| Research Article | ||
Open Vet. J.. 2026; 16(1): 136-146 Open Veterinary Journal, (2026), Vol. 16(1): 136-146 Review Article Effect of mangosteen peel extract nanocapsule on the hematological profile and total bacterial count in broiler chickens experimentally infected with avian pathogenic Escherichia coliAndri Kusmayadi1*, Ristina Siti Sundari2, Endang Sujana3, and Yanti Yanti41Department of Animal Science, Faculty of Agriculture, Universitas Perjuangan Tasikmalaya, Tasikmalaya, Indonesia 2Department of Agribusiness, Faculty of Agriculture, Universitas Perjuangan Tasikmalaya, Tasikmalaya, Indonesia 3Department of Animal Science, Faculty of Animal Science, Universitas Padjadjaran, Sumedang, Indonesia 4Department of Mechatronics Engineering, Faculty of Engineering, Universitas Mayasari Bakti, Tasikmalaya, Indonesia *Corresponding Author: Andri Kusmayadi, Department of Animal Science, Faculty of Agriculture, Universitas Perjuangan Tasikmalaya, Tasikmalaya, Indonesia. Email: andrikusmayadi [at] unper.ac.id Submitted: 09/10/2025 Revised: 12/12/2025 Accepted: 23/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
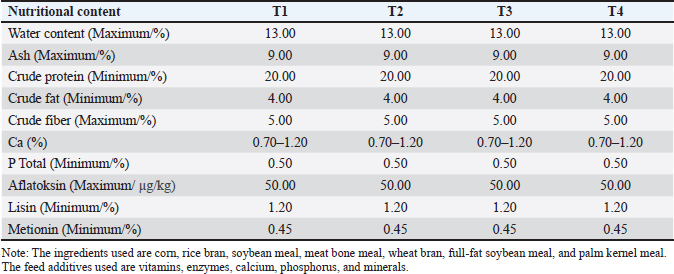
AbstractBackground: Infection by avian pathogenic Escherichia coli (APEC) in broiler chickens leads to serious health complications, including systemic inflammation, oxidative stress, and impaired immune response. Aim: This study aimed to evaluate the effect of mangosteen peel extract nanocapsule (MPE-NC) on the hematological profiles and total bacteria in APEC-infected broilers. Methods: A total of 200 day old chick broiler chickens were randomly allocated to four treatments with five replicates, including T1 (negative control group), fed a basal diet without APEC infection; T2 (positive control group), fed a basal diet with APEC infection; T3: a basal diet supplemented with 0.5% MPE-NC and APEC infection; and T4: a basal diet supplemented with 1.0% MPE-NC and APEC infection. The observed parameters were the hematological blood profile and total bacterial count. Results: APEC infection significantly (p < 0.05) impaired the physiological status of broiler chickens by reducing hematological parameters and had a significant effect on total bacteria (p < 0.05). The hematological parameters were significantly improved, and the intestinal bacterial count was reduced in broiler chickens supplemented with MPE-NC compared to control treatment. Conclusion: MPE-NC treatment provided optimal improvement for several tested measurements. These results indicate that MPE-NC could be used as a natural phytobiotic agent to improve physiological health and reduce the risk of bacterial infection. Keywords: Avian pathogenic Escherichia coli, Broiler, Hematological profile, Mangosteen peel extract, Total bacteria. IntroductionBroiler chickens are a commodity of poultry that have become an important livestock sector in fulfilling the animal protein needs of Indonesian society. Broiler chickens were preferred due to their benefits, such as quick growth, high feed conversion efficiency, and good quality meat productivity (Choi et al., 2023). The success of broiler production largely depends on the health of the birds. Infectious bacterial diseases can be a major economic burden to farmers. The avian pathogenic Escherichia coli is a bacterium that can cause severe pathogenesis in poultry. This bacterium is a dominant pathogen of colibacillosis, which can invade several organs, including the liver, heart, lungs, and respiratory tract (Nawaz et al., 2024). Systemic inflammations, including pericarditis, perihepatitis, and airsacculitis, are frequently accompanied by avian pathogenic Escherichia coli (APEC) infection. These circumstances result in lower growth performance, high mortality, and carcass rejection at slaughter (Kathayat et al., 2021). The extract of mangosteen peel (Garcinia mangostana L.) has the potential to prevent APEC-induced bacterial infection in broiler chickens because it is an abundant source of bioactive compounds. Mangosteen peel is a rich source of xanthones, flavonoids, tannins, and saponins with antimicrobial, antioxidant, anti-inflammatory and immunomodulatory potential. Xanthones, in particular α-mangostin and γ-mangostin, have been reported to disrupt bacterial cell membranes, inhibit protein synthesis, and disrupt the energy metabolism of cells, thereby affecting the ability of bacteria to grow or reproduce (Górecka et al., 2025; Majdalawieh et al., 2025). Moreover, flavonoids and tannins have been shown to precipitate the proteins of the bacterial cell wall, which could lead to easy break-up in the cell structure (Shamsudin et al., 2022). In addition to its antibacterial effects, Mangosteen Peel extract is a potent antioxidant that can scavenge free radicals produced in response to APEC infection. Bacterial infections frequently induce oxidative stress and tissue injury (Nseme et al., 2022). Mangosteen peel bioactive substances also show anti-inflammatory properties and can reduce inflammation in infected organs, promoting recovery and restoring the physiological state of chickens (Sun et al., 2024). MPE-NC demonstrates higher effectiveness in preventing APEC-induced bacterial infections in broiler chickens than conventional extract forms. This is attributed to nanoencapsulation technology that increases the stability, solubility, and bioavailability of its bioactive compounds. Xanthones, flavonoids, and tannins, which are widely recognized as antibacterial, antioxidant, and immunomodulatory materials, are the major compounds present in mangosteen. However, in the normal extraction form, the compounds are easily degraded in the digestive system and thus lose their activity very soon (Bi et al., 2023). Nanoencapsulation stabilizes the active compounds within a polymer matrix, such as chitosan-Sodium Tripolyphosphate (STPP), thereby protecting them from enzymatic degradation and enhancing their ability to maintain sustained release in the target place (Chowdhury et al., 2024). This study aimed to evaluate the effects of mangosteen peel extract nanocapsules and the optimal dosage on APEC-infected broiler chickens. Materials and MethodsMaterialsThe materials used in this study were mangosteen peel, chitosan, STPP, ethanol, day old chick (DOC) broiler Lohmann strain (MB 202), and basal feed broilers supplied by PT. Charoen Pokphand Indonesia Tbk (Hi-Pro-Vite 511, Table 1), physiological sodium chloride, glacial acetic acid, Tween 80, disinfectant, laundry soap, zinc-bacitracin antibiotics, formaldehyde, alcohol, vitamins, medicines. The research equipment included a macerator, rotary evaporator, spray dryer, light microscope, microtome, spectrophotometer, centrifuge, vortex mixer, water bath, oven, desiccator, and bacterial culture tools (agar media, incubator, Eppendorf tubes). Housing equipment consisted of pens measuring 1 m³, feeders and drinkers, heating lamps, digital scales, and surgical tools for sample collection. Table 1. Nutritional content of the basal feed (Hi-Pro-Vite 511).
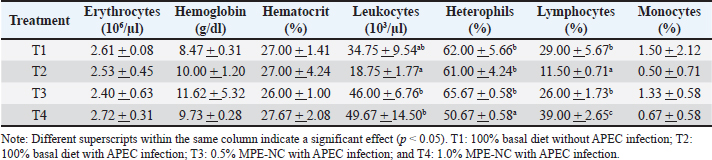
MethodsThe results of the synthesis and characterization of MPE-NC used in this study are as follows (Kusmayadi et al., 2025a,b). The encapsulant material used was chitosan:STPP with a 1:1 ratio. The results of the MPE-NC characterization test on the formula were obtained. The particle size of MPE-NC was approximately 234.00 ± 62.00 nm, and the zeta potential of MPE-NC was approximately −22.70 ± 1.30 mV. An experimental study was conducted to evaluate the efficacy of MPE-NC as a feed additive for broiler chickens and its influence on their hematological profile and total bacterial count. A total of 200 DOC Lohmann strain broiler chickens (unsexed) were randomly divided into four experimental groups: T1: 100% basal diet only without APEC infection; T2: 100% basal diet with APEC infection; T3: supplemented as 0.5% MPE-NC supplemented and challenged with APEC; and T4: supplemented as 1.0% MPE-NC supplemented and challenged with APEC. This experiment was repeated five times per treatment, with 10 broiler chickens kept in each cage. Rearing duration was 35 days, comprising the adaptation period (weeks 1–2) and the treatment period (weeks 3–5). Feed and water were offered twice daily (in the morning and evening) ad libitum. In the treatment groups, broilers were intratracheally infected with a local strain of APEC intratracheally infection (108 cells/ml) 0.5 ml at a dilution equivalent to the McFarland standard. Day 21 was the date of the oral gavage with infection. After 14 days of E. coli infection, five birds from both groups were bled for hematological parameters. Hematological profiles (erythrocytes, hemoglobin, hematocrit, leukocytes, heterophils, lymphocytes, and monocytes) and total bacterial counts (coliforms and lactic acid bacteria) were measured. The tests of each parameter are presented as follows: Hematological profileBroilers were randomly selected and fasted for several hours before blood collection. Blood samples were drawn from the brachial or jugular vein using a syringe, mixed with Ethylene Diamine Tetraacetic Acid anticoagulant solution to prevent clotting, stored in test tubes, and placed in a cool box. Blood samples were then transported to the laboratory for analysis. The hematological parameters analyzed included erythrocyte, hemoglobin, hematocrit, leukocyte, heterophil, lymphocyte, and monocyte counts. Total bacteriaThe Total Plate Count method was used to estimate the total count of bacteria. Broilers were sampled at the age of 35 days. A single chicken was randomly chosen from each flock, slaughtered, and the small intestine was retrieved. Duodenal fluid was collected, transferred into plastic sampling bags, and analyzed in the laboratory. After incubation, colonies were counted according to bacterial type, and actual bacterial counts were determined using Excel formulas. Data analysisThe data obtained were analyzed using analysis of variance to determine treatment effects, and the significant differences among treatments were further analyzed using Duncan’s New Multiple Range Test with SPSS version 25.0. Ethical approvalAll experiments, procedures, and treatment of laboratory animals were performed in strict adherence to the Animal Policy and Welfare Committee of the Faculty of Agriculture, Universitas Perjuangan Tasikmalaya No. 469/U/FP-UP/12/2025 on date December 10, 2025). ResultsHematological profilesSupplementation with mangosteen peel extract nanocapsule had no significant effect on the erythrocyte count of broiler chickens infected with APEC (p > 0.05) (Table 2). However, the decrease in the mean erythrocyte count was not significant compared with the negative control (T1). Table 2. Mean values of the hematological profiles of blood in broiler chickens.
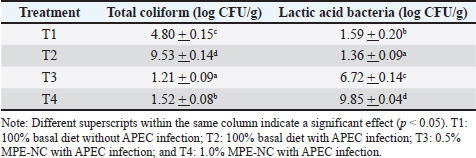
As shown in Table 1, dietary supplementation with mangosteen peel extract nanocapsules had no effect on the Hb level of APEC-infected broilers (p > 0.05). This implies that infection and/or MPE-NC supplementation had little effect on the physiological condition of chicken blood. The control group (T1) had an Hb value of 8.47 g/dl. Hb in the positive control (T2) increased to 10.00 g/dl. A high hemoglobin level is not necessarily indicative of good health because inflammation can also lead to oxidative stress. The average in treatment T1 was 27% with heterogeneity between individuals, indicating a normal physiological state. The mean value remained the same in T2 (a positive control, APEC-infected) (27%). This suggests that although APEC infection did not cause a significant difference in hematocrit values (p > 0.05), it induced individual variation. The hematocrit level in T3 (0.5% MPE-NC) was slightly decreased to 26%. This result indicates that the relatively low dosage of 0.5% was insufficient to confer maximum protective effects against APEC-induced oxidative stress. The average hematocrit was 27.67% for T4 (1% MPE-NC). There was a slight increase in this value, which, however, remained within the normal range, with no significant difference from T1 (p > 0.05). Table 2 shows a significant difference (p < 0.05) in the blood leukocyte levels of broiler chickens infected with APEC under different treatments. The negative control group (T1) had an average leukocyte count of 34.75 × 10³/µl, which was within the normal range for broilers. APEC infection without treatment (T2) significantly reduced the leukocyte count to 18.75, indicating leukopenia due to infection. The leukocyte decline in this group shows that APEC infection can weaken the host’s immune response, as evidenced by the limited number of circulating immune cells. The results showed that the heterophil levels in broiler chicken blood significantly differed (p < 0.05) among the treatments. In T1 (negative control), the heterophil count was 62.00%, whereas in T2 (positive control), it was 61.00%, with no significant difference between the two groups. This indicates that APEC infection triggered an increased inflammatory response while maintaining high heterophil levels. The results showed a significant difference (p < 0.05) in lymphocyte levels in broiler chicken blood among treatments. In T1 (negative control), the lymphocyte level was 29.00%, whereas it drastically decreased to 11.50% in T2 (positive control, APEC infection). Monocyte levels in broiler chicken blood differed significantly (p < 0.05) among treatments. In T1 (negative control), the monocyte level was 1.50%, whereas it significantly decreased to 0.50% in T2 (positive control, APEC infection). Total bacteriaTable 3 shows a significant difference among treatments of broiler chickens (p < 0.05) for total coliform bacteria in swab sample results. The maximum coliform number was obtained in T2 (positive control, infected with APEC), which was 9.53 log CFU/g, and APEC infection led to a significant increase in the coliform population within the gut content. On the other hand, 4.80 log CFU/g of coliforms was obtained in T1 (negative control), representing a more stable gut microbiota in healthy chickens. Treatments T2 and nanoencapsulated mangosteen peel extract significantly reduced coliform counts compared with T1. Treatments T3 (0.5%) and T4 (1%) reduced the coliform count to 1.21 and 1.52 log CFU/g, respectively, both of which were significantly lower than those of the negative control. This marked difference is proof that the 1% dose was more efficient for reducing the coliform load in broiler chickens infected by APEC. Table 3. Mean values of total bacteria in broiler chickens.
There was a significant difference between treatments in the total lactic acid bacteria (LAB) number (p < 0.05). In the negative control (T1), the LAB count was 1.59 log CFU/g, and in the positive control (T2, APEC-infected), it decreased to 1.36 log CFU/g, suggesting that APEC could inhibit the growth of LAB in the digestive tract of broiler chickens. However, the LAB population was significantly increased by the addition of MPE-NC. When LAB counts were expressed as 3.6 log CFU/g in T2 (0%), they were increased to 6.72 log CFU/g in T3 (0.5%) and even more by affecting the maximum level of 9.85 log CFU/g for T4 (1%), being higher than negative control as well. Therefore, MPE-NC supplementation at 1% was the most effective in improving the LAB population in APEC-infected broilers. DiscussionHematological profileThe average erythrocyte count remained within the normal physiological range for broilers, namely 2.5–3.5 × 106/µl (Weiss and Wardrop, 2006; Campbell, 2015), suggesting that the treatments did not have profound effects on blood count. Supplementation with 0.5% MPE-NC (T3) showed lower erythrocyte counts compared to T1 and T2 (Table 2). This reduction could be correlated with the pathogenic actions of APEC by which hemolysis, inflammation, and tissue damage lead to host immune response (Watts and Wigley, 2024). This shows that the suboptimal protection against APEC-induced erythrocyte lesions was achieved with the subprotective dosage. These discrepancies might be attributed to a lack of bioactive efficacy in balancing oxidative stress (Hu et al., 2022). The erythrocyte concentration was the highest (2.72 × 106/µl) using the latter supplementation level, with this value being higher than that of the negative control (2.61 × 106/µl). This shows that 1% MPE-NC was more effective in providing protective effects. Bioactive compounds, such as xanthones and flavonoids, have antioxidant, anti-inflammatory, and antibacterial activities that contribute to the maintenance of erythrocyte cell membrane integrity and prevention of hemolysis due to bacterial infection (Subroto et al., 2025). Although the treatments did not have a statistically significant effect on erythrocyte counts, there was a tendency for increased erythrocyte levels with 1% MPE-NC supplementation. This is in agreement with the assumption that a higher dose could offer immunomodulatory and protective effects against oxidative stress and erythrocyte damage induced by APEC infection, as recently reported for withdrawing problem-biotic compounds on broiler chickens’ hematological health (Widowati et al., 2020; Pratiwi et al., 2025). Throughout all treatments, Hb levels fluctuated within the normal physiological range for broilers (7–13 g/dl) (Weiss and Wardrop, 2006; Campbell, 2015). The Hb value was the highest (11.62 g/dl) with 0.5% MPE-NC treatment (T3). This is an indication that the effect of the lower dose used was not very stable in inducing different reactions among subjects. A small proportion of chickens may have been exposed to high Hb elevations because of the antimetabolite nature of flavonoids and xanthones (Desbruslais and Wealleans, 2022), while others responded little. This high variability made the results less reliable. Conversely, the results observed in the 1% MPE-NC-treated tumors (T4) were more consistent (9.73 g/dl Hb levels). This value fell within the normal range and was higher than T1 but lower than T2 and T3. This increase may be part of a compensatory mechanism in response to infection, as APEC induces inflammation, with some evidence suggesting an increased tissue oxygen demand and thus an upregulation of erythropoiesis (Watts and Wigley, 2024). The relatively stable T4 level indicates that the dose was better at a reasonable equilibrium of physiological homeostasis without extraordinary amplitude. This protective effect may be attributed to the higher optimum antioxidant and immunomodulatory potential of MPE-NC at increased doses (Nwaigwe et al., 2020). The effects were statistically nonsignificant (p > 0.05), yet the trend in the data suggested that APEC infection might increase Hb as a compensation response. The highest levels of Hb were achieved at 0.5% MPE-NC dosage with low stability, whereas 1% MPE-NC treatment was more effective for sustaining hematological stability. These results are consistent with those of recent studies, indicating that polyphenol-based phytobiotics could benefit blood poultry health via antioxidant and immunomodulation pathways (Kikusato, 2021). In this study, the hematocrit values of broiler chicken were between the reference range of 22%–35% (Weiss and Wardrop, 2006; Campbell, 2015). The results in Table 2 show that APEC infection did not lead to dramatic reductions in hematocrit but had a dramatic effect on inter-individual variation. Some chickens may have experienced a decrease in hematocrit due to hemolysis, while others showed an increase due to dehydration or compensatory erythropoiesis (Bienvenu et al., 2021). This result is in accordance with that of Akhtar et al. (2015), who reported that bacterial infections in chickens may present diverse hematological responses depending on infection severity, host physiological condition, and individual immune ability. Although blood homogeneity values were small in the 0.5% MPE-NC-treated group (T3), the mean hematocrit was not significantly higher in T3. This indicates that the low dose was insufficient to offer complete protection against erythrocyte injury. According to Urban et al. (2025), a low concentration of antioxidant plant extracts would have an incomplete effect compared with the ideal dosage level. Whereas treatment with 1% (T4) gave more uniform performance. Although it was not statistically significant (p > 0.05), the hematocrit in this group tended to increase and remained stable among the subjects. The presence of xanthones and flavonoids in mangosteen has antioxidant and anti-inflammatory activity, which can protect erythrocytes from oxidative damage (Rizaldy et al., 2022). Therefore, it can be inferred that 1% MPE-NC was more efficient than 0.5% in preserving the hematocrit stability of broiler chickens infected with APEC. These results confirm the potential of antioxidant phytogenic supplementation as an option for improving hematological health in chickens with an infected-stressed status. The normal leukocyte count in broiler chickens ranges from 12–30 × 10³/µl (Weiss and Wardrop, 2006; Campbell, 2015). This condition may be attributed to leukocyte migration into infected tissues, leukocyte consumption during phagocytosis, and the suppressive effect of APEC endotoxins on bone marrow activity, thereby reducing leukocyte production (Leick et al., 2014). MPE-NC supplementation at doses of 0.5% (T3) and 1% (T4) significantly increased leukocyte levels compared with the positive control group. Group T3 recorded an average leukocyte count of 46.00 ± 6.76, whereas T4 reached 49.67 ± 14.50. This increase is likely associated with the antioxidant, antibacterial, and immunomodulatory properties of xanthones, flavonoids, and tannins in mangosteen peel (Abate et al., 2022; Yuvanatemiya et al., 2022). These compounds can suppress oxidative damage to immune cells, enhance phagocytic activity, and stimulate lymphocyte proliferation, thereby increasing the number of circulating leukocytes. Moreover, nanoencapsulation improves the bioavailability of the active compounds of mangosteen, optimizing their protective effects on the immune system (Gallorini et al., 2022). Interestingly, although both T3 and T4 differed significantly from T2, leukocyte counts in T4 were relatively higher than in T3 but with greater individual variation (SD 14.50). This indicates a possible dose-dependent response, where the 1% concentration provided a stronger leukocyte-stimulating effect, albeit not uniformly across all individuals. Physiologically, an increase in leukocyte counts within the normal range indicates activation of the body’s defense system against infection. Mangosteen peel extract nanocapsules can be considered effective in improving the hematological profile of APEC-infected broiler chickens. These findings are consistent with previous reports that antioxidant-rich herbal extract supplementation can enhance leukocyte counts and strengthen immune responses in broilers under infectious and environmental stress (Nehme et al., 2025; Wiggins et al., 2025). Groups T3 and T4 had significantly higher mean leukocyte counts than T2. Meanwhile, T1 did not differ significantly from the other treatments. Several biological mechanisms may explain the leukocyte reduction observed in T2 in severely infected chickens. These include leukocyte migration or sequestration into infected tissues, particularly during systemic or focal infections such as peritonitis or airsacculitis, resulting in reduced circulating counts. Severe T2 infection may have led to phagocytosis and immune cell consumption at infection sites, thereby lowering peripheral counts. In addition, responses to exotoxins may trigger hemodynamic changes and leukocyte redistribution, sometimes causing an early leukopenia during acute sepsis before the development of leukocytosis. Severe systemic infections or inflammatory mediators may also suppress erythropoiesis and leukopoiesis, reducing leukocyte production. Furthermore, elevated corticosterone due to stress and infection can alter leukocyte composition, such as lymphopenia, thereby decreasing total leukocyte counts (Al-Qahtani et al., 2024). The higher leukocyte values in T3 and T4 can be caused by several factors, including the immunomodulatory effects of mangosteen peel extract. Mangosteen peel contains xanthones, flavonoids, and tannins, which stimulate lymphocyte proliferation, macrophage/heterophil phagocytic activity, and both humoral and cell-mediated immune responses (Yendri et al., 2022). An increase in total leukocyte count may reflect immune cell activation. In addition, the antibacterial properties of mangosteen bioactives can reduce the APEC burden by reducing leukocyte destruction and consumption, and peripheral leukocyte counts may recover or increase (Sun et al., 2021). The antioxidants in the mangosteen peel extract may also prevent oxidative damage to immune cells (including bone marrow), thereby supporting leukocyte production and viability. Chowdhury et al. (2024) reported that nanoencapsulation enhances the stability, bioavailability, and controlled release of active compounds. Effective concentrations in target tissues are higher and more consistent, producing stronger immunomodulatory effects than conventional extracts. Based on statistical analysis, both treatment doses (T3 and T4, 0.5% and 1%, respectively) significantly increased leukocyte counts compared with T2. The absolute difference (with slightly higher T4) indicates a dose-dependent effect. The negative control (T1) reflected the basal or normal immune condition of the healthy chickens. Statistical results showed that T1 did not significantly differ from T2, T3, or T4, suggesting that its values lay between the leukopenic condition of infection (T2) and the leukocyte-stimulating effect of treatments (T3/T4). This is reasonable because APEC infection suppresses leukocyte counts (T2), whereas immunostimulatory treatments increase them (T3/T4), while the healthy control group lies in between. The leukopenia observed in T2 indicates that APEC infection had systemic and acute effects, highlighting the need for medical intervention. The normal heterophil value in broiler chickens ranges from 15% to 50% (Weiss and Wardrop, 2006; Campbell, 2015). In the T3 treatment, the heterophil level rose as high as 65.67% but remained higher than in the T1 and T2 treatments. This increase is thought to be a form of nonspecific immune stimulation by bioactive compounds in mangosteen peel, such as xanthones and flavonoids, which can activate leukocytes and enhance phagocytic cell mobilization (Naing et al., 2023). Heterophils are the main innate immune cells that are rapidly mobilized for phagocytosis, degranulation, and the release of antimicrobial mediators, making their elevation an indicator of acute inflammation caused by bacterial infection (Rosales, 2018). Although excessively high heterophil levels may also reflect physiological stress in chickens, this response is beneficial in strengthening the initial defense against pathogen exposure (Alkie et al., 2019). In contrast, in T4 (1% MPE-NC), heterophils dropped significantly to 50.67%, which was below those of all other treatments. This decline illustrates that a high dose of MPE-NC can inhibit an excessive inflammatory response. The mechanism is correlated with the antioxidant and anti-inflammatory property of α-mangostin; it inhibits NF-κB and MAPK pathway activation to decrease the production of proinflammatory cytokines and the recruitment of heterophils into the circulation (Xu et al., 2024). This protection is also reinforced by nanoencapsulation technology, which improves the bioavailability of biological drugs and induces a more controlled immune response (Rezagholizade-Shirvan et al., 2024). This agrees with a previous study suggesting that extremely high heterophil levels are often referred to as oxidative stress, excessive inflammation, and growth-retarded performance in broiler chickens (Dash et al., 2025). Therefore, T4’s lower heterophil levels reflect better immune modulation. The 0.5% MPE-NC dosage (T3) was likely to promote the mobilization of heterophils for defense action, whereas the 1% MPE-NC dosage (T4) had a stronger ability to inhibit inflammation and immune balance. These findings demonstrate that the nanocoencapsulation of mangosteen peel extract may serve as a promising immunomodulatory agent. The 1% dose was preferable to decrease inflammation and to keep physiological parameters more stable in APEC-infected broiler chickens. The lymphocyte percentage in T3 was 26.00%, which was higher than that of T2 but not significantly different from that of T1. This means that a small amount of MPE-NC caused the number of lymphocytes to recover near normal. This indicates a significant lymphopenia caused by APEC infection, which is consistent with reports that systemic infections in poultry can induce immunosuppression through lymphocyte redistribution to lymphoid tissues, apoptosis induced by bacterial toxins, and increased corticosterone levels that suppress lymphocyte proliferation (Wang et al., 2024). Xanthones, flavonoids, and polyphenols of mangosteen have been shown to possess immunomodulatory activities, such as modulating lymphocyte proliferation and the functioning of adaptive immune cells (Li et al., 2023). T4 exhibited the highest proportion of lymphocytes, 39.00%, which was significantly different from all other groups. This increase represents a more significant effect of immunomodulation in the high dose, and it is strengthened by the influence of nanoencapsulation in increasing the bioavailability of active compounds. Xanthones, especially α-mangostin, can act as antioxidants and anti-inflammatory agents that shield lymphocytes from oxidative stress and apoptosis and promote the proliferation and differentiation of T and B cells (Majdalawieh et al., 2025). From a biological standpoint, the increase in lymphocytes in T4 signifies increased activation of the adaptive immune response, which is needed to fight bacterial infections by improving antibody production and effector cell responses (Chapman and Chi, 2022). The marked difference between T3 and T4 verifies the effect of extract concentration on immune-modulating potency, since 0.5% treatment tends to maintain a lymphocyte level compatible with the physiological standard, whereas 1% treatment induces markedly excessive proliferation above that range. MPE-NC, especially at the 1% dosage, was effective as a natural immunomodulatory agent to mitigate immunosuppression triggered by APEC infection, adding lymphocyte counts, and strengthening the defense mechanism in broiler chicken against stress induced by infection (Hooda et al., 2024). The normal monocyte counts in broiler chickens ranged from 0% to 7% (Weiss and Wardrop, 2006; Campbell, 2015). In the T3 treatment, the monocyte level was 1.33%, which was significantly higher than that in T2 and was not significantly different from that in T1. This indicates that APEC infection can suppress circulating monocytes, possibly through mechanisms such as monocyte migration to lymphoid and inflamed tissues for differentiation into macrophages, or through apoptosis induced by bacterial toxins and inflammatory mediators (Dash et al., 2024). This indicates that the bioactive compounds in mangosteen peel have a protective effect against monocyte reduction caused by infection. Xanthones and flavonoids possess immunomodulatory and phagocytic activities, which can enhance monocyte–macrophage proliferation and function, including increased phagocytosis and cytokine production (Oriola and Kar, 2024). Interestingly, the monocyte count decreased to 0.67% in T4, which was significantly different from that of T1 and T3. This reduction could be the result of the strong anti-inflammatory activity at high doses, where polyphenols (xanthones and α-mangostin) suppressed pro-inflammatory pathways such as NF-κB and MAPK, which in turn led to a decrease in monocyte migration from bone marrow into circulation. Consequently, whereas T4 boosted adaptive immunity (reflected by the rise in lymphocytes), there were fewer circulatory monocytes due to more active tissue migration or stronger influence of inflammation (Alam et al., 2023). Overall, these findings indicate that APEC infection (T2) reduced circulating monocyte levels, whereas low-dose T3 supplementation maintained normal monocyte levels. On the contrary, the greater dose (T4) showed a stronger immunomodulation toward inflammation control, which resulted in low peripheral monocytes. This is consistent with the fact that monocytes are not the major indicators of acute inflammation in APEC infection, but rather play an important role as precursors of tissue macrophages. Thus, the interpretation of the immune response may be improved by considering changes in heterophils and lymphocytes as well as the H/L ratio as measures of immunological stress (Bråthen et al., 2025). Total bacteriaThe APEC infection has been shown to significantly increase coliform counts, which is in line with Khairullah et al. (2024), who observed that pathogenic E. coli can cause distortion dysbiosis and consequently induce a higher ratio of coliforms. This state is unfavorable to gut health, leads to diarrhea, and impairs nutrient absorption. The marked decrease in coliform count numbers in T3 and T4 indicated that the mangosteen rind extract had antibacterial potential as a natural compound. The active components of mangosteen, such as xanthones and flavonoids, disrupt the bacterial cell membrane and block several key metabolic enzymes, leading to the growth inhibition of pathogenic bacteria (Zhang et al., 2025). Higher efficacy in T4 (1%) than T3 (0.5%) can be attributed to the concept of nanoencapsulation. It enhances the stability, solubility, and bioavailability of bioactive compounds and improves their antibacterial activity (Zhuo et al., 2024 ). Therefore, the protective effect of 1% MPE-NC supplementation was more uniform against APEC infection. Treatment with T3 and T4 could also be beneficial to balance the gut microbiota in addition to suppressing coliform bacteria. Rachwal and Gustaw et al. (2025) also indicated that phytogenic compounds can regulate microbiota by inhibiting pathogens while allowing beneficial bacteria, such as Lactobacillus spp., to receive nutrition and substrate support. Gut microbiota balance directly influences digestive health and growth performance of broilers. These data indicate that APEC infection promotes coliform growth, whereas MPE-NC supplementation inhibits the proliferation of pathogenic coliform (p < 0.05). Treatments T3 and T4 resulted in superior criteria, indicating that the mangosteen peel extract could be an interesting natural phytogenic to improve the gut health of broiler chickens. The decrease in LAB in the T2 group was also in line with the findings of Awad et al. (2017), who reported that infectious agents, such as pathogenic E. coli infection, can disturb gut microbial homeostasis by inhibiting probiotic microorganisms and facilitating pathogen adherence. This mechanism includes nutrient competition, toxin synthesis, and injury to small intestinal epithelial cells, leading to an adverse luminal environment for LAB growth (Vinayamohan et al., 2024). The rise in LAB levels in T3 and T4 indicates the protective role of MPE-NC. The predominant active compounds of mangosteen peel, in particular α-mangostin and flavonoids, show better antibacterial activity against gram-negative E. coli than against gram-positive LAB. This force also provides a competitive advantage for LAB growth (Liu et al., 2025). The nanoencapsulation technique plays an equally important role, as it shields the bioactive components from gastric degradation and increases their absorption in the small intestine. As a result, antimicrobial action was more robust and selective, thereby promoting the positive modulation of gut microbiota (Zhang and Cheng, 2022). The LAB level increase found in T4 (1%), which was ~27 times as high as the healthy control, indicates that a dose somewhat above this is most effective for changing microbiota toward favoring a healthier composition. The contribution of LAB to gut health is critical. LAB secretes lactic acid and bacteriocins, which help to reduce intestinal pH, prevent pathogen adhesion, and promote mucosal immunity by increasing IgA induction and promoting epithelial integrity (Vieco-Saiz et al., 2019; Ismael et al., 2021). Thus, the elevation of LAB in the treated groups showed better gut health and resistance to APEC infection. These findings support the findings of the intestinal content analysis that APEC infection reduced LAB populations and MPE-NC supplementation significantly increased LAB counts (p < 0.05). The optimum dosage of 1% mangosteen peel is believed to have potential as a natural phytogenic agent that not only suppresses pathogens but also increases the survival effect of beneficial bacteria on broiler chickens, thereby positively influencing performance and health (Kusmayadi et al., 2025a,b; Kadiwano et al., 2025). ConclusionInfection with avian pathogenic E. coli significantly reduced the physiological health status of broiler chickens, as indicated by decreased hematological parameters (erythrocytes, hemoglobin, hematocrit, and lymphocytes), increased heterophil counts, and reduced intestinal lactic acid bacteria populations. These conditions indicate immunological stress, inflammation, and gut microbiota imbalance due to pathogen colonization. MPE-NC supplementation demonstrated positive effects in modulating immune responses and microbiota. The supplementation of 0.5% MPE-NC was able to increase most blood indices and recover LAB counts up to the control level, whereas that in higher dose (1%) produced an outstanding role in the reduction of heterophil percentages, increases in lymphocyte values, as well as tremendous growth of LAB populations. These observations demonstrate the immunomodulatory, anti-inflammatory, and antibacterial-inducing potentials of xanthones from the mangosteen peel as a bioactive compound. In conclusion, MPE-NC is a promising natural phytogenic candidate for enhancing hematological health, attenuating pathogenic bacterial infection, modulating the gut microbiome, and boosting immune resistance in broiler chickens against APEC infection. AcknowledgmentAcknowledgments are expressed sincere gratitude to the Directorate of Research and Community Service, Directorate General of Research and Development, Ministry of Higher Education, Science, and Technology, Republic of Indonesia, for the grant No 0070/C3/AL.04/2025 of the Regular Fundamental Research Grants. Conflict of interestThe authors have no conflicts of interest to declare. FundingThis study was supported by the Directorate of Research and Community Services, Directorate General of Research and Development, Ministry of Higher Education, Science, and Technology, Republic of Indonesia, through the Fundamental Research Scheme in 2025 (Grant No. 0070/C3/AL.04/2025). Authors’ contributionThis study was carried out in collaboration with all the authors (AK, RSS, ES, and YY) and the research theme, methods, and experiments were defined. AK conducted the experiments, analyzed the data, interpreted the results, and wrote the manuscript. All authors have read, reviewed, and approved the final version of the manuscript. Data availabilityAll data generated during this study have been published in the form of a manuscript. ReferencesAbate, M., Pagano, C., Masullo, M., Citro, M., Pisanti, S., Piacente, S. and Bifulco, M. 2022. Mangostanin, a xanthone derived from Garcinia mangostana fruit, exerts protective and reparative effects on oxidative damage in human keratinocytes. Pharmaceuticals 15(1), 84; doi:10.3390/ph15010084 Akhtar, M., Awais, M.M., Anwar, M.I., Ehtisham-Ul-Haque, S., Nasir, A., Saleemi, M.K. and Ashraf, K. 2015. The effect of infection with mixed Eimeria species on hematology and immune responses following Newcastle disease and infectious bursal disease booster vaccination in broilers. Vet. Quart. 35(1), 21–26; doi: 10.1080/01652176.2014.991048 Alam, M., Rashid, S., Fatima, K., Adnan, M., Shafie, A., Akhtar, M.S., Ganie, A.H., Eldin, S.M., Islam, A., Khan, I. and Hassan, M.I. 2023. Biochemical features and therapeutic potential of α-mangostin: mechanism of action, medicinal values, and health benefits. Biomed. Pharmacother. 163, 114710; doi:10.1016/j.biopha.2023.114710 Alkie, T.N., Yitbarek, A., Hodgins, D.C., Kulkarni, R.R., Taha-Abdelaziz, K. and Sharif, S. 2019. Development of innate immunity in chicken embryos and newly hatched chicks: a disease control perspective. Avian. Pathol. 48(4), 288–310; doi:10.1080/03079457.2019.1607966 Al-Qahtani, A.A., Alhamlan, F.S. and Al-Qahtani, A.A. 2024. Pro-inflammatory and anti-inflammatory interleukins in infectious diseases: a comprehensive review. Trop. Med. Infect. Dis. 9(1), 13; doi:10.3390/tropicalmed9010013 Awad, W., Hess, C. and Hess, M. 2017. Enteric pathogens and their toxin-induced disruption of the intestinal barrier through alteration of tight junctions in chickens. Toxins 9(2), 60; doi:10.3390/toxins9020060 Bi, C., Xu, H., Yu, J., Ding, Z. and Liu, Z. 2023. Botanical characteristics, chemical components, biological activity, and potential applications of mangosteen. PeerJ 11, e15329; doi: 10.7717/peerj.15329 Bienvenu, A., Martinez, E. and Bonazzi, M. 2021. Undercover agents of infection: the stealth strategies of T4SS-equipped bacterial pathogens. Toxins 13(10), 713; doi:10.3390/toxins13100713 Bråthen, V.S., Skomsø, D.B. and Bech, C. 2025. The heterophil-to-lymphocyte (H/L) ratio indicates varying physiological characteristics in nestlings compared to adults in a long-lived seabird. Birds 6(1), 4; doi:10.3390/birds6010004 Campbell, T.W. 2015. Exotic Animal Hematology and Cytology. 4th Edition. CO: John Wiley & Sons, Inc. Chapman, N.M. and Chi, H. 2022. Metabolic adaptation of lymphocytes in immunity and disease. Immunity 55(2), 224–239; doi:10.1016/j.immuni.2021.12.012 Choi, J., Kong, B., Bowker, B.C., Zhuang, H. and Kim, W.K. 2023. Nutritional strategies to improve meat quality and composition in the challenging conditions of broiler production: a review. Animals 13, 1386; doi:10.3390/ani13081386 Chowdhury, S., Kar, K. and Mazumder, R. 2024. Exploration of different strategies of nanoencapsulation of bioactive compounds and their ensuing approaches. Future. J. Pharm. Sci. 10(1), 644; doi:10.1186/s43094-024-00644-y Dash, S.P., Gupta, S. and Sarangi, P.P. 2024. Monocytes and macrophages: origin, homing, differentiation, and functionality during inflammation. Heliyon 10(8); doi:10.1016/j.heliyon.2024.e29686 Dash, U.C., Bhol, N.K., Swain, S.K., Samal, R.R., Nayak, P.K., Raina, V., Panda, S.K., Kerry, R.G., Duttaroy, A.K. and Jena, A.B. 2025. Oxidative stress and inflammation in the pathogenesis of neurological disorders: mechanisms and implications. Acta Pharm. Sin. B 15(1), 15–34; doi:10.1016/j.apsb.2024.08.006 Desbruslais, A. and Wealleans, A. 2022. Oxidation in poultry feed: impact on the bird and the efficacy of dietary antioxidant mitigation strategies. Poultry 1(4), 246–277; doi:10.3390/poultry1040022 Gallorini, M., Carradori, S., Resende, D.I.S.P., Saso, L., Ricci, A., Palmeira, A., Cataldi, A., Pinto, M. and Sousa, E. 2022. Natural and synthetic xanthone derivatives counteract oxidative stress via Nrf2 modulation in inflamed human macrophages. Int. J. Mol. Sci. 23(21), 13319; doi:10.3390/ijms232113319 Górecka, H., Guźniczak, M., Buzalewicz, I., Ulatowska-Jarża, A., Korzekwa, K. and Kaczorowska, A. 2025. Alpha-mangostin: a review of current research on its potential as a novel antimicrobial and anti-biofilm agent. Int. J. Mol. Sci. 26, 5281; doi:10.3390/ijms26115281 Hooda, P., Malik, R., Bhatia, S., Al-Harrasi, A., Najmi, A., Zoghebi, K., Halawi, M.A., Makeen, H.A. and Mohan, S. 2024. Phytoimmunomodulators: a review of natural modulators for complex immune system. Heliyon 10, e23790; doi:10.1016/j.heliyon.2023.e23790 Hu, J., Afayibo, D.J.A., Zhang, B., Zhu, H., Yao, L., Guo, W., Wang, X., Wang, Z., Wang, D., Peng, H., Tian, M., Qi, J. and Wang, S. 2022. Characteristics, pathogenic mechanism, zoonotic potential, drug resistance, and prevention of avian pathogenic Escherichia coli (APEC). Front. Microbiol. 13, 1049391; doi:10.3389/fmicb.2022.1049391 Ismael, A.B., Ashmawy, N.A. and Mostafa, M.M. 2021. Evaluation of the antimicrobial and antioxidant activities of mangosteen (Garcinia mangostana L.) rind and its effects on growth performance and immunity of broiler chickens. Egypt. J. Vet. Sci. 52(3), 367–377; doi:10.21608/ejvs.2021.71663.1212 Kadiwano, F.A., Bidura, I.G.N.G., Siti, N.W. and Melati, N.P.Y. 2025. Effect of water extract of mangosteen peel (Garcinia mangostana L.) fermented by probiotics on meat chemistry, abdominal fat, and blood lipid profile of broilers. World. J. Bio. Pharm. Health. Sci. 21(1), 503–510; doi:10.30574/wjbphs.2025.21.1.0088 Kathayat, D., Lokesh, D., Ranjit, S. and Rajashekara, G. 2021. Avian pathogenic Escherichia coli (APEC): an overview of virulence and pathogenesis factors, zoonotic potential, and control strategies. Pathogens 10, 467; doi:10.3390/pathogens10040467 Khairullah, A.R., Afnani, D.A., Riwu, K.H.P., Widodo, A., Yanestria, S.M., Moses, I.B., Effendi, M.H., Ramandinianto, S.C., Wibowo, S., Fauziah, I., Kusala, M.K.J., Fauzia, K.A., Furqoni, A.H. and Raissa, R. 2024. Avian pathogenic Escherichia coli: epidemiology, virulence and pathogenesis, diagnosis, pathophysiology, transmission, vaccination, and control. Vet. World 17(12), 2747–2762; doi:10.14202/vetworld.2024.2747-2762 Kikusato, M. 2021. Phytobiotics to improve health and production of broiler chickens: functions beyond the antioxidant activity. Anim. Biosci. 34(3), 345–353; doi:10.5713/ab.20.0842 Kusmayadi, A., Mardianingrum, R. and Yanti, Y. 2025a. Formulation and characterization of mangosteen rind extract nanocapsules as a feed additive candidate of poultry. Open. Vet. J. 15(4), 1771–1783; doi:10.5455/OVJ.2025.v15.i4.29 Kusmayadi, A., Mardianingrum, R., Wijayanti, D. and Yanti, Y. 2025b. The effect of mangosteen rind extract nanocapsule as a feed additive on the growth performance and blood lipid profile of broiler chicken. Open. Vet. J. 15(6), 2762–2773; doi:10.5455/OVJ.2025.v15.i6.44 Leick, M., Azcutia, V., Newton, G. and Luscinskas, F.W. 2014. Leukocyte recruitment in inflammation: basic concepts and new mechanistic insights based on new models and microscopic imaging technologies. Cell. Tissue. Res. 355(3), 647–656; doi:10.1007/s00441-014-1809-9 Li, R., Inbaraj, B.S. and Chen, B.H. 2023. Quantification of xanthone and anthocyanin in mangosteen peel by UPLC-MS/MS and preparation of nanoemulsions for studying their inhibition effects on liver cancer cells. Int. J. Mol. Sci. 24(4), 3934; doi:10.3390/ijms24043934 Liu, X., Song, M., Liu, Y., Yang, S., Chen, S., Kang, J., Shen, J. and Zhu, K. 2025. Rational design of natural xanthones against Gram-negative bacteria. Adv. Sci. 12(4), 2411923; doi:10.1002/advs.202411923 Majdalawieh, A.F., Khatib, B.K. and Terro, T.M. 2025. Α-Mangostin is a xanthone derivative from mangosteen with potent immunomodulatory and anti-inflammatory properties. Biomolecules 15, 681; doi:10.3390/biom15050681 Naing, S., Sandech, N., Maiuthed, A., Chongruchiroj, S., Pratuangdejkul, J. and Lomarat, P. 2023. Garcinia mangostana L. pericarp extract and its active compound, mangostin, as potential inhibitors of immune checkpoint programmed death ligand-1. Molecules 28(19), 6991; doi:10.3390/molecules28196991 Nawaz, S., Wang, Z., Zhang, Y., Jia, Y., Jiang, W., Chen, Z., Yin, H., Huang, C. and Han, X. 2024. Avian pathogenic Escherichia coli (APEC): current insights and future challenges. Poult. Sci. 103(12), 104359; doi:10.1016/j.psj.2024.104359 Nehme, R., Chervet, A., Decombat, C., Habanjar, O., Longechamp, L., Rousset, A., Chalard, P., Gainche, M., Senejoux, F., Fraisse, D., Filaire, E., Berthon, J.Y., Diab-Assaf, M., Delort, L. and Caldefie-Chezet, F. 2025. Selected plant extracts regulating the inflammatory immune response and oxidative stress: focus on Quercus robur. Nutrients 17(3), 510; doi:10.3390/nu17030510 Nseme, Y.D.M., Mandeng, K.F.P., Mounpou, J., Djuikoo, I.L.N., Nguedjo, M.W., Baleba, R.M.M., Mouafo, H.T. and Medoua, G.N. 2022. Bioactive compounds from mangosteen fruit peels (Garcinia mangostana L.) and assessment of their antioxidant potential. Microbiol. Res. J. Int. 32(11–12), 40–52; doi:10.9734/mrji/2022/v32i113008 Nwaigwe, C.U., Ihedioha, J.I., Shoyinka, S.V. and Nwaigwe, C.O. 2020. Evaluation of the hematological and clinical biochemical markers of stress in broiler chickens. Vet. World. 13(10), 2294–2300; doi:10.14202/vetworld.2020.2294-2300 Oriola, A.O. and Kar, P. 2024. Naturally occurring xanthones and their biological implications. Molecules 29(17), 4241; doi:10.3390/molecules29174241 Pratiwi, Y.S., Rini, D.M., Defri, I., Qubra, T. and Maulidi, N.M.I. 2025. Utilization of Garcinia mangostana L. peel as an immunomodulator to improve the quality of human resources: a systematic review. Amerta. Nutr. 9(2), 377–388; doi:10.20473/amnt.v9i2.2025.377-388 Rachwał, K. and Gustaw, K. 2025. Plant-derived phytobiotics as emerging alternatives to antibiotics against foodborne pathogens. Appl. Sci. 15(12), 6774; doi:10.3390/app15126774 Rezagholizade-Shirvan, A., Soltani, M., Shokri, S., Radfar, R., Arab, M. and Shamloo, E. 2024. Bioactive compound encapsulation: characteristics, applications in food systems, and implications for human health. Food. Chem. X. 24, 101953; doi:10.1016/j.fochx.2024.101953 Rizaldy, D., Hartati, R., Nadhifa, T. and Fidrianny, I. 2022. Chemical compounds and pharmacological activities of mangosteen (Garcinia mangostana L.)—updated review. Biointerface Res. Appl. Chem. 12(2), 2503–2516; doi:10.33263/BRIAC122.25032516 Rosales. and C. 2018. Neutrophil: a cell with many roles in inflammation or several cell types?. Front. Physiol. 9, 113; doi:10.3389/fphys.2018.00113 Shamsudin, N.F., Ahmed, Q.U., Mahmood, S., Ali Shah, S.A., Khatib, A., Mukhtar, S., Alsharif, M.A., Parveen, H. and Zakaria, Z.A. 2022. Antibacterial effects of flavonoids and their structure-activity relationship study: a comparative interpretation. Molecules 27(4), 1149; doi:10.3390/molecules27041149 Subroto, E., Afifah, T.N., Harlina, P.W., Indiarto, R., Pangawikan, A.D., Huda, S., Wiguna, B. and Geng, F. 2025. Solid lipid nanoparticles of mangosteen peel extract based on monoacylglycerol–diacylglycerol-rich fat and stearic acid: study on physicochemical properties and encapsulation efficiency. Future Foods 12, 100719; doi:10.1016/j.fufo.2024.100719 Sun, H., Huo, Y. and Fan, Z. 2021. Editorial: imaging and mechanism of leukocyte recruitment and function in inflammation and infections. Front. Cell Dev. Biol. 9, 690003; doi:10.3389/fcell.2021.690003 Sun, Y., Guo, B., Liang, X., Luo, Z., Han, J. and Qu, D. 2024. Evaluation of the antioxidant activity and prebiotic properties of mangosteen peel proanthocyanidin extracts through simulated in vitro digestion and colonic fermentation. LWT. -. Food. Sci. Technol. 212, 116992; doi:10.1016/j.lwt.2024.116992 Urban, J., Kareem, K.Y., Matuszewski, A., Bień, D., Ciborowska, P., Lutostański, K. and Michalczuk, M. 2025. Enhancing broiler chicken health and performance: the impact of phytobiotics on growth, gut microbiota, antioxidants, and immunity. Phytochem. Rev. 24, 2131–2145; doi:10.1007/s11101-024-09994-0 Vieco-Saiz, N., Belguesmia, Y., Raspoet, R., Auclair, E., Gancel, F., Kempf, I., Drider, D. and Everaert, N. 2019. Benefits and inputs from lactic acid bacteria and their bacteriocins as alternatives to antibiotic growth promoters during food-animal production. Front. Microbiol. 10; doi:10.3389/fmicb.2019.00057 Vinayamohan, P., Joseph, D., Viju, L.S., Baskaran, S.A. and Venkitanarayanan, K. 2024. Efficacy of probiotics in reducing pathogenic potential of infectious agents. Fermentation 10(12), 599; doi:10.3390/fermentation10120599 Wang, Z., Zhang, W., Chen, L., Lu, X. and Tu, Y. 2024. Lymphopenia in sepsis: a narrative review. Crit. Care 28(1), 99; doi:10.1186/s13054-024-05099-4 Watts, A. and Wigley, P. 2024. Avian pathogenic Escherichia coli: an overview of infection biology, antimicrobial resistance and vaccination. Antibiotics 13(809), 13; doi:10.3390/antibiotics13090809 Weiss, D.J and dan K.J. Wardrop. 2006. Schalm’s Veterinary Hematology 6th Edition. IA: Wiley-Blackwell. Widowati, W., Ginting, C.N., Lister, I.N.E., Girsang, E., Amalia, A., Wibowo, S.H.B., Kusuma, H. and Rizal, R. 2020. Anti-aging effects of mangosteen peel extract and its phytochemical compounds: antioxidant activity, enzyme inhibition and molecular docking simulation. Trop. Life Sci. Res. 31(3), 127–144; doi:10.21315/tlsr2020.31.3.9 Wiggins, R.W., Woo, J., Cauba, J.N. and Mito, S. 2025. Evaluating the potential of herbal extracts as treatment in immune thrombocytopenia: a review of evidence and limitations. Appl. Biosci. 4(1), 1; doi:10.3390/applbiosci4010001 Xu, F., Li, Z., Jiang, Y., Liao, T., Aschner, M. and Wei, Q. 2024. Ononin delays the development of osteoarthritis by down-regulating MAPK and NF-κB pathways in rat models. PLos One. 19(10), 310293; doi:10.1371/journal.pone.0310293 Yendri, E., Azizah, Z. and Mustika, M. 2022. Phytochemical and pharmacological effects of mangosteen (Garcinia mangostana L): a review. IOSR. J. Pharm. Biol. Sci. 17(3, Ser. III), 1–4. Yuvanatemiya, V., Srean, P., Klangbud, W.K., Venkatachalam, K., Wongsa, J., Parametthanuwat, T. and Charoenphun, N. 2022. A review of the influence of various extraction techniques and the biological effects of the xanthones from mangosteen (Garcinia mangostana L.) pericarps. Molecules 27(24), 8775; doi:10.3390/molecules27248775 Zhang, F. and Cheng, W. 2022. The mechanism of bacterial resistance and potential bacteriostatic strategies. Antibiotics 11(9), 1215; doi:10.3390/antibiotics11091215 Zhang, Z., Cao, M., Shang, Z., Xu, J., Chen, X., Zhu, Z., Wang, W., Wei, X., Zhou, X., Bai, Y. and Zhang, J. 2025. Research progress on the antibacterial activity of natural flavonoids. Antibiotics 14(4), 334; doi:10.3390/antibiotics14040334 Zhuo, Y., Zhao, Y.G. and Zhang, Y. 2024. Enhancing drug solubility, bioavailability, and targeted therapeutic applications through magnetic nanoparticles. Molecules, 29(20), 4854; doi: 10.3390/molecules29204854 | ||
| How to Cite this Article |
| Pubmed Style Kusmayadi A, Sundari RS, Sujana E, Yanti Y. Effect of mangosteen peel extract nanocapsule on the hematological profile and total bacterial count in broiler chickens experimentally infected with avian pathogenic Escherichia coli. Open Vet. J.. 2026; 16(1): 136-146. doi:10.5455/OVJ.2026.v16.i1.13 Web Style Kusmayadi A, Sundari RS, Sujana E, Yanti Y. Effect of mangosteen peel extract nanocapsule on the hematological profile and total bacterial count in broiler chickens experimentally infected with avian pathogenic Escherichia coli. https://www.openveterinaryjournal.com/?mno=289195 [Access: January 31, 2026]. doi:10.5455/OVJ.2026.v16.i1.13 AMA (American Medical Association) Style Kusmayadi A, Sundari RS, Sujana E, Yanti Y. Effect of mangosteen peel extract nanocapsule on the hematological profile and total bacterial count in broiler chickens experimentally infected with avian pathogenic Escherichia coli. Open Vet. J.. 2026; 16(1): 136-146. doi:10.5455/OVJ.2026.v16.i1.13 Vancouver/ICMJE Style Kusmayadi A, Sundari RS, Sujana E, Yanti Y. Effect of mangosteen peel extract nanocapsule on the hematological profile and total bacterial count in broiler chickens experimentally infected with avian pathogenic Escherichia coli. Open Vet. J.. (2026), [cited January 31, 2026]; 16(1): 136-146. doi:10.5455/OVJ.2026.v16.i1.13 Harvard Style Kusmayadi, A., Sundari, . R. S., Sujana, . E. & Yanti, . Y. (2026) Effect of mangosteen peel extract nanocapsule on the hematological profile and total bacterial count in broiler chickens experimentally infected with avian pathogenic Escherichia coli. Open Vet. J., 16 (1), 136-146. doi:10.5455/OVJ.2026.v16.i1.13 Turabian Style Kusmayadi, Andri, Ristina Siti Sundari, Endang Sujana, and Yanti Yanti. 2026. Effect of mangosteen peel extract nanocapsule on the hematological profile and total bacterial count in broiler chickens experimentally infected with avian pathogenic Escherichia coli. Open Veterinary Journal, 16 (1), 136-146. doi:10.5455/OVJ.2026.v16.i1.13 Chicago Style Kusmayadi, Andri, Ristina Siti Sundari, Endang Sujana, and Yanti Yanti. "Effect of mangosteen peel extract nanocapsule on the hematological profile and total bacterial count in broiler chickens experimentally infected with avian pathogenic Escherichia coli." Open Veterinary Journal 16 (2026), 136-146. doi:10.5455/OVJ.2026.v16.i1.13 MLA (The Modern Language Association) Style Kusmayadi, Andri, Ristina Siti Sundari, Endang Sujana, and Yanti Yanti. "Effect of mangosteen peel extract nanocapsule on the hematological profile and total bacterial count in broiler chickens experimentally infected with avian pathogenic Escherichia coli." Open Veterinary Journal 16.1 (2026), 136-146. Print. doi:10.5455/OVJ.2026.v16.i1.13 APA (American Psychological Association) Style Kusmayadi, A., Sundari, . R. S., Sujana, . E. & Yanti, . Y. (2026) Effect of mangosteen peel extract nanocapsule on the hematological profile and total bacterial count in broiler chickens experimentally infected with avian pathogenic Escherichia coli. Open Veterinary Journal, 16 (1), 136-146. doi:10.5455/OVJ.2026.v16.i1.13 |