| Research Article | ||
Open Vet. J.. 2026; 16(1): 536-549 Open Veterinary Journal, (2026), Vol. 16(1): 536-549 Research Article Assessment of low-intensity pulsed ultrasound in enhancing alveolar bone repair and minimizing relapse in a rabbit modelOmar Alluazy1, Sarmad S. Salih Al Qassar1*, Ahmed Khalaf Ali2 and Mohammed Ghassan Saeed31Department of Pedodontics, Orthodontics and Prevention, College of Dentistry, University of Mosul, Mosul, Iraq 2Department of Surgery and Theriogenology, College of Veterinary Medicine, University of Mosul, Mosul, Iraq 3Department of Pathology and Poultry Diseases, College of Veterinary Medicine, University of Mosul, Mosul, Iraq *Corresponding Author: Sarmad S. Salih Al Qassar. Department of Pedodontics, Orthodontics and Prevention, College of Dentistry, University of Mosul, Mosul, Iraq. Email: sarmadsobhi [at] uomosul.edu.iq Submitted: 03/10/2025 Revised: 12/12/2025 Accepted: 27/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
AbstractBackground: The exogen device delivers low-intensity pulsed ultrasound (LIPUS), which stimulates osteogenesis and angiogenesis without causing harmful thermal effects. In veterinary practice, it can speed fracture healing in animals and horses, enhancing bone strength and shortening recovery time. Aim: This experimental study evaluated the veterinary application of the Exogen LIPUS device in promoting alveolar bone healing and reducing post-orthodontic relapse in rabbits. Methods: Twenty adult rabbits underwent orthodontic tooth movement, followed by appliance removal. Animals were divided into two groups: control group without LIPUS treatment and an Exogen group receiving daily LIPUS exposure for 20 days. Clinical measurements of relapse distance (RD) were recorded at days 0, 10, and 20. Histological and histomorphometric analyses were performed to assess osteoblast and osteoclast counts, vascular density, and periodontal ligament (PDL) width in both cervical and apical regions. Results: Results showed that the Exogen group exhibited a significantly smaller RD from day 10 onwards (p ≤ 0.01) and approximately 35% less relapse at day 20 compared with controls. Histomorphometric data revealed significant osteoblast counts, lower osteoclast counts, increased vascular density, and narrower PDL width in the Exogen group, indicating enhanced bone formation, reduced bone resorption, and improved periodontal stability. Histological examination confirmed denser osteoblastic lining and more pronounced vascularization in treated animals. Conclusion: These findings suggest that the Exogen LIPUS device may serve as an effective adjunct tool in veterinary medicine to accelerate bone healing—particularly alveolar bone—and to achieve greater stability of teeth following maxillofacial surgical interventions in rabbits, and potentially in other animals. Keywords: Bone healing, Low-intensity pulsed ultrasound, Relapse, Rabbits, Veterinary dentistry. IntroductionEnhancing alveolar bone healing is of paramount importance in veterinary dentistry and maxillofacial surgery, particularly in cases requiring rapid restoration of oral function to maintain masticatory efficiency, prevent malocclusion, and ensure overall animal welfare. In species such as rabbits, which serve both as research models to stimulate teeth movement in humans, post-treatment relapse of tooth alignment following orthodontic intervention or appliance removal is a common clinical challenge. This problem is also relevant in other veterinary patients, including horses, small ruminants, and companion animals such as dogs and cats, where loss or delayed healing of alveolar bone can compromise oral health, feeding efficiency, and long-term tooth stability (Ribka and Niemiec, 2022; Mosaddad et al., 2024; Patel et al., 2025). Consequently, there is growing interest in developing innovative adjunctive therapies to promote bone regeneration and preserve periodontal integrity in veterinary practice. Low-intensity pulsed ultrasound (LIPUS), as delivered by the Exogen device, has been shown to accelerate bone repair by enhancing osteoblastic activity, increasing expression of bone morphogenetic proteins (BMPs), and promoting angiogenesis without generating harmful thermal effects (Ball et al., 2023; Zhu et al., 2024). In veterinary medicine, these properties offer substantial benefits for the management of fractures and bone injuries in both small animals and equines, where rapid recovery is critical for restoring mobility and preventing secondary complications. In horses, where limb fractures and joint-associated bone injuries can severely impact performance and welfare, LIPUS may serve as a non-invasive adjunct to conventional fixation methods, potentially shortening healing time and improving bone strength (Hegmann et al., 2012). Studies in animal models and clinical veterinary cases have reported improved callus formation, enhanced mineralization, and reduced healing duration, supporting the potential translation of this technology to a wide range of orthopedic applications in domestic and performance animals. The ability of LIPUS to accelerate bone repair is largely attributed to its capacity to enhance osteoblast activity, stimulate osteogenic differentiation, and upregulate bone-specific growth factors such as BMP-2 and vascular endothelial growth factor (VEGF), while simultaneously promoting angiogenesis, thereby improving the mechanical and structural properties of the healing bone (Yan et al., 2016). In veterinary contexts, LIPUS has been successfully applied in long bone fracture repair in horses and small animals, while preclinical evidence in rabbits has demonstrated its ability to accelerate mandibular fracture healing, supporting its potential applicability in alveolar bone repair (Bigham-Sadegh and Oryan, 2015; Auer and Grainger, 2015). Within the scope of alveolar bone remodeling, animal studies—particularly those involving rodent models—indicate that LIPUS can modulate orthodontic tooth movement and post-treatment bone adaptation through molecular signaling pathways involving HGF/Runx2/BMP-2 and receptor activator of nuclear factor kappa-Β ligand (RANKL) expression, thus enhancing post-orthodontic bone stability (Krishnan et al., 2015; Toghranegar 2018). Although direct veterinary studies on LIPUS applications in dental contexts are limited, broader preclinical evidence supports its regenerative efficacy in tendon, ligament, and bone–soft tissue junction injuries across multiple domestic animal species, with demonstrated improvements in collagen organization, angiogenesis, and mechanical strength (Khanna et al., 2009; Lai et al., 2021). Collectively, these findings underscore the translational potential of LIPUS for clinical veterinary applications. Veterinary literature emphasizes that LIPUS differs from traditional continuous-wave therapeutic ultrasound in its low-intensity pulsed mode, which induces micromechanical effects at the cellular level rather than thermal effects. This mechanism activates integrin-mediated intracellular signaling, thereby stimulating osteoblast proliferation, extracellular matrix deposition, and vascular responses—key mechanisms in the healing of bone and periodontal supporting tissues in animals (Savva, n.d.; Man, n.d.). Building on this mechanistic rationale and the available preclinical evidence, the present study investigates the feasibility of using an Exogen-based LIPUS protocol to enhance alveolar bone remodeling and to mitigate post-orthodontic relapse following appliance removal in a rabbit model. The experimental rabbit model was selected for its anatomical and physiological similarities to small mammal dentition, allowing both quantitative histomorphometric analysis and qualitative histological evaluation to determine its potential as an adjunctive therapeutic modality in veterinary dental practice. Rabbits were selected as the experimental model due to their established use in preclinical orthodontic and craniofacial bone remodeling research. Despite the continuous eruption of rabbit incisors, this model offers several advantages, including rapid bone turnover, high sensitivity of the alveolar bone to mechanical stimuli, and suitability for histological and histomorphometric assessment. Importantly, relapse evaluation in rabbits is interpreted within a relative intra-animal framework rather than direct extrapolation to humans. Previous studies have demonstrated that, when standardized landmarks intra-observer reliability, and short observation intervals are applied, rabbit models provide reproducible and biologically meaningful insights into post-orthodontic alveolar bone adaptation and periodontal ligament (PDL) remodeling (Abdulhaddi et al., 2024). The primary objective of this research is to assess whether daily LIPUS application using the Exogen device substantially improves alveolar bone remodeling and reduces post-orthodontic relapse compared to a sham-treated (control) group in rabbits. Materials and MethodsThroughout the study, animals were monitored daily for signs of discomfort, appetite changes, or impaired mobility. A standard peri-operative analgesic protocol was followed, in which rabbits received routine postoperative pain management using veterinary-approved analgesics, and additional doses were administered if any animal showed signs of pain or distress. Humane endpoints were established to prevent unnecessary suffering, including criteria such as persistent weight loss, reduced activity, self-mutilation, or inability to feed. No animal reached any humane endpoint during the study. General anesthesia was induced using a combination of ketamine (35 mg/kg) and xylazine (5 mg/kg), administered intramuscularly. Postoperative analgesia was provided using meloxicam (0.2 mg/kg) for 3 consecutive days. Euthanasia was performed humanely by overdose of sodium pentobarbital in accordance with institutional and international animal welfare guidelines. Experimental design and sample sizeWe employed a randomized, controlled, parallel-arm preclinical trial using adult New Zealand White rabbits (Oryctolagus cuniculus), weighing 1.5–2.5 kg, selected for their well-established relevance in maxillofacial and bone regeneration studies (Ozawa et al., 2022). For achieving sufficient statistical power, we based our sample size estimate on previous rabbit mandibular LIPUS studies, which used approximately six animals per group (Shah et al., 2016). Accordingly, we allocated rabbits randomly into two groups: Control (sham application) and Exogen (LIPUS), with six rabbits per group. Animal housing and husbandryRabbits were individually housed in stainless-steel cages within a controlled environment (20°C–22°C; 12/12 hours light/dark cycle), with ad libitum access to food and water, and monitored daily to ensure welfare—a critical aspect of ARRIVE compliance. Orthodontic appliance and device removalEach rabbit received a clinically modeled orthodontic appliance placed on the mandibular central incisors’ general anesthesia, maintained for 14 days to simulate orthodontic tooth movement. Upon removal, baseline measurements (day 0) of incisors’ edge relapse were recorded. Orthodontic appliance design and force applicationOrthodontic tooth movement was induced using a custom-fabricated orthodontic appliance adapted for the mandibular incisors of rabbits. The appliance consisted of a stainless-steel orthodontic wire (0.014-inch diameter) configured as a closed-coil spring and secured between the two mandibular central incisors using light-cured composite resin. The appliance was calibrated to deliver a constant orthodontic force of approximately 25 g, verified using a tension gauge prior to placement. This magnitude of force was selected based on previous rabbit orthodontic models to induce controlled tooth movement while minimizing tissue trauma. The appliance remained in situ for 14 consecutive days, after which it was carefully removed under general anesthesia to initiate the relapse observation period. Intervention protocol• Control group: Received sham LIPUS application (device turned off) for 20 minutes daily. • Exogen group (LIPUS): Received daily LIPUS treatment via the Exogen device at a frequency of 1.5 MHz, spatial-average intensity 30 mW/cm², pulsed at 20% duty cycle (e.g., 200 µs on/800 µs off), for 20 minutes per day—parameters aligned with previous rabbit mandibular bone healing protocols (Puts, n.d.) Treatments commenced on the day of device removal (day 0) and continued daily until euthanasia. Timepoints and measurementsTimepoints and tissue samplingAnimals were followed clinically for relapse measurements across the 20-day post-removal period. For histological and radiographic assessments, mandibles were harvested at days 10 and 20 post-removal. The day 0 measurements were considered baseline clinical records obtained immediately after appliance removal and were not treated as an endpoint representing LIPUS exposure. The number of animals allocated to each endpoint was predefined to ensure balanced group representation at each sampling time. Randomization and blindingAllocation to groups was determined by computer-generated random numbers. Investigators performing measurements and tissue analysis were blinded to group assignments to minimize bias—a key component of high-quality animal research. Application of LIPUS (Exogen) on rabbit teethThe Exogen LIPUS device was applied according to the manufacturer’s guidelines, with modifications suitable for rabbit oral anatomy. After removal of the orthodontic appliance, the ultrasound transducer—equipped with a coupling gel to ensure optimal acoustic transmission—was positioned extraorally over the alveolar bone region corresponding to the treated teeth. The handpiece was held in a stable position using a custom-fabricated support to prevent movement during the session. Each application consisted of a 20-minute continuous exposure at the preset output parameters of the device (1.5 MHz frequency, 200 μs pulse duration, 1 kHz repetition rate, and 30 mW/cm² spatial average temporal average intensity). Treatments were administered once daily for the designated post-removal observation period, ensuring that the coupling gel was replenished if any drying occurred during the session to maintain consistent ultrasound transmission (Fig. 1).
Fig. 1. The Exogen® LIPUS device (Bioventus LLC, Durham, NC) used for daily treatment in the experimental group. The device delivers 1.5 MHz pulsed ultrasound at an intensity of 30 mW/cm² for 20 minutes per session. EXOGEN ultrasound enhances upregulation of the processes that are critical to bone repair (Fig. 2).
Fig. 2. Steps of application of bone stimulation (Device Exogen). Relapse measurementRelapse was monitored over a 20-day post-removal period using an electronic digital vernier caliper (accuracy: 0.01 mm). The inter-incisal space was recorded at the mesial incisal tips of the two mandibular incisors at day 0 (immediately after appliance removal), and days 2, 4, 7, 10, 13, 17, and 20, following previously reported protocols (Naruse et al., 2010; Al-Fakhry and Al-Sayagh, 2022). To reduce measurement variability related to the curved incisor profile, the same anatomical landmarks were consistently used at each time point, and measurements were performed under standardized head positioning. Relapse distance (RD) was calculated as the change in space relative to baseline as RD=S1−S0, where S0 is the baseline space at day 0, and S1 is the space at the specified follow-up day. RD was additionally expressed as a percentage of S0 to enable standardized comparison between animals. Digital radiographical measurementDigital radiographic assessment was performed using Carestream Imaging Software (v7.0.3) to evaluate bone density in the para-symphysis region of the right mandibular cancellous bone between the alveolar crest and bifurcation. Measurements were expressed in gray values (0–255 GV) and analyzed using standardized coronal and apical reference points (Pollard and Phillips, 2017). Radiographic recordings employed a size 1 dental digital sensor (RVG 5200, Carestream Dental LLC). Method of radiographyThe jaw sample was placed on the sensor with its lingual part touching the sensor surface sleaved and the cone of the dental X-ray machine (Getidy intraoral X-ray system, tube). The source image distance was about 20 cm so that the central ray directed in perpendicular direction to sample surface which was parallel to sensor surface that in contact with it. Tube model K 127-0.8-70, settings were 70 kv and 7 mA with exposure time 0.30 as in Figure 5A and B. Radiographic bone density assessment is shown in Figure 4.
Fig. 4. (A): digital bone density assessment of target area of the rabbit mandibular jaw. (B): RVG 5,200 dental sensor (size 1); TROPHY,4, rue F. pelloutier, Croissy-Beabourg, 77,435 Marne La Vallee Cedex 2, France Carestream Dental LLC, Cumberland Boulevard, Suite 700, Atlanta, GA USA.
Fig. 5. Shows dental intraoral X-ray machine with target area for rabbit lower jaw imaging technique (A) and control panel of Getidy intraoral X-ray machine (B). Histological assessmentTissue samples were fixed in 10% neutral buffered formalin, dehydrated through graded alcohols, and embedded in paraffin. Sections of 4–5 μm thickness were prepared and stained with hematoxylin and eosin (H&E) for general morphology, while special stains were applied when required to highlight connective tissue or other structures. The stained slides were examined under a light microscope to evaluate tissue architecture, cellular morphology, and pathological alterations such as inflammatory infiltration, fibrosis, and vascular changes. Where applicable, morphometric measurements and semi-quantitative scoring were performed to provide an objective evaluation of histological changes. The relapse measurement procedure is illustrated in Figure 3.
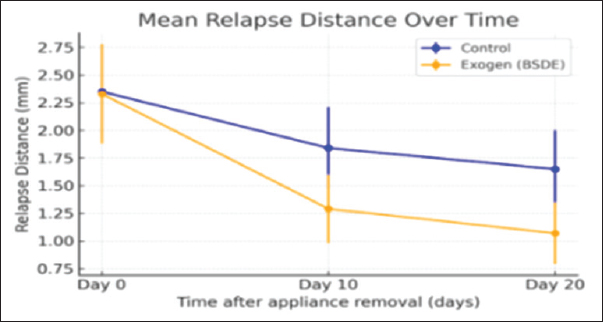
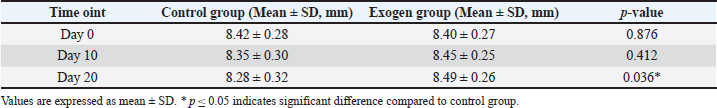
Fig. 3. Measurement of the relapse amount. Statistical analysisData normality (Shapiro–Wilk) and homogeneity of variances (Levene) were verified. The primary outcome (relapse over time) was analyzed using a linear mixed-effects model (group × time, rabbit as a random effect). Secondary outcomes (radiographic density, histomorphometric parameters) were analyzed via two-way ANOVA (group × time) with Bonferroni or Holm corrections for multiple comparisons. Post-hoc comparisons were performed using the Bonferroni correction to identify significant pairwise differences between groups at each time point. We reported effect sizes (partial η²) and 95% confidence intervals. Statistical significance was set at p < 0.05. Effect sizes were calculated to complement p-values and to quantify the magnitude of observed differences, thereby providing a more comprehensive interpretation of treatment effects beyond statistical significance alone. Ethical approvalThis experimental protocol was approved by the Institutional Animal Care and Use Committee (IACUC) of the College of Dentistry, University of Mosul (REC reference no. UoM.Dent. 24/1025) on April 21, 2024, in full compliance with ARRIVE 2.0 guidelines to ensure robustness, transparency, and reproducibility in animal research. All procedures were performed in accordance with ARRIVE 2.0 guidelines and under the approval of the Institutional Animal Care and Use Committee (IACUC). ResultsClinical relapse measurementsAt baseline (day 0, immediately after appliance removal), the mean RD was comparable between the control and Exogen groups, confirming successful randomization p > 0.05. Over the observation period, both groups exhibited a gradual reduction in RD; however, the Exogen group demonstrated a significantly lower mean relapse from day 10 onwards compared with controls (p < 0.01). By day 20, the mean relapse in the Exogen group was reduced by approximately 35% relative to controls, indicating a clear benefit in maintaining post-orthodontic tooth position (Table 1, Fig. 6).
Fig. 6. Line graph showing RD over time for both groups. Table 1. Mean ± SD of clinical RD (mm) in control and exogen groups at days 0, 10, and 20 after orthodontic appliance removal.
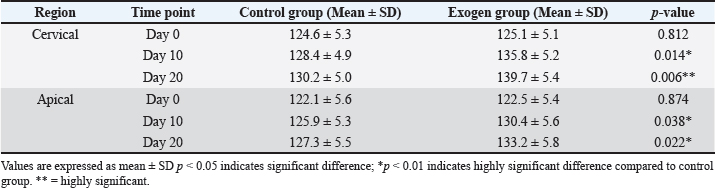
Although relapse was recorded at multiple follow-up points (days 0, 2, 4, 7, 10, 13, 17, and 20), the main comparative summaries are reported at days 0, 10, and 20 to align with the planned observation windows and to provide clinically interpretable checkpoints. Radiographic bone densityQuantitative radiographic analysis revealed progressive increases in relative alveolar bone density over time in both groups. While no significant difference was observed at day 0, the Exogen group exhibited significantly higher bone density values at both day 10 and 20 (p < 0.05), with the most pronounced difference recorded at day 20. Region-specific analysis showed that the cervical alveolar region responded more rapidly to LIPUS stimulation, displaying an earlier increase in density compared to the apical region, as shown in Table 2. Table 2. Relative alveolar bone density (mean ± SD) in cervical and apical regions for control and Exogen groups at days 0, 10, and 20 after orthodontic appliance removal.
Alveolar ridge heightMeasurements of alveolar ridge height indicated preservation of structural dimensions in the Exogen group throughout the study period, in contrast to a mild but statistically significant reduction in ridge height observed in the control group by day 20 (p < 0.05). This finding suggests that LIPUS may contribute to minimizing post-treatment alveolar resorption. Histological findingsAt day 10, specimens from the exogen-treated group predominantly exhibited woven bone and osteoid-rich trabeculae, reflecting active early bone formation rather than fully mature lamellar architecture. By day 20, specimens from this group revealed a well-organized lamellar bone matrix, a continuous and dense osteoblastic lining along the trabecular surfaces, and rich vascular networks permeating the marrow spaces—features consistent with accelerated and well-coordinated bone remodeling. Alveolar ridge height measurements are summarized in Table 3. Table 3. Mean ± SD of alveolar ridge height (mm) in control and exogen groups at days 0, 10, and 20 after orthodontic appliance removal.
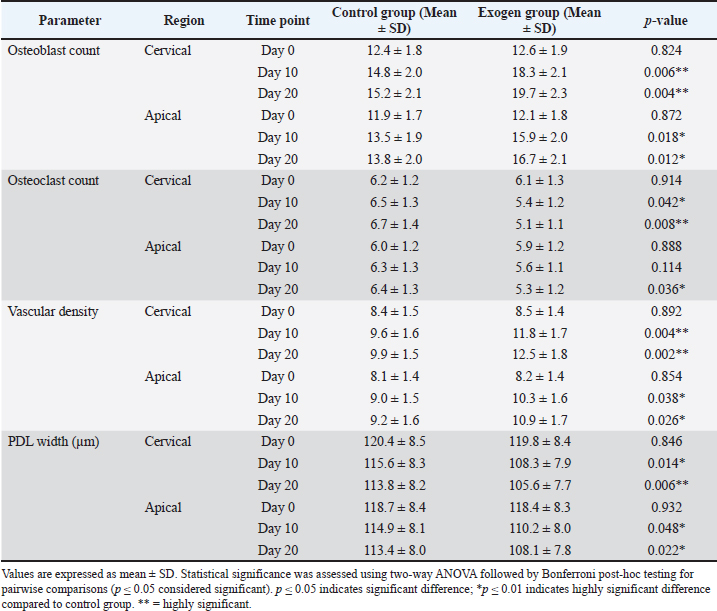
In contrast, the control group at comparable time points displayed a more disorganized trabecular pattern, wider marrow cavities, and a notably reduced osteoblast population, suggesting slower bone regeneration and less structural maturation. Detailed histomorphometric parameters are presented in Table 4. Table 4. Histomorphometric parameters (mean ± SD) in cervical and apical alveolar bone regions for control and exogen groups at days 0, 10, and 20 after orthodontic appliance removal.
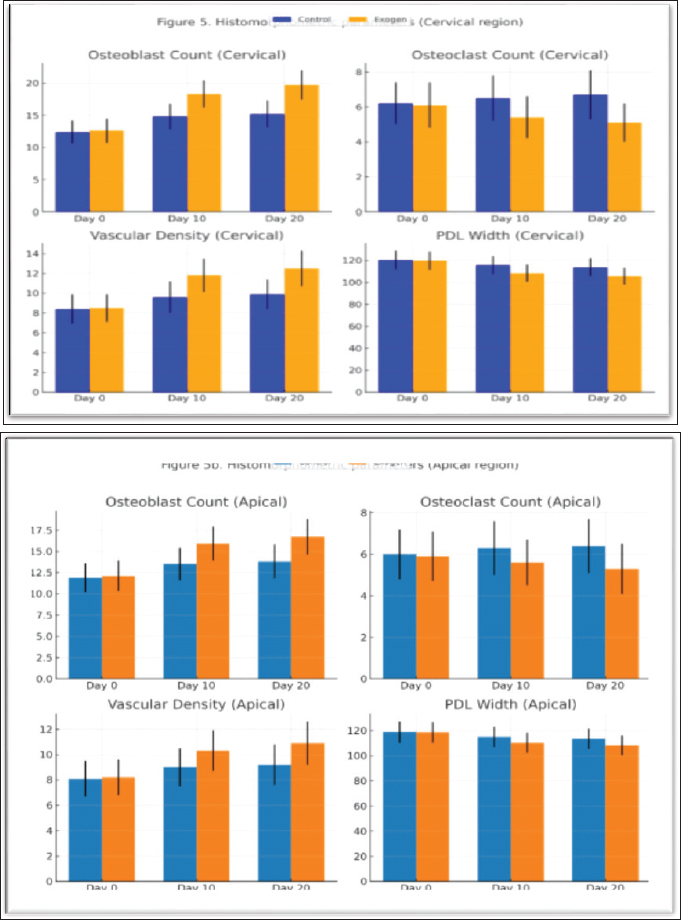
Histomorphometric analysisBased on the histomorphometric findings, exogen treatment markedly enhanced bone-forming activity, as evidenced by a statistically significant increase in osteoblast count at both day 10 and 20 compared with the control group (p < 0.01), indicating accelerated bone formation. In contrast, osteoclast count was significantly reduced at day 20 (p < 0.05), reflecting suppression of bone resorption. Vascular density showed a consistent and significant increase across all post-removal time points (p < 0.01), providing a more favorable blood supply for bone and periodontal tissue healing. Additionally, PDL width was narrower in the Exogen group by day 20, suggesting enhanced tooth stability after orthodontic appliance removal. Representative histological sections at day 10 are shown in Figures 7 and 8.
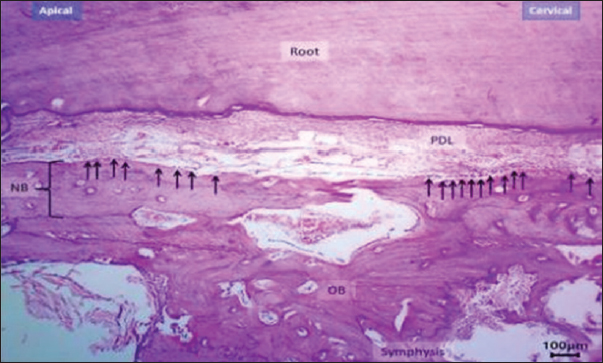
Fig. 7. Histological section of the exogen-treated group at day 10 showing a broad zone of newly formed bone (NB) with aligned osteoblasts (arrows). H&E stain, 40×, scale bar=100 μm.
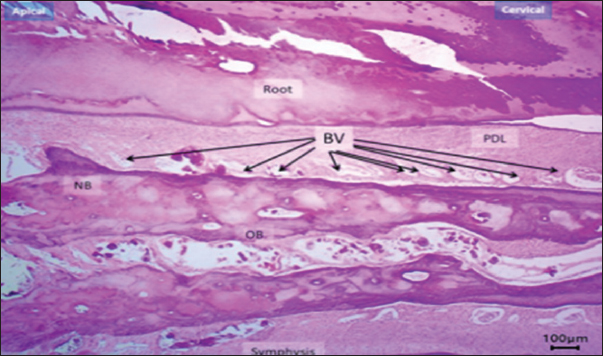
Fig. 8. Histological section at day 10 (non-Exogen group) showing multiple blood vessels (BV) within newly formed bone (NB). H&E stain, 40×, scale bar=100 μm. This pattern of results translates into accelerated healing of bone and periodontal tissues with an improved vascular environment, supporting the hypothesis that LIPUS can reduce post-treatment tooth relapse and promote long-term stability (Fig. 9a and b). New bone surface distribution is demonstrated in Figure 10.
Fig. 9. Histological section of the control group at day 20 showing disorganized new bone (NB), widened PDL space, and multiple osteoclasts (arrows). H&E stain, 40×, scale bar=100 μm.
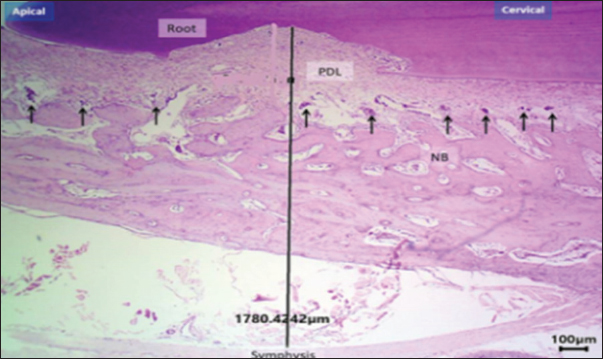
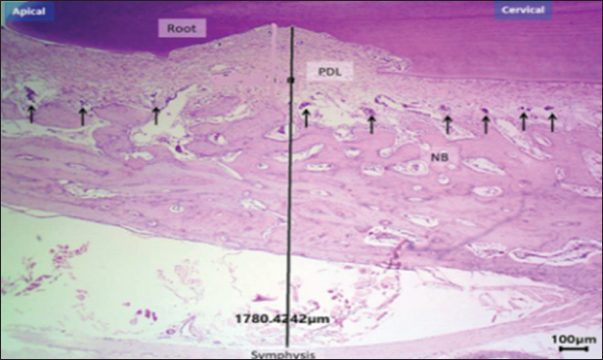
Fig. 10. H&E –stained sections representing new bone surface area in control and exogen groups along cervical and apical lines at day 0. H&E, 100×, scale bar=100 μm. DiscussionThe present findings underscore the significant veterinary potential of LIPUS, as delivered by the exogen device, in promoting bone regeneration and minimizing post-treatment relapse. While the current model focused on alveolar bone in rabbits, the underlying mechanisms and observed outcomes have broader implications for veterinary orthopedics and maxillofacial surgery across multiple animal species. In animals—particularly performance horses, companion dogs, and small ruminants—rapid osseous repair is essential for restoring function, preventing chronic disability, and maintaining welfare standards (Naruse et al., 2010; Al-Fakhry and Al-Sayagh, 2022). Mechanistically, LIPUS exerts its regenerative effect through micromechanical stimulation of bone cells, resulting in integrin-mediated activation of intracellular signaling cascades, including the focal adhesion kinase pathway and downstream transcription factors such as Runx2. Recent findings on orthodontic relapse support the present observations. Mohammed et al. (2023) reported that enhancing osteoblastic activity and vascular supply in rabbit incisors significantly reduced relapse after appliance removal—a pattern closely aligned with the current study, where LIPUS-treated animals showed narrower PDL width and higher osteoblast counts. Similarly, Al-Fakhry and Al-Sayagh (2022)demonstrated that biological stimulation accelerates post-movement alveolar remodeling and increases tissue stiffness, thereby limiting tooth rebound. These parallels reinforce the interpretation that the reduced RD observed in the Exogen group arises from an accelerated shift toward bone formation and improved periodontal structural integrity. This activation promotes osteoblastic proliferation, differentiation, and the upregulation of BMP-2, VEGF, and other osteogenic mediators (Nielsen, 2023; Mohammed et al., 2023). In parallel, LIPUS reduces osteoclastic activity by modulating the RANKL/osteoprotegerin balance, thereby suppressing bone resorption. The current study's histomorphometric data—showing increased osteoblast counts, reduced osteoclast numbers, and enhanced vascular density—are consistent with these molecular pathways supporting the histomorphometric outcomes observed (Fig. 11a and b).
Fig. 11. (a). Histomorphometric parameters (cervical region): osteoblast count, osteoclast count, vascular density, and PDL width at days 0, 10, and 20 in control and exogen groups. (b). Histomorphometric parameters (Apical region): osteoblast count, osteoclast count, vascular density, and PDL width at days 0, 10, and 20 in control and exogen groups. Comparable outcomes have been documented in preclinical models beyond the oral cavity. Comparable outcomes have been reported in animal models evaluating alveolar bone regeneration. A systematic analysis by Lai et al. (2021) demonstrated that LIPUS promotes early osteoid deposition and improves trabecular orientation in craniofacial bone defects, findings consistent with the more organized lamellar bone pattern detected in our exogen-treated specimens by day 20. Likewise, Hatt et al. (2022) emphasized the role of angiogenesis in stabilizing early bone formation in cranio-maxillofacial models, supporting our observation of significantly increased vascular density in both cervical and apical regions. Together, these studies confirm that LIPUS enhances orchestrated tissue maturation rather than merely accelerating mineral deposition. For instance, in equine long bone fracture models, daily LIPUS application improved callus mineralization and mechanical strength, shortening convalescence by up to 30% (Reis et al., 2024). Similarly, canine studies have reported accelerated union in radial and tibial fractures, with improved radiographic density and histological maturity of bone tissue (Alas et al., 2025). These outcomes align with the present findings and strengthen the rationale for species-specific veterinary adoption. Furthermore, comparable findings have been reported in large-animal veterinary contexts. In equine patients, LIPUS has been applied to metacarpal and metatarsal fractures, yielding faster cortical bridging and improved callus quality, thereby reducing stall rest duration and risk of secondary complications (Mitchell and Bertorini, 2010; Khedir et al., 2023). More recent literature (2023–2025) reinforces these mechanistic findings. Reis et al. (2024)demonstrated that LIPUS significantly improves cortical bridging and callus mineralization in equine musculoskeletal injuries, highlighting its translational relevance for veterinary orthopedics. Similarly, Alas et al. (2025)documented enhanced radiographic bone union in canine tibial injuries treated with ultrasound-assisted rehabilitation. These contemporary studies parallel the current results, indicating that the Exogen device not only accelerates cellular events such as osteoblast differentiation but also improves macro-level outcomes like structural bone preservation and relapse reduction. In canine models, LIPUS treatment of radial and ulnar fractures has been associated with earlier radiographic union and superior histological bone maturity compared to standard fixation alone (Pollard and Phillips, 2017). Similarly, studies in ovine tibial defects have demonstrated enhanced mineral apposition rates and improved biomechanical strength following LIPUS application (Reichert et al., 2010). These cross-species outcomes reinforce the present study's findings, suggesting that the mechanisms observed in rabbits are consistent with those operating in other domestic and performance animals, supporting broader clinical translation. The translational relevance of the rabbit model lies in its similarity to small mammal bone physiology, allowing extrapolation to companion animal practice. However, differences in cortical-to-trabecular bone ratios, vascular supply, and mechanical loading patterns across species necessitate careful parameter optimization. For example, while the present study employed 1.5 MHz at 30 mW/cm² for 20 minutes daily, equine limb bone applications may require adjustments in transducer positioning and coupling to account for tissue depth and bone geometry (Leighton et al., 2021). An additional clinically relevant finding was the preservation of alveolar ridge height in LIPUS-treated animals, indicating its capacity to counteract post-intervention resorption. In veterinary orthopedics, similar principles apply to preventing bone loss following fracture fixation or limb surgery, where maintenance of bone volume directly impacts functional prognosis. Moreover, the significant increase in vascular density observed in this study supports the use of LIPUS in cases complicated by compromised perfusion, such as open fractures or wounds with periosteal stripping (Hatt et al., 2022; Verduzco-Mendoza and Olmos-Hernández, 2025). From a broader veterinary perspective, LIPUS presents several advantages: non-invasive application reduces stress and morbidity in animal patients compared to surgical interventions (Cordelle et al., 2025). It also shows species versatility, with potential application in mammals and possibly avian orthopedics, given adequate anatomical adaptation of the device (Bhattacharjee and Sharma, 2025). Additionally, LIPUS has field utility in large animals, especially horses, where portable ultrasound units can be deployed in stables to deliver daily treatments without hospitalization (Samee et al., 2008; Alkhayat et al., 2023). Although alveolar bone is unique in its rapid turnover and high remodeling sensitivity, the biological response observed here mirrors patterns reported in long-bone healing. Leighton et al. (2021) and Nielsen (2023)emphasized that LIPUS enhances integrin-mediated mechanotransduction and upregulates osteogenic markers across skeletal sites. This mechanistic similarity suggests that the accelerated alveolar healing documented in rabbits may be part of a broader LIPUS-mediated biological response applicable to multiple bone types, supporting its wider veterinary implementation. Although advanced immunohistochemical markers such as Runx2, osteocalcin, and TRAP staining were not employed in the present study, the combined use of quantitative histomorphometry (osteoblast and osteoclast counts, vascular density, and PDL width) alongside radiographic and clinical relapse data provided a multi-level assessment of bone remodeling. Future investigations should incorporate molecular and immunohistochemical analyses to further elucidate the underlying signaling pathways involved in LIPUS-mediated alveolar regeneration. These mechanisms are consistent with previous veterinary and experimental findings (Nielsen, 2023; Ozawa et al., 2022; Patel et al., 2025; Meduri, 2023). In conclusion, the present study demonstrates that daily application of LIPUS via the Exogen device significantly accelerates bone regeneration, enhances vascularization, and reduces post-treatment relapse in a rabbit alveolar bone model. These results, supported by comparable findings in diverse animal models, indicate strong potential for incorporating LIPUS into veterinary protocols for fracture management, post-extraction bone preservation, and maxillofacial reconstruction. Future research should prioritize large-animal trials, optimization of dosing regimens for different skeletal sites, and long-term assessments of functional outcomes to ensure the safe and effective translation of this technology across veterinary species. ConclusionThese findings suggest that LIPUS may represent a promising adjunctive approach for enhancing alveolar bone remodeling and reducing post-orthodontic relapse under controlled experimental conditions. Treated animals showed significantly reduced RD, higher bone density, increased osteoblast activity, enhanced vascularization, and narrower PDL width, collectively reflecting accelerated bone remodeling and improved post-treatment stability. These outcomes highlight the potential value of LIPUS as a non-invasive adjunct in veterinary maxillofacial and orthopedic practice, particularly in managing alveolar bone loss and supporting postoperative recovery. Further investigation in larger animal models is warranted to optimize dosing parameters and validate long-term clinical applicability across species. AcknowledgmentsThe authors would like to express their gratitude to the College of Dentistry, University of Mosul, for providing laboratory facilities and technical assistance during the experimental procedures. Conflict of interestThe authors declare that there is no conflict of interest regarding the publication of this research. FundingThis research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. Authors' contributionsAll authors contributed equally to the conception, design, experimentation, data analysis, and manuscript preparation. Data availabilityThe datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request. ReferencesAbdulhaddi, A., Qassar, A., Salih, S.S. and Mohammed, A.M., 2024. Assessment of the mechanical properties and antimicrobial efficiency of orthodontic adhesive modified with Salvadora Persica oil. Romanian J. Stomatol. 70(2). Alkhayat, Z.I., Al Qassar, S.S.S. and Qasim, A.A., 2023. The effect of the static magnetic field on some of the mechanical properties of glass ionomer cements. Rom J Stomatol, 69(4), pp.224-29. Alas, O., Gallastegui, A., Hernandez, J. and Johnson, M. 2025. Comprehensive radiographic grading system and clinical outcomes of canine tibial avulsion fractures in dogs. Vet. Radiol. Ultrasound. 66, e13473. Al-Fakhry, H.H. and Al-Sayagh, N.M. 2022. Effects of injectable platelet rich fibrin (i-PRF) on reduction of relapse after orthodontic tooth movement: rabbits model study. J. Orthod. Sci. 11, 10. Auer, J.A. and Grainger, D.W. 2015. Fracture management in horses: where have we been and where are we going?. Vet. J. 206(5), 5–14. Ball, J.R., Shelby, T., Hernandez, F., Mayfield, C.K. and Lieberman, J.R. 2023. Delivery of growth factors to enhance bone repair. Bioengineering 10, 1252. Bhattacharjee, U. and Sharma, I. 2025. Integrating modern modalities for advancements in veterinary medicine. In: Biofilm associated livestock diseases and their management. Cham, Switzerland: Springer, pp: 265–307. Bigham-Sadegh, A. and Oryan, A. 2015. Selection of animal models for pre-clinical strategies in evaluating fracture healing, bone graft substitutes and bone tissue regeneration. Connect. Tissue. Res. 56, 175–194. Cordelle, M.Z., Snelling, S.J.B. and Mouthuy, P.A. 2025. Skeletal muscle tissue engineering: from tissue regeneration to biorobotics. Cyborg. Bionic. Syst. 6(6), 279. Hatt, L.P., Thompson, K., Helms, J.A., Stoddart, M.J. and Armiento, A.R. 2022. Clinically relevant animal models for testing novel cranio-maxillofacial biomaterials. Clin. Transl. Med. 12, 690. Hegmann, K.T., Hoffman, H.E., Belcourt, R.M., Byrne, K., Glass, L. and Melhorn, J.J. 2012. Elbow disorders. In Occupational medicine practice guidelines. ACOEM, pp: 1–69. Khanna, A., Nelmes, R.T., Gougoulias, N., Maffulli, N. and Gray, J. 2009. Effects of LIPUS on soft-tissue healing: a review. Br. Med. Bull. 89, 169–182. Khedir, R., Taqa, G. and Al Qassar, S. 2023. Evaluating systemic effect of magnesium oxide on gene expression of osteocalcin and vitamin D receptors in rabbits. Egypt. J. Vet. Sci. 54, 71–86. Krishnan, V., Zahrowski, J.J. and Davidovitch, Z.E. 2015. The effect of drugs and diet on orthodontic tooth movement. In Biological mechanisms of tooth movement. Wiley, pp: 173–87. Lai, W.C., Iglesias, B.C., Mark, B.J. and Wang, D. 2021. Low-intensity pulsed ultrasound augments tendon, ligament, and bone-soft tissue healing. Arthroscopy 37, 2318–2333. Leighton, R., Phillips, M., Bhandari, M. and Zura, R. 2021. LIPUS for management of non-unions: systematic review. BMC. Musculoskelet. Disord. 22, 532. Man, J.S. Biological effects of low frequency ultrasound on bone and tooth cells. PhD thesis, University of Birmingham, Birmingham, UK. Meduri, C. 2023. Focused ultrasound methods for the treatment of tendon injuries. PhD Thesis. Milan, Italy: University of Milan. Mitchell, M. and Bertorini, T. 2010. Ultrasound stimulation in equine fracture healing. Veterinary Orthopedics and Traumatology. 23(4), 210–218. Mohammed, R.E., Al Qassar, S.S. and Taqa, G.A. 2023. Clinical and histological evaluation of magnesium oxide on relapse after orthodontic movement. J. Orthod. Sci. 12, 19. Mosaddad, S.A., Hussain, A. and Tebyaniyan, H. 2024. Exploring animal models in craniofacial regenerative medicine. Tissue. Eng. Part. B. 30, 29–59. Naruse, K., Sekiya, H., Harada, Y., Iwabuchi, S., Kozai, Y., Kawamata, R., Kashima, I., Uchida, K., Urabe, K., Seto, K., Itoman, M. and Mikuni-Takagaki, Y. 2010. Prolonged endochondral bone healing. Ultrasound Med Biol. 236, 1098–1108. Nielsen, B.D. 2023. Review of research dedicated to equine bone strength. Animals 13, 789. Ozawa, S., Thomson, A. and Petritz, O. 2022. Safety and efficacy of oral mirtazapine in rabbits. J. Exotic. Pet. Med. 40, 16–20. Patel, R., Chauhan, T., Srivastava, S., Mishra, P. and Jain, P. 2025. Parasitism in farm animals. In Prospects of fungal biotechnologies for livestock. Springer, pp: 1–42. Pollard, R.E. and Phillips, K.L. 2017. Orthopedic diseases of young dogs and cats. In: Textbook of Veterinary Diagnostic Radiology. Ed., Thrall, D.E. St. Louis, MO: Elsevier, 348–365. Puts, R. 2023. Physico-biological mechanisms of focused LIPUS in musculoskeletal regeneration. PhD Thesis. Utrecht, Netherlands: Utrecht University. Reichert, J.C., Epari, D.R., Wullschleger, M.E., Saifzadeh, S., Steck, R., Lienau, J., Sommerville, S., Dickinson, I.C., Schütz, M.A., Duda, G.N. and Hutmacher, D.W. 2010. Establishing a preclinical ovine model for tibial segmental bone defect repair by applying bone tissue engineering strategies. Tissue Eng. Part B: Rev. 16(1), 93–104. Reis, I.L., Lopes, B., Sousa, P., Ferreira, F., Oliveira, J.M., Silva, T.H. 2024. Equine musculoskeletal pathologies: clinical perspectives and regenerative approaches. Vet. Sci. 11(4), 190. Ribka, E.P. and Niemiec, B.A. 2022. Diseases of the oral cavity and teeth. In Clinical Medicine of the Dog and Cat. Eds., Silverstein, D.C. and Hopper, K. Boca Raton, FL: CRC Press, 163–187. Samee, M., Kasugai, S., Kondo, H., Ohya, K., Shimokawa, H. and Shimizu, Y. 2008. Bone morphogenetic protein-2 and vascular endothelial growth factor transfection enhances osteoblast differentiation and mineralization. J. Pharmacol. Sci. 108(1), 18–31. Savva, J. Effects of low intensity pulsed ultrasound on bone cells using controlled in vitro exposure. PhD thesis, University of Glasgow, Glasgow, UK. Shah, S.R., Young, S., Goldman, J.L., Jansen, J.A., Wong, M.E. and Mikos, A.G. 2016. A composite rabbit mandibular defect model for evaluation of craniofacial bone regeneration strategies. Nat. Protocols 11(10), 1989–2009. Toghranegar, S. 2018. Minimally invasive accelerated orthodontic techniques: rat model. PhD thesis, Nova Southeastern University, Davie, FL. Verduzco-Mendoza, A. and Olmos-Hernández, A. 2025. Thermal imaging applications in biomedical and veterinary research: current advances. Front. Vet. Sci. 12, 1544112. Yan, H., Liu, X., Zhu, M., Li, Y., Zhang, J. and Wang, L. 2016. Hybrid delivery of growth factors combined with low-intensity pulsed ultrasound enhances bone regeneration in composite scaffolds. J. Biomed. Mat. Res. Part A. 104(1), 195-208 Zhu, J., Wang, Y., Liu, X. and Zhang, H. 2024. Low-intensity pulsed ultrasound promotes bone regeneration through angiogenic and osteogenic signaling pathways. Ultrasound Med. Biol. 250(3), 455–468. | ||
| How to Cite this Article |
| Pubmed Style Alluazy O, Qassar SSSA, Ali AK, Saeed MG. Assessment of low-intensity pulsed ultrasound in enhancing alveolar bone repair and minimizing relapse in a rabbit model. Open Vet. J.. 2026; 16(1): 536-549. doi:10.5455/OVJ.2026.v16.i1.51 Web Style Alluazy O, Qassar SSSA, Ali AK, Saeed MG. Assessment of low-intensity pulsed ultrasound in enhancing alveolar bone repair and minimizing relapse in a rabbit model. https://www.openveterinaryjournal.com/?mno=288101 [Access: February 06, 2026]. doi:10.5455/OVJ.2026.v16.i1.51 AMA (American Medical Association) Style Alluazy O, Qassar SSSA, Ali AK, Saeed MG. Assessment of low-intensity pulsed ultrasound in enhancing alveolar bone repair and minimizing relapse in a rabbit model. Open Vet. J.. 2026; 16(1): 536-549. doi:10.5455/OVJ.2026.v16.i1.51 Vancouver/ICMJE Style Alluazy O, Qassar SSSA, Ali AK, Saeed MG. Assessment of low-intensity pulsed ultrasound in enhancing alveolar bone repair and minimizing relapse in a rabbit model. Open Vet. J.. (2026), [cited February 06, 2026]; 16(1): 536-549. doi:10.5455/OVJ.2026.v16.i1.51 Harvard Style Alluazy, O., Qassar, . S. S. S. A., Ali, . A. K. & Saeed, . M. G. (2026) Assessment of low-intensity pulsed ultrasound in enhancing alveolar bone repair and minimizing relapse in a rabbit model. Open Vet. J., 16 (1), 536-549. doi:10.5455/OVJ.2026.v16.i1.51 Turabian Style Alluazy, Omar, Sarmad S. Salih Al Qassar, Ahmed Khalaf Ali, and Mohammed Ghassan Saeed. 2026. Assessment of low-intensity pulsed ultrasound in enhancing alveolar bone repair and minimizing relapse in a rabbit model. Open Veterinary Journal, 16 (1), 536-549. doi:10.5455/OVJ.2026.v16.i1.51 Chicago Style Alluazy, Omar, Sarmad S. Salih Al Qassar, Ahmed Khalaf Ali, and Mohammed Ghassan Saeed. "Assessment of low-intensity pulsed ultrasound in enhancing alveolar bone repair and minimizing relapse in a rabbit model." Open Veterinary Journal 16 (2026), 536-549. doi:10.5455/OVJ.2026.v16.i1.51 MLA (The Modern Language Association) Style Alluazy, Omar, Sarmad S. Salih Al Qassar, Ahmed Khalaf Ali, and Mohammed Ghassan Saeed. "Assessment of low-intensity pulsed ultrasound in enhancing alveolar bone repair and minimizing relapse in a rabbit model." Open Veterinary Journal 16.1 (2026), 536-549. Print. doi:10.5455/OVJ.2026.v16.i1.51 APA (American Psychological Association) Style Alluazy, O., Qassar, . S. S. S. A., Ali, . A. K. & Saeed, . M. G. (2026) Assessment of low-intensity pulsed ultrasound in enhancing alveolar bone repair and minimizing relapse in a rabbit model. Open Veterinary Journal, 16 (1), 536-549. doi:10.5455/OVJ.2026.v16.i1.51 |