| Research Article | ||
Open Vet. J.. 2026; 16(1): 515-520 Open Veterinary Journal, (2026), Vol. 16(1): 515-520 Research Article Molecular and conventional detection of Salmonella spp. and Helicobacter pylori in licensed and individually prepared foods in the city of Al-Kut, IraqAkmam Ali Habeeb*Department of Biology, College of Education for Pure Sciences, University of Wasit, Al Kut City, Iraq *Corresponding Author: Akmam Ali Habeeb. Department of Biology, College of Education for Pure Sciences, Submitted: 14/09/2025 Revised: 10/12/2025 Accepted: 22/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
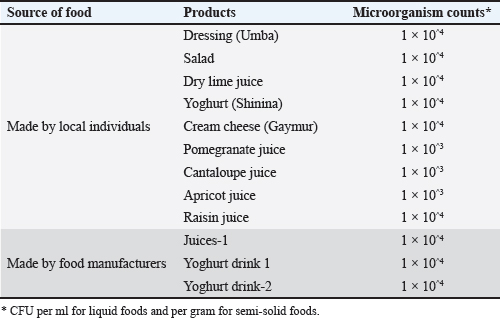
AbstractBackground: A wide range of licensed foods (LFs) and foods prepared by individuals (FPIs) in local stores and restaurants have been frequently blamed for food poisoning in many consumers. To the best of our knowledge, no data are available on the status of contamination in these foods. Aim: This study was conducted to estimate the contamination levels and detect some bacterial pathogens in different food sources prepared by individuals in local stores and restaurants in Al-Kut City, Iraq. Methods: Food samples were subjected to microscopic examination and counting. These samples were also examined for the presence of Salmonella spp. and Helicobacter pylori using Quantitative Reverse Transcription Polymerase Chain Reaction. The ureC gene of H. pylori was detected by quantitative reverse transcription polymerase chain reaction using published primers. Deoxyribonucleic Acid was extracted using a commercial kit and amplified using SYBR Green (a DNA-binding fluorescent dye) Green chemistry. Results: The results revealed high bacterial and fungal contamination as discovered by the microscopic method. Furthermore, these samples showed a significant (p < 0.05) presence of Salmonella spp. in many food samples at different rates ranging from 76% to 80%, especially in FPIs. In addition, these samples showed a significant (p < 0.05) presence of H. pylori, as it ranged from 12% to 17% of the FPIs. The ureC gene of H. pylori was detected in 16% of the food samples by quantitative polymerase chain reaction. The highest positivity was recorded in cheese (22%) and cream cheese (18%), while lower rates were found in juices (10%) and yogurt (12%). The difference between cheese products and juices was statistically significant (p < 0.05). These findings confirm that unprocessed dairy products are more contaminated with H. pylori ureC than other foods. Conclusion: The results suggest that high levels of contamination in different food samples need control procedures for better protection against food-borne diseases. Keywords: Food-borne diseases, Food poisoning, Helicobacter pylori, QPCR, Salmonella spp. IntroductionFoodborne pathogens, such as bacteria, can induce foodborne diseases (FBDs). A FBD is defined as the manifestation of comparable symptoms that arise from the consumption of a shared food source (Switaj et al., 2015; Wu et al., 2018; Todd, 2020). A foodborne illness is a condition that arises when a causative agent is consumed through food and successfully colonizes within the human host, often leading to replication. Alternatively, foodborne illness can occur when a toxigenic pathogen in a food item generates its toxins. FBD is commonly categorized as either foodborne infection (FBRI) or foodborne poisoning (FBRP). In the context of FBRIs, the duration between ingestion and the manifestation of symptoms is typically longer than that of FBRP, primarily due to the presence of an incubation period. Over 200 distinct FBDs have been discovered. The most severe instances of the condition are commonly observed in individuals who are advanced in age, in individuals who are in their early stages of life, in individuals with compromised immune system functionality, and in individuals who are in good health but have been exposed to a significantly elevated dosage of a particular organism (Pires et al., 2021; Menini et al., 2022; Mengistu et al., 2022). In 2015, more than 4000 FBDs were reported by 26 member states within the EU. The outbreaks resulted in a total of 45,874 reported cases of illness, which is 209 more than the previous year. Additionally, there were 3,892 hospitalizations, which is 2,546 fewer than the previous year. Furthermore, 17 deaths were reported, which is 10 fewer deaths than the previous year (9,10). Bacterial toxins were the second most prevalent cause of illness in foodborne and waterborne outbreaks, comprising 19.5% of the total disease outbreaks (EFSA, 2017; European Food Safety Authority et al., 2018). The food sources that were found to be associated with the outbreaks were primarily derived from animals, specifically eggs and egg products, pig meat, chicken meat, cheese, seafood and fish items, dairy products, beef meat, and crustaceans. These food categories accounted for varying percentages of strong evidence incidents. In 2015, the occurrence of Salmonella spp. in different foods, such as eggs, was linked to the highest number of documented FBDs. Additionally, it ranked among the top five combinations of food and pathogens according to the total number of indicated instances of FBDs and hospitalizations resulting from these outbreaks (EFSA and ECDC, 2018; Shati et al., 2021). A wide range of different licensed foods and foods prepared by individuals (FPIs) in local stores and restaurants have frequently been blamed for food poisoning. To the best of our knowledge, no data are available on the status of contamination in these foods. The current study was conducted to estimate the contamination levels and detect some bacterial pathogens in different food sources prepared by individuals in Al-Kut City, Iraq. Materials and MethodsSamplesThe current study included the collection of 10 samples from each LF, such as juices and yogurt, and 10 samples from each FPI, such as juices, cheese, cream cheese, and yogurt. Samples were collected from Al-Kut City, Iraq, in June 2023. The samples were transported in an icebox to the Laboratory of Microbiology. The samples were examined by microscopy. Microscopic evaluation in this study employed DMC techniques. Each food sample was blended for 20 seconds to prepare a homogenate. A small portion was then drawn with a micropipette and placed in a hemocytometer for a 1:1 dilution with 0.85% saline. The sample was then placed on a light microscope and focused on while rotating from the 10× to 40× objectives for total microbial cell visualization and enumeration. In this way, the microbial cells in food samples could be counted without the need for any culturing, which was the main advantage of this procedure. The examination steps followed standard protocols for direct microbial cell enumeration from food samples. qRT–PCRThe samples were used in a DNA Extraction Kit (Qiagen, USA) to extract bacterial DNA and following the extraction procedure by the kit manufacturer. DNA was measured using a NanoDrop. The resulting DNA was kept in a deep freezer until qRT-PCR. For Salmonella spp., the invA gene was targeted using the following primers: F: GTGAAATTATCGCCACGTTCGGGCAA and R: TCATCGCACCGTCAAAGGAACC (Tomar and Jyoti, 2016). qRT-PCR evaluations were conducted using Bio-Rad (USA). The amplification processes followed the use of a total volume of 25 μl. The reaction mixture consisted of SYBR Green (USA), 10 mM (pH 8.8) Tris-HCl, 0.1% Triton, 150 mM KCl, 5 mM MgCl2, 100μM dNTPs, 0.5μM per primer, 0.6 U polymerase, and 5 μl DNA. The amplification process consisted of an initial denaturation cycle at 95°C for 5 minutes, followed by 40 cycles of denaturation at 95°C for 15 seconds, primer annealing at a temperature range of 50°C–70°C for 20 seconds, extension at 72°C for 30 seconds, and a further incubation step at 80°C–85°C for 30 seconds to gather the fluorescent data. UreC gene-RT-PCRDNA was extracted from each food sample using the QIAamp DNA Mini Kit (Qiagen, Germany) according to the manufacturer’s instructions. The concentration and purity of DNA were checked using a NanoDrop spectrophotometer (Thermo Fisher, USA). The primers targeting the ureC gene of Helicobacter pylori were selected from He et al. (2002). The forward and reverse primers were TTATCGGTAAAGACACCAGAAA, and the reverse primer was ATCACAGCGCATGTCTTC, generating a 132 bp product. Each PCR reaction contained 12.5 μl of SYBR Green PCR Master Mix (Applied Biosystems, USA), 0.5 μM of each primer, 2 μl of DNA template, and nuclease-free water to a total volume of 25 μl. Amplification was performed in a Bio-Rad CFX96 real-time PCR system with the following program: initial denaturation at 95°C for 10 minutes, followed by 40 cycles of denaturation at 94°C for 10 seconds, annealing at 54°C for 5 seconds, and extension at 72°C for 8 seconds. A melting curve was performed at the end to confirm specificity. Each run included a no-template control, a positive control containing DNA from a reference H. pylori strain, and standard cloned ureC fragment dilutions for quantification. Data were analyzed by comparing the Ct values with the standard curve to estimate the copy numbers. Results are expressed as relative gene copies per gram of food sample. All reactions were performed in triplicate. To prevent contamination, strict aseptic conditions were maintained during DNA extraction and PCR setup. Statistical analysisThe Statistical Package for the Social Sciences software (IBM, USA) was used for data analysis. The analyses consisted of calculating frequencies, percentages, means, and standard deviations. Chi-square (χ²) tests were used to compare prevalence data between food groups, whereas one-way Analysis of Variance was used to compare means of continuous data. A 5% significance level (p < 0.05) was used to indicate statistical significance. Ethical approvalNot needed for this study. ResultsMicrobial load in the food samplesThe results revealed high levels of bacterial and fungal contamination as discovered by the microscopic method. All LF samples, such as juices and yogurt, and 10 FPI samples, such as juices, cheese, cream cheese, and yogurt, contained bacterial and fungal counts ranging from 1 × 10^3 to 1 × 10^4 per ml for liquid foods and per gram for semi-solid foods (Table 1). Table 1. Microbial load in food samples obtained from the Al-Kut city market.
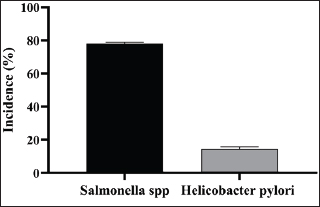
SalmonellaFurthermore, these samples showed a significant (p < 0.05) presence of Salmonella spp. in many food samples at different rates ranging from 76% to 80%, especially in FPIs. In addition, these samples showed a significant (p < 0.05) presence of H. pylori, which ranged from 12% to 17% of the FPIs (Fig. 1).
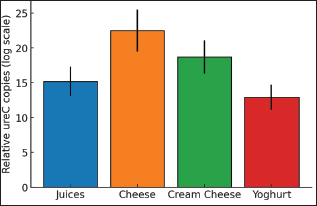
Fig. 1. Incidence of Salmonella spp. and Helicobacter pylori in food samples from Al-Kut city markets. The qPCR assay targeting the ureC gene of H. pylori showed positive results in a proportion of the examined food samples. Of all the samples tested, 16% carried detectable ureC sequences. The overall mean copy number per gram was higher in dairy foods than in juices. Cheese showed the highest contamination rate, with 22% of the samples being positive. Cream cheese was next with 18% positive samples. In contrast, 12% positivity was found in yogurt drinks and 10% in juices. The mean gene copy values were also higher in cheese and cream cheese, suggesting a greater bacterial load in these products. A statistical comparison revealed a significant difference between dairy items and juices (p < 0.05). No significant difference was observed between cream cheese and cheese. The relative abundance values confirmed that unprocessed or locally prepared dairy foods carried more contamination risk than other categories. The results are summarized in Figure 2, where each food group is represented with its mean copy numbers and error bars.
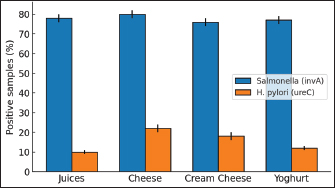
Fig. 2. Detection of the ureC gene of H. pylori in different food samples. Bars represent the mean relative gene copy numbers per gram of food for juices, cheese, cream cheese, and yogurt. The bar chart compares the prevalence of Salmonella invA and H. pylori ureC across the four food types. Salmonella was consistently more common in all samples, with rates close to or above 76%. In contrast, H. pylori was less frequent and concentrated in dairy products. Cheese and cream cheese had the highest H. pylori positivity, whereas juices and yogurt had lower values. This figure highlights that while Salmonella remains the main contaminant, H. pylori is still relevant in unprocessed dairy products (Fig. 3).
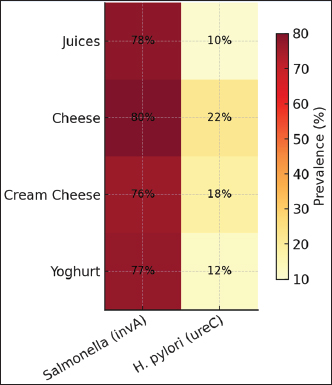
Fig. 3. Prevalence of Salmonella (invA) and Helicobacter pylori (ureC) across four food groups. The heatmap displays the contamination levels of both pathogens across the examined foods. Darker colors indicate higher positivity, making the differences easy to see. The map confirms that Salmonella was widespread in all food groups, whereas H. pylori was mainly associated with cheese and cream cheese. This visual also shows that yogurt and juices carried a lower risk for H. pylori but remained positive for Salmonella (Fig. 4).
Fig. 4. Heatmap of Salmonella and Helicobacter pylori contamination in different food samples. DiscussionThis assemblage of Enterobacteriaceae exhibits pathogenic attributes and represents a prevalent global etiological agent responsible for enteric infections, commonly referred to as food poisoning. Salmonella has two main species, namely S. enterica and S. bongori, with the potential to induce illness in humans. Salmonella is classified into distinct serotypes using the Kaufmann-White typing scheme, which was initially introduced in 1934. This scheme enables the differentiation of various Salmonella strains based on their specific surface and flagellar antigenic characteristics. Salmonella species are frequently denoted by their serotype designations. An illustration of this can be seen in the case of Salmonella enterica subsp. enterica, which exhibits a considerable degree of subdivision into various serotypes, such as S. enterica. Enteritidis and Streptococcus typhimurium. Specific serovars of S. enterica are responsible for more severe illnesses, such as typhoid fever. Salmonella spp. Salmonella species are the predominant bacterial agents responsible for foodborne illnesses in the United States. The annual incidence of Salmonella infections in the United States exceeds 1 million cases, resulting in almost 20,000 hospitalizations and 400 fatalities. Salmonella species commonly reside within the intestinal tracts of various livestock and numerous wildlife species. Salmonella species infection typically arises from the consumption of contaminated fecal-based food. Salmonella FBDs are frequently associated with the consumption of eggs, meat, and poultry. However, these bacteria can contaminate a broader range of food items, including fruits and vegetables. According to the current study, H. pylori was found in unprocessed ewe milk. Helicobacter pylori was detected in 1:38 positive PCR unprocessed ewe milk specimens and in 1:6 of positive PCR biopsy specimens. Helicobacter pylori in unpasteurized milk specimens has become infrequent following the discovery of these findings. During an investigation carried out in Japan, H. pylori was observed in unpasteurized milk from cows that tested positive for PCR. The bacterium was detected in 4 out of 20 samples of unpasteurized milk from cows in Greece, as well as in 2 out of 11 PCR-positive specimens of unprocessed sheep milk and 1 out of 15 PCR-positive specimens of unprocessed buffalo milk in Iran. Subsequently, an elevated incidence of bacteria in unprocessed milk within Iranian regions was documented. Moreover, the investigation of H. pylori presence in dairy has been limited to a few studies. Mousavi et al. (2014) found that a significant proportion of local cheese (30%), cream (15%), butter (5%), and ice cream (27%) samples tested positive for H. pylori, all of which were produced using unprocessed milk. There is a scarcity of research examining the identification of H. pylori in foods. Helicobacter pylori was detected in approximately 14%–37% of meat samples obtained from different sources, such as cows and camels. In addition, conducted an analysis on 550 ready-to-consume samples of foods. Their findings revealed the presence of H. pylori in approximately 74% of the samples. Notably, the highest levels of contamination were observed in olive-based salads (36%), fruit-prepared salads (30%), and soups (22%). Furthermore, found that H. pylori was detected in 20% of specimens of foods that were ready to be consumed, representing a prevalence rate of 20%. The potential cause for the elevated occurrence of the bacterium in foods that were ready to be consumed, such as meats and vegetables, may be contamination that occurs after the processing stage. This study analyzed various locally sourced and commercially licensed products obtained from Al-Kut City. Reported results show significant bacterial and fungal contamination. Fungal and bacterial counts ranged from 10³ to 104 cells/g or ml. The range indicates poor adherence to microbial measures and storage handling standards. Such findings correlate with and build upon the established findings of microbe-laden foods in developing nations. Salmonella prevalence at 76%–80% across the varied foods illustrates the confirmed pathogen. Consistent Salmonella contamination across prepared and licensed foods suggests weak hygiene protocols. Salmonella presence indicates a risky supply chain and/or infrastructural food handling practices. Fungal and bacterial contamination present additional food safety concerns. Overall, the findings of this study correlate with previously published works and provide a novel data contribution to the specific field. Simultaneously, the detection of H. pylori ureC in approximately 16% of samples occurred with the highest positivity for cheese and cream cheese. This also raises concern for the first time in documenting these data from Al-Kut, as it aligns with previous works of other countries, as it suggests the presence of unprocessed and/or minimally processed dairy foods as potential sources of risk in the community. The current results also refine the previous studies and illustrate specific product groups, especially locally prepared cheese and cream cheese, which may require H. pylori and Salmonella testing due to the risk of salmonella being widespread in the contaminated fruits and vegetables. The results also indicate that the wider Salmonella risk suggests that more drastic public health measures may be needed with some product groups than others. ConclusionThe findings of the current study indicate that the food samples collected in Al-Kut City, whether made in-house or commercially produced, are highly contaminated and contaminated with Salmonella and, to a much lesser degree, H. pylori ureC. Salmonella’s widespread occurrence in various food types indicates serious deficiencies in food preparation, handling, and storage hygiene. In particular, H. pylori ureC contamination of soft and cream cheeses suggests that these products pose a significant risk. The present situation suggests a clear need for improved and integrated sanitation, increased controls on the manufacture of dairy products and juice, and practical and live microbial testing to diminish the risk of foodborne diseases in the population. AcknowledgmentThe authors thank the College of Veterinary Medicine, University of Al-Qadisiyah, for their support in this study. Conflict of interestThe author declares that there is no conflict of interest. FundingSelf-funding. No external funding source was available. Authors’ contributionsOne author for this study. Data availabilityData are available when requested by the corresponding author. ReferencesEuropean Food Safety Authority (EFSA). (2017). The European Union summary report on trends and sources of zoonoses, zoonotic agents and food-borne outbreaks in 2016. EFSA J. 15(12), e05077; doi: 10.2903/j.efsa.2017.5077 European Food Safety Authority (EFSA). European Centre for Disease Prevention. and Control, (.E.C.D.C.). 2018. The European Union summary report on trends and sources of zoonoses, zoonotic agents and food-borne outbreaks in 2017. EFSA. J. 16(12), e05500; doi:10.2903/j.efsa.2018.5500 He, Q., Wang, J.P., Osato, M. and Lachman, L.B. 2002. Real-time quantitative PCR for detection of Helicobacter pylori. J. Clin. Microbiol. 40(10), 3720–3728. Mengistu, D.A., Belami, D.D., Tefera, A.A. and Alemeshet Asefa, Y. 2022. Bacteriological quality and public health risk of ready-to-eat foods in developing countries: systematic review and meta-analysis. Microbiol. Insights 15, 11786361221113916; doi:10.1177/11786361221113916 Menini, A., Mascarello, G., Giaretta, M., Brombin, A., Marcolin, S., Personeni, F., Pinto, A. and Crovato, S. 2022. The critical role of consumers in the prevention of foodborne diseases: an ethnographic study of Italian families. Foods 11(7), 1006; doi:10.3390/foods11071006 Pepper, I.L. and Gerba, C.P. 2015. Environmental microbiology (3rd ed.). San Diego, CA: Academic Press. Pires, S.M., Desta, B.N., Mughini-Gras, L., Mmbaga, B.T., Fayemi, O.E., Salvador, E.M., Gobena, T., Majowicz, S.E., Hald, T., Hoejskov, P.S., Minato, Y. and Devleesschauwer, B. 2021. Burden of foodborne diseases: think global, act local. Curr. Opinion Food Sci. 39, 152–159; doi:10.1016/j.cofs.2021.02.011 Shati, A.A., Al Qahtani, S.M., Shehata, S.F., Alqahtani, Y.A., Aldarami, M.S., Alqahtani, S.A., Alqahtani, Y.M., Siddiqui, A.F. and Khalil, S.N. 2021. Knowledge, attitudes, and practices towards food poisoning among parents in Aseer Region, Southwestern Saudi Arabia. Healthcare 9(12), 1650; doi:10.3390/healthcare9121650 Switaj, T.L., Winter, K.J. and Christensen, S.R. 2015. Diagnosis and management of foodborne illness. Am. Fam. Physician 92(5), 358–365. Todd, E. 2020. Food-borne disease prevention and risk assessment. Int. J. Environ. Res. Public Health 17(14), 5129; doi:10.3390/ijerph17145129 Tomar, R.S. and Jyoti, A. 2016. Culture-independent quantification of Salmonellae in food samples by real-time PCR. J. Appl. Pharm. Sci. 6(11), 153–157. Wu, G., Yuan, Q., Wang, L., Zhao, J., Chu, Z., Zhuang, M., Zhang, Y., Wang, K., Xiao, P., Liu, Y. and Du, Z. 2018. Epidemiology of foodborne disease outbreaks from 2011 to 2016 in Shandong Province, China. Med. (Baltimore). 97(45), e13142; doi:10.1097/MD.0000000000013142 | ||
| How to Cite this Article |
| Pubmed Style Akmam Ali Habeeb. Molecular and conventional detection of Salmonella spp. and Helicobacter pylori in licensed and individually prepared foods in the city of Al-Kut, Iraq. doi:10.5455/OVJ.2026.v16.i1.49 Web Style Akmam Ali Habeeb. Molecular and conventional detection of Salmonella spp. and Helicobacter pylori in licensed and individually prepared foods in the city of Al-Kut, Iraq. https://www.openveterinaryjournal.com/?mno=283813 [Access: February 01, 2026]. doi:10.5455/OVJ.2026.v16.i1.49 AMA (American Medical Association) Style Akmam Ali Habeeb. Molecular and conventional detection of Salmonella spp. and Helicobacter pylori in licensed and individually prepared foods in the city of Al-Kut, Iraq. doi:10.5455/OVJ.2026.v16.i1.49 Vancouver/ICMJE Style Akmam Ali Habeeb. Molecular and conventional detection of Salmonella spp. and Helicobacter pylori in licensed and individually prepared foods in the city of Al-Kut, Iraq. doi:10.5455/OVJ.2026.v16.i1.49 Harvard Style Akmam Ali Habeeb (2026) Molecular and conventional detection of Salmonella spp. and Helicobacter pylori in licensed and individually prepared foods in the city of Al-Kut, Iraq. doi:10.5455/OVJ.2026.v16.i1.49 Turabian Style Akmam Ali Habeeb. 2026. Molecular and conventional detection of Salmonella spp. and Helicobacter pylori in licensed and individually prepared foods in the city of Al-Kut, Iraq. doi:10.5455/OVJ.2026.v16.i1.49 Chicago Style Akmam Ali Habeeb. "Molecular and conventional detection of Salmonella spp. and Helicobacter pylori in licensed and individually prepared foods in the city of Al-Kut, Iraq." doi:10.5455/OVJ.2026.v16.i1.49 MLA (The Modern Language Association) Style Akmam Ali Habeeb. "Molecular and conventional detection of Salmonella spp. and Helicobacter pylori in licensed and individually prepared foods in the city of Al-Kut, Iraq." doi:10.5455/OVJ.2026.v16.i1.49 APA (American Psychological Association) Style Akmam Ali Habeeb (2026) Molecular and conventional detection of Salmonella spp. and Helicobacter pylori in licensed and individually prepared foods in the city of Al-Kut, Iraq. doi:10.5455/OVJ.2026.v16.i1.49 |