| Research Article | ||
Open Vet. J.. 2026; 16(2): 1281-1288 Open Veterinary Journal, (2026), Vol. 16(2): 1281-1288 Research Article Identification of ticks from conflict Sumatran tigers (Panthera tigris sumatrae) in Bukittinggi, West Sumatra, IndonesiaLisa Hidayati1*, Yoli Zulfanedi2, Supriyono Supriyono3, Fitrine Ekawasti4, Bilan Diurai Viawan2, Rita Gusmiati1, Nike Puspita Alwi1, Halima Thusadya1 and Badriyatul Humaida11Department of Nursing and Public Health, Prima Nusantara University Bukittinggi, Bukittinggi, Indonesia 2Kinantan Wildlife and Cultural Park, Bukittinggi City, Indonesia 3Division of Parasitology and Health Entomology, Department of Animal Diseases and Veterinary Public Health, School of Veterinary Medicine and Biomedicine, IPB University, Bogor, Indonesia 4Research Centre for Veterinary Science, National Research and Innovation Agency (BRIN), Cibinong, Indonesia *Corresponding Author: Lisa Hidayati, Department of Nursing and Public Health, Prima Nusantara University Bukittinggi, Bukittinggi, Indonesia. Email: lisahidayatidnr [at] gmail.com Submitted: 21/09/2025 Revised: 17/01/2026 Accepted: 27/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
AbstractBackground: Vector-borne diseases transmitted by ticks pose risks to both wildlife and humans. In Indonesia, tick-borne infections are rarely considered in differential diagnoses of febrile illnesses such as dengue and malaria, leading to potential underdiagnosis. Wildlife–human conflict, including cases of Sumatran tigers entering residential areas, may facilitate pathogen spillover at the human–animal interface. Aim: This study aimed to identify and characterize molecular features of tick-borne pathogens associated with conflict-affected wildlife using polymerase chain reaction (PCR) amplification and sequencing and to assess their potential zoonotic risk. Methods: Ticks were collected from Sumatran tigers involved in human–wildlife conflict events reported by the public, from both live and deceased animals. Tick species were identified using morphological and molecular approaches. Pathogen detection was conducted using PCR amplification and sequencing of bacterial DNA. Results: Morphological and molecular analyses identified three tick species, including Amblyomma javanense, Haemaphysalis bispinosa, and Haemaphysalis wellingtoni. Molecular screening detected the presence of Rickettsia spp. and Anaplasma spp. in the collected ticks, indicating circulation of multiple tick-borne pathogens in conflict-affected wildlife. Conclusion: The detection of Rickettsia spp. and Anaplasma spp. in Amblyomma and Haemaphysalis ticks highlights their potential role as vectors of zoonotic pathogens. These findings underscore the importance of integrating tick-borne disease surveillance into wildlife monitoring and adopting a One Health approach to improve early detection and prevention of neglected zoonotic diseases in Indonesia, particularly in Bukittinggi, West Sumatra. Keywords: Tick-borne diseases, Zoonosis, Rickettsia spp., Anaplasma spp., Sumatran tiger, One Health. IntroductionVector-borne diseases are caused by bacteria, viruses, and parasites transmitted by arthropods such as ticks, fleas, and mites. Several important zoonotic vector-borne diseases include rickettsioses, leptospirosis, granulocytic ehrlichiosis, Lyme borreliosis, and Japanese encephalitis. Among these, tick-borne infections caused by Rickettsia, Anaplasma, and Borrelia are of particular concern due to their wide host range, zoonotic potential, and frequent misdiagnosis (Supriyono et al., 2019; Sophia et al., 2024). Rickettsial infections are recognized as neglected zoonotic diseases and represent a major cause of non-malarial febrile illness in Southeast Asia, second only to dengue fever (Acestor et al., 2012; Acestor et al., 2012; Tomassone et al., 2018). Clinical manifestations often overlap with dengue, leptospirosis, and typhoid fever, leading to frequent misdiagnosis and delayed treatment, which can increase morbidity and mortality (Hidayati et al., 2017a; Lokida et al., 2020; Salje et al., 2021). Globally, more than one million rickettsial cases are estimated to occur annually, with certain spotted fever group infections such as Rocky Mountain spotted fever exhibiting high case fatality rates (Chikeka and Dumler, 2015; Clark, 2019). Environmental changes, including deforestation, land-use conversion, and climate variability, have altered vector ecology and increased contact between humans, wildlife, and domestic animals. These processes facilitate the spillover of tick-borne pathogens, particularly in tropical regions such as Indonesia, where climatic conditions favor vector survival and reproduction (Hidayati et al., 2017b). West Sumatra is considered a potential hotspot for vector-borne diseases due to its suitable environmental conditions and increasing human–wildlife interactions. The Sumatran tiger (Panthera tigris sumatrae), an endemic and critically endangered species, increasingly comes into contact with human settlements due to habitat loss and fragmentation. Beyond threats from poaching and habitat destruction, infectious and parasitic diseases also pose risks to tiger conservation. Ticks parasitizing large carnivores may act as reservoirs or vectors of zoonotic pathogens, posing risks not only to wildlife but also to surrounding human populations. While tick species such as Haemaphysalis bispinosa have been reported on tigers in other Asian countries (Kumar et al., 2018), data on tick diversity and tick-borne pathogens associated with Sumatran tigers in Indonesia remain extremely limited. Molecular diagnostic tools such as polymerase chain reaction (PCR) provide higher sensitivity and specificity than conventional methods for detecting tick-borne pathogens, particularly in asymptomatic wildlife hosts or cases with low pathogen loads. Molecular approaches are therefore essential for accurate pathogen detection, species identification, and zoonotic risk assessment. This study aimed to identify tick species parasitizing conflict-affected Sumatran tigers and to detect tick-borne pathogens (Rickettsia, Anaplasma, and Borrelia) using molecular methods, as part of an integrated zoonotic surveillance approach. Material and MethodsSample collectionEctoparasites (ticks) were collected from a Sumatran tiger wildlife conflict (reported by the community to the West Sumatra Province Natural Resources Conservation Agency). Ectoparasites collected from each wildlife were placed in separate 1.5 mL tubes with 70% ethanol and stored at +4°C until examination (Fig. 1). The sampling technique used was convenience sampling. Morphology identificationThere were two Sumatran tigers reported from the public (one of them was dead, caught in a wild boar snare set by residents. Eighteen ticks from two Sumatran tigers were collected from the body using forceps. Tick species, stage, and sex were identified based on morphological features under stereo microscopy following taxonomic keys and molecular analysis at the Biomedical Laboratory of Prima Nusantara University, Bukittinggi, and the Kinantan Wildlife and Cultural Park laboratory, Bukittinggi, West Sumatra (Hidayati et al., 2024). Molecular Analysis using PCRa. DNA extraction and PCR amplificationDNA extraction was performed using the GENEAID® Genomic DNA Mini Kit (Tissue). The obtained ticks were divided into 5 pools distinguished by species and collection location. After being divided into 5 pools in Eppendorf tubes, 1 micropestle was used for each to crush the tick body and grind. DNA extraction according to the available protocol procedures. The DNA extraction results were stored at −20°C until used for detection using PCR. DNA amplification preparation will be carried out using the MyTaq HS Red® master mix according to the available procedures. DNA amplification was carried out on a LongGene A600 Super Gradient® Thermal Cycler. Pathogen detection was carried out against Anaplasma, Rickettsia, and Borrelia. The oligonucleotide primer pair used in this study was a 17-kDa antigen for Rickettsia detection, the groEL gene to detect Anaplasma, and 16s rDNA with flaB for Borrelia detection (Supriyono et al., 2019). b. DNA gene electrophoresisThe DNA amplification results were then subjected to electrophoresis. Electrophoresis was performed using a 1.5% agarose gel stained with flourosafe. Electrophoresis was carried out with 1X TAE buffer at 100 V for 35 minutes. The electrophoresis gel was then placed under UV light to detect the presence of target DNA using the Cleaver Scientific gelONE® gel documentation system. c. DNA detectionThe PCR detection program for Rickettsia with the 17-kDa gene included a pre-denaturation temperature of 95°C for 5 minutes, followed by 40 cycles of denaturation at 94°C for 30 seconds, annealing at 46.5°C for 30 seconds, and elongation at 72°C for 30 seconds. Finally, a final elongation of 72°C for 7 minutes. The PCR program for detecting Borrelia with the flaB gene included a pre-denaturation temperature of 94°C for 5 minutes, followed by 40 cycles of denaturation at 94°C for 45 seconds, annealing at 50°C for 30 seconds, and elongation at 72°C for 30 seconds. Finally, a final elongation of 72°C for 10 minutes. Nested PCR program for the detection of Anaplasma spp. The first stage included a pre-denaturation temperature of 94°C for 3 minutes, followed by 40 cycles of denaturation at 94°C for 30 seconds, annealing at 57°C for 30 seconds, and elongation at 72°C for 30 seconds. Finally, a final elongation at 72°C for 7 minutes. The second phase of the nested PCR program for Anaplasma was the same as the first stage, except that the annealing temperature was changed to 54°C (Supriyono et al., 2019). The base sequence of nucleotide data as a result of manual sequencing was reprinted into the Basic Local Alignment Search Tool (BLAST) program on the website www.ncbi.nlm.nih.gov. so that the similarity information of the sample sequence with the organism sequence in the BLAST data. The nucleotide sequencing results were manually edited based on the chromatogram. Multiple alignment of the edited nucleotide sequence with several ITS sequences of 2 genera (GenBank) using Clustal W integrated in MEGA 11 (Molecular Evolutionary Genetics Analysis).
Fig. 1. The process of taking tick samples from Sumatran tigers (Panthera tigris) Ethical approvalThe study has been approved by the Animal Research Ethics Committee from the National Research and Innovation Agency, Indonesia (BRIN No. 175/KE.02/SK/08/2025). ResultsTicks collected from conflict-affected Sumatran tigers (Panthera tigris sumatrae) in Pasaman Regency were morphologically identified as three species: H. bispinosa, Haemaphysalis wellingtoni, and Amblyomma javanense. Morphological characterization of A. javanense belonging to the rigid tick family (Ixodidae) was identified based on a rectangular basis capituli, small and deep porose areas, wide anterior and narrow posterior palpus, with the second segment longer than the third; dark crimson and heart-shaped scutum without enamel; small and flat eyes with only one imprint; anal groove behind the anus; and comma-shaped peritrema. In addition, coxa I has two expansive, blunt, and well-separated spurs, and coxae II, III, and IV each have expansive, blunt, and rounded spurs (Jabin et al., 2019) (Fig. 2). Haemaphysalis wellingtoni was characterized by medium body size, small blunt cornua, a posterointernally directed spur on palpal article III, and a lateral groove on the conscutum (Kwak and Ng, 2022) (Fig. 3). Haemaphysalis bispinosa exhibited short cervical grooves, lateral grooves extending posteriorly on the idiosoma, dense punctuations, the genital opening is parallel to the second coxa, and the anal opening is opposite the oval-shaped spiracle. In addition, the louse has long, thick legs (Sahara et al., 2023) (Fig. 4). In addition, it has the characteristics of rectangular base capitula; small and deep shaft area; wide anterior and narrow posterior part of the palp, with the second segment longer than the third; dark red and heart-shaped scutum without enamel; small and flat eyes with only one imprint; anal groove behind the anus; comma-shaped peritrema. In addition, coxa I has two very wide, blunt, and well-separated spurs, and coxa II, III, and IV each have very wide, blunt, and rounded spurs (Fig. 4).
Fig. 2. Morphology of A. javanense (a) dorsal and (b) ventral.
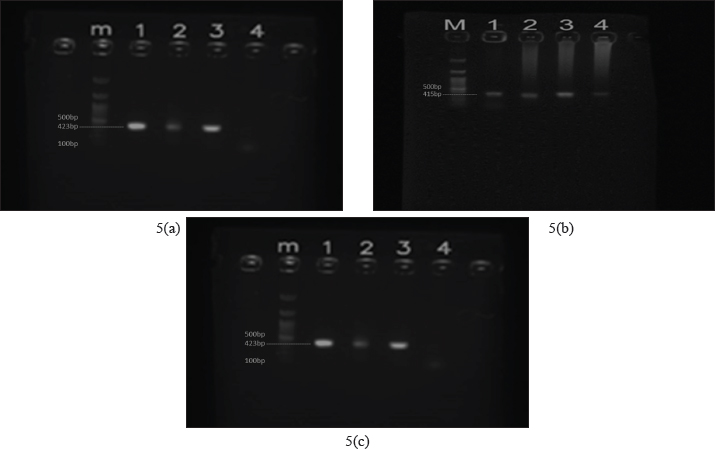
Fig. 3. Morphology of H. wellingtoni 3(a) dorsal and 3(b) ventral These tick species have previously been reported on wildlife and livestock in Indonesia; however, this study represents the first documented record of tick infestation on Sumatran tigers from Bukitinggi, West Sumatra, Indonesia. Human–wildlife conflict presents a significant opportunity for vectors, particularly ticks, to transmit pathogens to humans through bites. The study of tick infestation in Panthera tigris sumatrae presented in this paper is the first of its kind in Indonesia. Ticks previously reported in wildlife and livestock in Indonesia include lizards (Varanus salvator) infested with A. javanense (Supriyono et al., 2019), Dermacentor auratus, and H. bispinosa, which infest the Moa buffalo (Bubalus bubalis) (Utami and Kunda, 2023), cattle infested with H. bispinosa (Sahara et al., 2023), Ixodes cordifer cuscus (Utami, Budianto, and Sahara 2021), and dogs infested with Rhipicephalus sanguineus (Kesumawati Hadi and Soviana, 2015). Molecular detection of tick-borne pathogensFollowing morphological identification, ticks were subjected to molecular analysis using PCR. Genomic DNA amplification was confirmed by electrophoresis prior to pathogen screening. PCR assays targeting Rickettsia spp., Anaplasma spp., and Borrelia spp. yielded amplicons of expected sizes (423 bp for Rickettsia spp.; and 415 bp for Anaplasma spp.), consistent with previous studies (Supriyono et al., 2019). The results of DNA electrophoresis (Fig. 5a) showed that the sample was detected as Rickettsia spp. (samples 1–4 consisting of A. javanense, male H. wellingtoni, female H. wellingtoni, male H. bispinosa), detected as Anaplasma spp. shown in Fig. 5b and not detected Borelia spp. in Fig. 5c.
Fig. 4. Morphology of Haemaphysalis bispinosa 4(a) dorsal and 4(b) ventral.
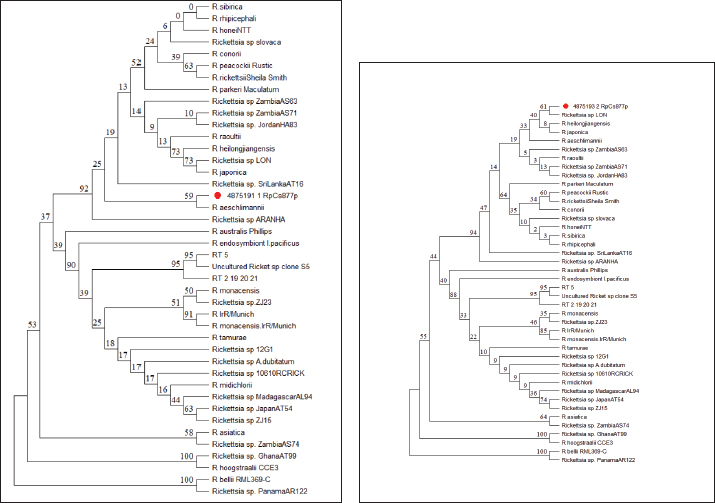
Fig. 5. Results of pathogen DNA electrophoresis based on the type of species found (sample 1–4, namely A. javanense, male H. wellingtoni, female H. wellingtoni, male Haemaphysalis bispinosa). (a) Rickettsia spp. detected with clear bands in samples 1 and 2. (b) Anaplasma spp. detected in samples 1, 2, and 3. (c) Borrelia spp. not detected. Phylogenetic analysisSequencing of PCR-positive samples targeting the mitochondrial 16S rRNA gene was performed to confirm pathogen identity. Phylogenetic analysis (maximum likelihood with bootstrap resampling) revealed that Sample R1 clustered closely with Rickettsia aeschlimannii, showing 99.09% sequence similarity based on nBLAST analysis (Fig. 6a) (McGinley et al., 2021). Sample R2 showed close genetic relatedness to Rickettsia spp. LON from Japan (Fujita, 2008) 98.78% similarity, and clustered with Rickettsia heilongjiangensis from China (Zhang et al., 2000) and Rickettsia japonica from Thailand (Gaywee et al., 2007) as sister taxa (Fig. 6b). These findings indicate the circulation of pathogenic Rickettsia lineages with known zoonotic potential in ticks parasitizing conflict wildlife. DiscussionThis study represents the first report of tick-borne pathogens detected in vectors parasitizing conflict-affected Sumatran tigers (Panthera tigris sumatrae) in Indonesia. Although ticks were collected from only two tigers, one deceased and one alive, both of which entered residential areas, the identification of Rickettsia spp. and Anaplasma spp. in A. javanense and H. wellingtoni highlights the potential role of conflict wildlife as sentinels for zoonotic pathogens at the human wildlife interface. The detection of these bacterial genera is consistent with reports from other parts of Southeast Asia, where Haemaphysalis and Amblyomma species have been implicated in the circulation of zoonotic pathogens. For example, H. bispinosa has been associated with Rickettsia spp. in Thailand and Bartonella spp. in Peninsular Malaysia, while H. wellingtoni has been documented feeding on humans, albeit less frequently. Similarly, A. javanense has been shown to harbor Jingmen tick virus in China, demonstrating its competence as a vector of emerging pathogens. Together, these findings suggest that the tick species identified in this study are not incidental parasites but may participate in broader regional transmission cycles involving wildlife, livestock, and humans.
Fig. 6. (a) Phylogenetic tree of tick sample R1 from the Sumatran Tiger (P. tigris Sumatrae). (b). R2 phylogenetic tree of the Sumatran tiger (P. tigris Sumatrae). Importantly, Indonesia currently lacks documented human cases linked to Rickettsia or Anaplasma associated with these tick species. However, the close proximity between humans and wildlife during conflict events substantially increases opportunities for spillover. Given the demonstrated capacity of ticks to maintain pathogens through transovarial, transstadial, and horizontal transmission, these vectors may function not only as transmitters but also as reservoirs, sustaining pathogens in nature even in the absence of recognized human disease. While these transmission mechanisms were not directly examined in the present study, their well-established role in maintaining spotted fever group Rickettsia underscores the epidemiological relevance of our findings. From a One Health perspective, the detection of zoonotic bacterial genera in ticks collected from conflict wildlife reinforces the need for integrated surveillance across human, animal, and environmental sectors. As emphasized by Meurer (2025), neglecting any one component undermines timely diagnosis, prevention, and control of neglected zoonoses. Molecular surveillance of ticks associated with endangered wildlife such as Sumatran tigers can therefore provide early warning signals of emerging infectious threats, support public health preparedness, and inform biosurveillance strategies in regions where differential diagnosis of tick-borne diseases is rarely considered. Several limitations of this study must be acknowledged. The small sample size, lack of quantitative prevalence data, absence of uninfested control animals, and limited geographic scope restrict the generalizability of the findings. In addition, while molecular detection confirms the presence of bacterial DNA, it does not establish pathogen viability, vector competence, or pathogenicity in either tigers or humans. These gaps highlight the need for expanded longitudinal studies incorporating larger sample sizes, ecological data, and serological or clinical investigations in humans and domestic animals. Pathogenic bacterial genera have been detected in H. bispinosa in Thailand, including Rickettsia (Malaisri et al., 2015), and Bartonella in Peninsular Malaysia (Kho et al., 2015). Haemaphysalis wellingtoni has been recorded as sucking human blood, but this appears to be less common than H. bispinosa (Kho et al., 2015). Amblyomma javanense also has the potential to be a vector for zoonotic diseases, especially in humans. The Jingmen tick virus (JMTV) strain was isolated from A. javanense ticks into an embryo-derived tick cell line in China. It can accumulate in the salivary glands of experimentally infected A. javanense (Jia et al., 2019). Therefore, this study revealed that the wildlife conflict, especially Sumatran tigers close to humans and their related ticks, harbored a variety of unknown bacteria. Although the pathogenicity or biology of these detected bacteria remains unclear, garnering information about these novel bacteria is essential to devise diagnostic strategies for unknown infectious diseases. However, some literature reports that ticks acquire Rickettsia and Anaplasma or some bacteria of the Spot Fever Group infection in three ways, namely through transovarial transmission (from the female to her eggs/offspring), transstadial (from eggs to the next life cycle stage to adulthood), and horizontal transmission (infection acquired during a blood meal on a host containing Rickettsia). Ticks can also act as reservoirs for these bacteria, as with Amblyomma spp. for R. africae, and play a role in maintaining the agent in nature (Fournier and Raoult, 2020). This study demonstrates that ticks associated with conflict-affected Sumatran tigers harbor bacterial genera with recognized zoonotic potential. These findings underscore the importance of incorporating wildlife-associated vectors into molecular surveillance programs and adopting a One Health framework that simultaneously supports public health preparedness and Sumatran tiger conservation in Indonesia. ConclusionTicks are recognized vectors capable of transmitting pathogens from wildlife to humans, particularly in situations of increased human–wildlife interaction. In this study, morphological and molecular analyses of ticks collected from conflict-affected Sumatran tigers identified three species: A. javanense, H. bispinosa, and H. wellingtoni. Molecular screening provided preliminary evidence of the presence of Rickettsia spp. and Anaplasma spp. in A. javanense, male H. wellingtoni, and female H. wellingtoni. Although the epidemiological significance and zoonotic potential of these findings remain unclear, the results highlight the need for further investigation involving larger sample sizes, broader geographic coverage, and integration with human and domestic animal surveillance. Such efforts are essential to clarify transmission dynamics and assess public health relevance within a One Health framework. AcknowledgmentsThe authors acknowledge the Ministry of Research and Technology of Higher Education, Republic of Indonesia, for providing funding support to complete this study. Conflict of interestThe authors declared that there is no conflict of interest. FundingThe authors acknowledge the Ministry of Research and Technology of Higher Education, Republic of Indonesia, for providing funding support to complete this study. Author’s contributionLH and YZ developed the concept and design. LH, YZ, S, FE, RG, and NPA contributed to data analysis/interpretation. LH, YZ, S, FE, RG, and NPA contributed to drafting the manuscript. LH, YZ, S, FE, RG, and NPA contributed to the critical revision of the manuscript. LH, S, and FE contributed to statistical analysis. LH, YZ, RG, and NPA contributed to admin and technical or material support. S and FE contributed to supervision. LH, YZ, S, FE, RG, and NPA contributed to the final approval. Data availabilityAll data are provided in the manuscript. ReferencesAcestor , N., Cooksey, R., Newton, P.N., Ménard, D., Guerin, P.J., Nakagawa, J., Christophel, E., González, I.J. and Bell, D. 2012. Mapping the Aetiology of Non-Malarial Febrile Illness in Southeast Asia through a Systematic Review—Terra Incognita Impairing Treatment Policies. PLos One 7(9), e44269; doi:10.1371/journal.pone.0044269 Fournier, P.E. and Raoult, D. 2020. Tick-Borne Spotted Fever Rickettsioses.In Hunter’s Tropical Medicine and Emerging Infectious Diseases. Elsevier, pp: 587–93. Gaywee, J., Sunyakumthorn, P., Rodkvamtook, W., Ruang-Areerate, T., Mason, C.J. and Sirisopana, N. 2007. Human Infection with Rickettsia Sp. Related to R. Japonica, Thailand. Emerg. Infect. Dis. 13(4), 657–659; doi:10.3201/eid1304.060585 Hidayati., Lisa., Zulfanedi, Y., Viawan, B.D., Supriyono., Alhamda, S., Ramadanti, T. and Gusmiati, R. 2024. The Identification of Ticks That Have the Potential to Transmit Diseases To Human (Ticks Borne Diseases) That Infested Sumatran Tiger (Panthera Tigris Sumatrae) in West Sumatera Province. Jurnal. Kesehatan. 15(3), 30–37; doi:10.35730/jk.v15i3.1152 Jia, N., Liu, H.B., Ni, X.B., Bell-Sakyi, L., Zheng, Y.C., Song, J.L., Li, J., Jiang, B.G., Wang, Q., Sun, Y., Wei, R., Yuan, T.T., Xia, L.Y., Chu, Y.L., Wei, W., Li, L.F., Ye, J.L., Lv, Q.Y., Cui, X.M., Guan, Y., Tong, Y.G., Jiang, J.F., Lam, T.T.Y. and Cao, W.C. 2019. Emergence of Human Infection with Jingmen Tick Virus in China: a Retrospective Study. EBioMedicine 43, 317–324; doi:10.1016/j.ebiom.2019.04.004 Kesumawati Hadi, U. and Soviana, S. 2015. Prevalence of Ticks and Tick-Borne Diseases in Indonesian Dogs. J. Vet. Sci. &. Technol. 07(03), 7; doi:10.4172/2157-7579.1000330 Kho, K.L., Koh, F.X., Jaafar, T., Hassan Nizam, Q.N. and Tay, S.T. 2015. Prevalence and Molecular Heterogeneity of Bartonella Bovis in Cattle and Haemaphysalis Bispinosa Ticks in Peninsular Malaysia. BMC Vet. Res. 11(1), 153; doi:10.1186/s12917-015-0470-1 Kumar, K. G. A., Ravindran, R., Johns, J., Chandy, G., Rajagopal, K., Chandrasekhar, L., George, A. J. and Ghosh, S. 2018. Ixodid Tick Vectors of Wild Mammals and Reptiles of Southern India. J Arthropod-Borne Dis., 12(3), 276–285. Malaisri, P., Hirunkanokpun, S., Baimai, V., Trinachartvanit, W. and Ahantarig, A. 2015. Detection of Rickettsia and Anaplasma from Hard Ticks in Thailand. J. Vector Ecol. 40(2), 262–268; doi:10.1111/jvec.12163 Mcginley, L., Hansford, K.M., Cull, B., Gillingham, E.L., Carter, D.P., Chamberlain, J.F., Hernandez-Triana, L.M., Phipps, L.P. and Medlock, J.M. 2021. First Report of Human Exposure to Hyalomma Marginatum in England: further Evidence of a Hyalomma Moulting Event in North-Western Europe?. Ticks. Tick-Borne. Dis. 12(1), 101541; doi:10.1016/j.ttbdis.2020.101541 Sahara, A., Budianto, B.H., Kunda, R.M. and Firdausy, L.W. 2023. Tick (Acari: ixodidae) Infestation in Cattle from Sleman, Yogyakarta Province, Indonesia. Biodiversitas J. Biol. Diversity 24(7), 4087–4094. doi:10.13057/biodiv/d240747 Sophia, H.F., Supriyono, S., Soviana, S., Novianto, D. and Hadi, U.K. 2024. Molecular Detection of Borrelia Spp. (Spirochaetales: borreliaceae) in Ticks (Acari: Ixodidae) Collected from Tortoises in Java, Indonesia. Biodiversitas J. Biol. Diversity 24(12), 6852–6857. doi:10.13057/biodiv/d241246 Supriyono., Takano, A., Kuwata, R., Shimoda, H., Hadi, U.K., Setiyono, A., Agungpriyono, S. and Maeda, K. 2019. Detection and Isolation of Tick‐borne Bacteria ( Anaplasma Spp., Rickettsia Spp., and Borrelia Spp.) in Amblyomma Varanense Ticks on Lizard ( Varanus Salvator ). Microbiol. Immunol. 63(8), 328–333; doi:10.1111/1348-0421.12721 Utami, P. and Kunda, R.M. 2023. Surface Ultrastructure of Tick (Acari: ixodidae) on Moa Buffalo from Southwest Maluku District, Indonesia. Biodiversitas. J. Biol. Diversity. 24(6), 3230–3235. doi:10.13057/biodiv/d240617 Utami, P., Budianto, B.H. and Sahara, A. 2021. Tick (Acari: ixodidae) Infestation of Cuscuses from Maluku Province, Indonesia. Vet. World 1465, 1465–1471; doi:10.14202/vetworld.2021.1465-1471 Zhang, J.Z., Fan, M.Y., Wu, Y.M., Fournier, P.E., Roux, V. and Raoult, D. 2000. Genetic Classification of ‘Rickettsia Heilongjiangii’ and ‘Rickettsia Hulinii,’ Two Chinese Spotted Fever Group Rickettsiae. J. Clin. Microbiol. 38(9), 3498–3501; doi:10.1128/JCM.38.9.3498-3501.2000 | ||
| How to Cite this Article |
| Pubmed Style Hidayati L, Zulfanedi Y, Supriyono S, Ekawasti F, Viawan BD, Gusmiati R, Alwi NP, Thusadya H, Humaida B. Identification of ticks from conflict Sumatran tigers (Panthera tigris sumatrae) in Bukittinggi, West Sumatra, Indonesia. Open Vet. J.. 2026; 16(2): 1281-1288. doi:10.5455/OVJ.2026.v16.i2.45 Web Style Hidayati L, Zulfanedi Y, Supriyono S, Ekawasti F, Viawan BD, Gusmiati R, Alwi NP, Thusadya H, Humaida B. Identification of ticks from conflict Sumatran tigers (Panthera tigris sumatrae) in Bukittinggi, West Sumatra, Indonesia. https://www.openveterinaryjournal.com/?mno=285455 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.45 AMA (American Medical Association) Style Hidayati L, Zulfanedi Y, Supriyono S, Ekawasti F, Viawan BD, Gusmiati R, Alwi NP, Thusadya H, Humaida B. Identification of ticks from conflict Sumatran tigers (Panthera tigris sumatrae) in Bukittinggi, West Sumatra, Indonesia. Open Vet. J.. 2026; 16(2): 1281-1288. doi:10.5455/OVJ.2026.v16.i2.45 Vancouver/ICMJE Style Hidayati L, Zulfanedi Y, Supriyono S, Ekawasti F, Viawan BD, Gusmiati R, Alwi NP, Thusadya H, Humaida B. Identification of ticks from conflict Sumatran tigers (Panthera tigris sumatrae) in Bukittinggi, West Sumatra, Indonesia. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 1281-1288. doi:10.5455/OVJ.2026.v16.i2.45 Harvard Style Hidayati, L., Zulfanedi, . Y., Supriyono, . S., Ekawasti, . F., Viawan, . B. D., Gusmiati, . R., Alwi, . N. P., Thusadya, . H. & Humaida, . B. (2026) Identification of ticks from conflict Sumatran tigers (Panthera tigris sumatrae) in Bukittinggi, West Sumatra, Indonesia. Open Vet. J., 16 (2), 1281-1288. doi:10.5455/OVJ.2026.v16.i2.45 Turabian Style Hidayati, Lisa, Yoli Zulfanedi, Supriyono Supriyono, Fitrine Ekawasti, Bilan Diurai Viawan, Rita Gusmiati, Nike Puspita Alwi, Halima Thusadya, and Badriyatul Humaida. 2026. Identification of ticks from conflict Sumatran tigers (Panthera tigris sumatrae) in Bukittinggi, West Sumatra, Indonesia. Open Veterinary Journal, 16 (2), 1281-1288. doi:10.5455/OVJ.2026.v16.i2.45 Chicago Style Hidayati, Lisa, Yoli Zulfanedi, Supriyono Supriyono, Fitrine Ekawasti, Bilan Diurai Viawan, Rita Gusmiati, Nike Puspita Alwi, Halima Thusadya, and Badriyatul Humaida. "Identification of ticks from conflict Sumatran tigers (Panthera tigris sumatrae) in Bukittinggi, West Sumatra, Indonesia." Open Veterinary Journal 16 (2026), 1281-1288. doi:10.5455/OVJ.2026.v16.i2.45 MLA (The Modern Language Association) Style Hidayati, Lisa, Yoli Zulfanedi, Supriyono Supriyono, Fitrine Ekawasti, Bilan Diurai Viawan, Rita Gusmiati, Nike Puspita Alwi, Halima Thusadya, and Badriyatul Humaida. "Identification of ticks from conflict Sumatran tigers (Panthera tigris sumatrae) in Bukittinggi, West Sumatra, Indonesia." Open Veterinary Journal 16.2 (2026), 1281-1288. Print. doi:10.5455/OVJ.2026.v16.i2.45 APA (American Psychological Association) Style Hidayati, L., Zulfanedi, . Y., Supriyono, . S., Ekawasti, . F., Viawan, . B. D., Gusmiati, . R., Alwi, . N. P., Thusadya, . H. & Humaida, . B. (2026) Identification of ticks from conflict Sumatran tigers (Panthera tigris sumatrae) in Bukittinggi, West Sumatra, Indonesia. Open Veterinary Journal, 16 (2), 1281-1288. doi:10.5455/OVJ.2026.v16.i2.45 |