked immunosorbent assay for detection of antibody to malignant catarrhal fever viruses. J. Vet. Diag. Invest. 13(4), 361–364. Li, H., Taus, N.S., Lewis, G.S., Kim, O., Traul, D.L. and Crawford, T.B. 2004. Shedding of ovine herpesvirus 2 in sheep nasal secretions: the predominant mode for transmission. J. Clinic. Microbiolo. 42, 5558–5564. Muszbek, L., Bagoly, Z., Bereczky, Z. and Katona, E. 2008. The involvement of blood coagulation factor XIII in fibrinolysis and thrombosis. Cardiovasc. Hematol. Agents. Med. Chem. 6(3), 190–205. O’Toole, D. and Li, H. 2014. The pathology of malignant catarrhal fever, with an emphasis on ovine herpesvirus 2. Vet. Pathol. 51(2), 437–452. Orono, S.A., Gitao, G.C., Mpatswenumugabo, J.P., Chepkwony, M., Mutisya, C., Okoth, E., Bronsvoort, B.M.C., Russell, G.C., Nene, V. and Cook, E.A.J. 2019. Field validation of the clinical and laboratory diagnosis of malignant catarrhal fever associated with wildebeest in cattle. BMC Vet. Res. 15, 69. Rebar, A.H., Mas Williams, P.S., Feldman, B.F., Metzger, F.L. and Pollock, R.V. 2005. Platelets: overview, morphology, quantity, platelet function disorders. Int. Vet. Infec. 21, 805–825. Russell, G.C., Stewart, J.P. and Haig, D.M. 2009. Malignant catarrhal fever: a review. Vet. J. 179, 324–335. Sharma, B., Parul, S., Basak, G. and Mishra, R. 2019. Malignant catarrhal fever (MCF): an emerging threat. J. Entomol. Zool. Stud. 7, 26–32. Smith, B.P. 2004. Large animal internal medicine, 4th ed., New York, NY: Mosby.. Sood, R., Kumar, N. and Bhatia, S. 2017. Malignant catarrhal fever. In Emerging and re-emerging infectious diseases in livestock. Ed., Bayry, J. Berlin/Heidelberg, Germany: Springer International Publishing. Swai, E.S., Kapaga, A.M., Sudi, F., Loomu, P.M. and Joshua, G. 2013. Malignant catarrhal fever in pastoral Maasai herds caused by wildebeest-associated alcelaphine herpesvirus-1: an outbreak report. Vet. Res. Forum. 4(2):133–136. Uzlu, E., Erkiliç, E.E., Adali, Y., Öğün, M., Can Şahna, K., Eryeğen, N., Eroğlu, H.A., Abayli, H., Ermutlu, C. and Kirmizigül, A.H. 2023. Blood and cerebrospinal fluid biochemistry, cytology, and hematological parameters in the head-and-eye form of malignant catarrhal fever in cattle. Erciyes. Univ. Vet. Fak. Derg. 20(2), 86–93. Van Wuijckhuise-sjouke, L. and Knibbe, G.C. 2007. Malignant catarrhal fever in cattle: a large outbreak. Tijdschr. Geneeskd. 132(19), 732–734. Weiss, D.J. and Wardrop, K.J. 2011. Schalm’s veterinary hematology. New York, NY: John Wiley & Sons. Zaki, A.A.M., El-Said, H.M., Abd El-aziz, A., Bastawecy, I.M., Abd El-wahab, S.A. and El-Sayed, M.M. 2016. Field study on malignant catarrhal fever. Life. Sci. 13, 83–98. | ||
| How to Cite this Article |
| Pubmed Style Atshan AY, Alsaad KM. Investigation of the oculo-head form of malignant catarrhal fever in Basrah Buffaloes in Iraq. doi:10.5455/OVJ.2026.v16.i1.47 Web Style Atshan AY, Alsaad KM. Investigation of the oculo-head form of malignant catarrhal fever in Basrah Buffaloes in Iraq. https://www.openveterinaryjournal.com/?mno=283247 [Access: February 01, 2026]. doi:10.5455/OVJ.2026.v16.i1.47 AMA (American Medical Association) Style Atshan AY, Alsaad KM. Investigation of the oculo-head form of malignant catarrhal fever in Basrah Buffaloes in Iraq. doi:10.5455/OVJ.2026.v16.i1.47 Vancouver/ICMJE Style Atshan AY, Alsaad KM. Investigation of the oculo-head form of malignant catarrhal fever in Basrah Buffaloes in Iraq. doi:10.5455/OVJ.2026.v16.i1.47 Harvard Style Atshan, A. Y. & Alsaad, . K. M. (2026) Investigation of the oculo-head form of malignant catarrhal fever in Basrah Buffaloes in Iraq. doi:10.5455/OVJ.2026.v16.i1.47 Turabian Style Atshan, Ali Yousif, and Kamal M. Alsaad. 2026. Investigation of the oculo-head form of malignant catarrhal fever in Basrah Buffaloes in Iraq. doi:10.5455/OVJ.2026.v16.i1.47 Chicago Style Atshan, Ali Yousif, and Kamal M. Alsaad. "Investigation of the oculo-head form of malignant catarrhal fever in Basrah Buffaloes in Iraq." doi:10.5455/OVJ.2026.v16.i1.47 MLA (The Modern Language Association) Style Atshan, Ali Yousif, and Kamal M. Alsaad. "Investigation of the oculo-head form of malignant catarrhal fever in Basrah Buffaloes in Iraq." doi:10.5455/OVJ.2026.v16.i1.47 APA (American Psychological Association) Style Atshan, A. Y. & Alsaad, . K. M. (2026) Investigation of the oculo-head form of malignant catarrhal fever in Basrah Buffaloes in Iraq. doi:10.5455/OVJ.2026.v16.i1.47 |