| Case Report | ||
Open Vet. J.. 2026; 16(2): 1380-1388
Open Veterinary Journal, (2026), Vol. 16(2): 1380-1388 Case Report First molecularly-confirmed canine rickettsiosis in Southern Philippines (Cebu): A case reportAdrian P. Ybañez1,2,3*†, Wagner Bekim Y. Cardenas4†, Rochelle Haidee D. Ybañez5, Maryneth B. Barrios6 and Vernadyn A. Morillo71College of Veterinary Medicine at CTU Barili Campus, Cebu Technological University, Cebu, Philippines 2Institute for Molecular Genetics Parasitology and Vector-borne Diseases, Cebu Technological University, Cebu City, Philippines 3GPY Veterinare Animale Veterinary Clinic, Tres de Abril, Punta Princesa, Cebu City, Philippines 4Vet Co. Animal Clinic, Central Nautical Highway, Consolacion, Cebu, Philippines 5PhilVets Veterinary Products, Cebu City, Philippines 6College of Veterinary Medicine, Capiz State University – Dumarao Campus, Capiz, Philippines 7College of Veterinary Medicine and Biomedical Sciences, Cavite State University, Cavite, Philippines †Contributed equally to this study *Corresponding Author: Adrian P. Ybañez. Cebu Technological University, Cebu City, Philippines. Email: dr.adrianpybanez [at] gmail.com Submitted: 21/10/2025 Revised: 30/12/2025 Accepted: 11/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
ABSTRACTBackground: Canine rickettsiosis has previously been reported only in the northern Philippines. The present report documents the first case of canine rickettsiosis in the southern Philippines, confirmed by in-clinic and standard polymerase chain reaction (PCR). It aims to highlight the importance of PCR and accurate diagnosis, which affect the course of treatment for tick-borne diseases, including canine rickettsiosis. Case Description: A 3-year-old 4-kg male Shih Tzu was presented for hospitalization to an animal clinic in Cebu, Philippines. Presenting clinical signs included vomiting, lethargy, inappetence, and jaundice. About 14 months prior, the dog was diagnosed with canine babesiosis and canine ehrlichiosis from another clinic and was tested using commercial serological kits. Point-of-care initial serological tests were positive for Ehrlichia, Babesia, and Leptospira spp., leading to a preliminary diagnosis of multiple vector-borne diseases. Treatment was initiated with doxycycline, along with clindamycin and penicillin-based medications targeting the assumed pathogens. Additional treatments were mainly symptomatic. On day 12, in-clinic PCR testing procedures were performed. The sample was further tested using standard PCR. The PCR results revealed negative detection for Ehrlichia, Babesia, Leptospira, Mycoplasma, and Hepatozoon spp. but positive for Rickettsia sp. Standard PCR also showed positive results for Rickettsia sp. Because of the new PCR results, the prescribed treatment was modified before the patient was discharged. Conclusion: This case demonstrated the challenges of serological detection, where cross-reactivity, persistent antibodies, and possible vaccine-induced antibodies complicated the initial diagnosis. It shows that a positive serological test indicates exposure but not necessarily active infection. The case also illustrates that, while doxycycline provides a broad safety net for common vector-borne diseases, unnecessary antimicrobials may be administered if confirmatory PCR is not performed. Further studies should clarify the epidemiological status of this pathogen in the southern Philippines. Keywords: Rickettsia, Point-of-care test, Dog, Cebu, Philippines. IntroductionRickettsial infections are increasingly recognized as significant vector-borne diseases that affect both animals and humans (Galay et al., 2018; Marquez et al., 2021; Fa-ngoen et al., 2024). The genus Rickettsia, particularly the spotted fever group (SFG), is primarily transmitted by ticks, although fleas and mites can also serve as vectors (Tian et al., 2024). In dogs, rickettsioses may manifest with common clinical signs, including fever, lethargy, loss of appetite, and musculoskeletal discomfort. It is often accompanied by hematological abnormalities such as thrombocytopenia and anemia (Tsokana et al., 2022; Ebani et al., 2023). These signs overlap with other canine vector-borne diseases (CVBD) common in the Philippines, including ehrlichiosis, anaplasmosis, and babesiosis, making precise diagnosis challenging (Ybañez et al., 2012; Marquez et al., 2021; Apart from similar clinical signs, co-infection with different CVBDs is quite common, as the same vectors can carry them. This can lead to a more complex treatment protocol, depending on the presenting clinical signs (Cevidanes et al., 2022). In tropical urban and peri-urban areas, conditions favor the year-round proliferation of competent vectors, including the brown dog tick (Rhipicephalus sanguineus sensu lato). This tick is a known vector of Rickettsia spp. (Do et al., 2024). However, documented cases of canine rickettsial infections in the Philippines are limited. To date, only six dogs have been reported to be positive for Rickettsia sp., and all were confined to the northern area (Luzon) (Galay et al., 2018; Marquez et al., 2021). Previous reports did not fully describe the clinical signs observed in the infected dogs. Local veterinary clinicians may not fully consider these pathogens in the differential diagnosis of acute febrile illness with thrombocytopenia due to the lack of reliable testing (Marquez et al., 2021; Luong et al., 2023). This diagnostic gap underscores the need for broader surveillance and advanced molecular diagnostics to accurately identify and characterize rickettsial species circulating in the canine population in the Philippines, particularly in previously under-investigated regions such as Cebu (Galay et al., 2018). Serological assays can support a presumptive diagnosis but are constrained by cross-reactivity among SFG antigens and the inability to distinguish current from past exposure (Ravishankar et al., 2024). However, these assays are not readily available in the Philippines and other developing countries. Recently, in-clinic qualitative polymerase chain reaction (PCR) has become available in the Philippines, although only selected institutions can perform standard PCRs. PCR assays targeting conserved Rickettsia genes from whole blood or vector specimens provide more substantial evidence of active infection and enable downstream sequencing for species-level characterization (Blanda et al., 2020; Choat et al., 2023). Integrating PCR with routine hematology and biochemical profiling enhances diagnostic confidence and informs the selection of appropriate therapy and vector control measures (Alwasi et al., 2024). This case report, to the best of the authors’ knowledge, describes the first PCR-confirmed Rickettsia sp. infection in a dog in the southern Philippines (Cebu). This report details the patient’s clinical presentation, hematologic and serum biochemical findings, molecular testing, treatment course, and outcome. The implications for the differential diagnosis and treatment of CVBDs, as well as practical considerations for veterinary clinicians in resource-constrained settings, are also discussed. Informed consent was obtained from the owner for diagnostic and publication purposes of the case details. Case DetailsClinical case presentation and historyA 3-year-old male Shih Tzu dog was presented to an animal clinic. The clinical signs were vomiting, lethargy, inappetence, and jaundice. About 14 months prior, the dog was diagnosed with canine babesiosis and canine ehrlichiosis from another clinic and was tested using commercial serological kits (according to the owner). During that time, the patient was recumbent, anemic, and thrombocytopenic. The patient also showed vomiting and diarrhea. Treatment and clinical progressionHospitalization and treatment were initiated on September 9, 2025 (day 1). Intravenous fluid therapy with B complex, electrolytes, amino acids, and dextrose solution (Amilyte C), butaphosphan (Coforta), omeprazole, and maropitant (Cerenia). As the commercial serological test results were positive for Ehrlichia sp. and Babesia gibsoni, oral antibiotics, including doxycycline and clindamycin, were administered. Hematological and serum biochemical profiles revealed anemia, thrombocytopenia, and hepatic problems. Hence, dexamethasone (injectable) and oral supplements for the liver (Tefrosol) and anemia (Hemashield) were administered. By September 11, 2025 (day 3), serological testing for Leptospira spp. revealed positive results (Fig. 1). Hence, procaine and benzathine penicillin injectable (Duplocillin) was added. Enalapril and amlodipine were given to manage potential hypertension that the probable kidney problem may cause, while maropitant was replaced with metoclopramide.
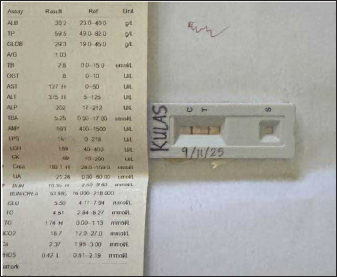
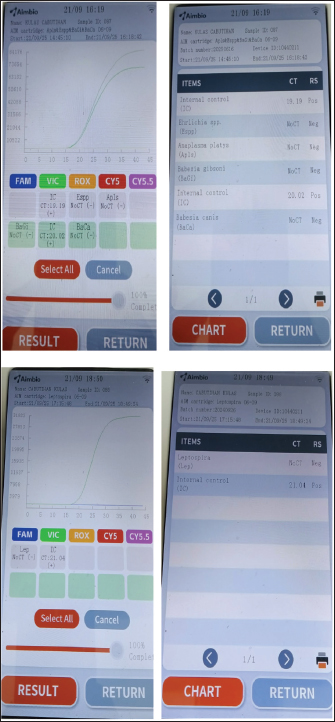
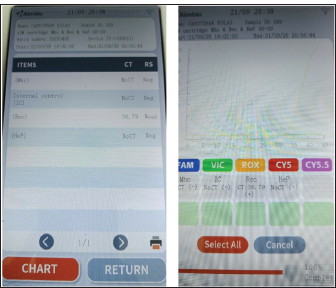
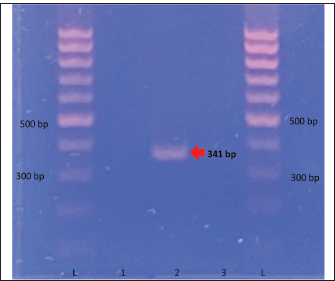
Fig. 1. Commercial test kit showing positive results for Leptospira sp. and serum biochemistry result strip at day 3. By September 12 (day 4), dexamethasone was discontinued and replaced with tolfenamic acid. The treatment remained the same for the next 4 days. By September 14 (day 6), clindamycin administration was stopped. By September 17 (day 9), all medications were continued except for metoclopramide. Enalapril and amlodipine were also given on this day. By September 18 (day 10), the patient was gradually weaned off most medications, receiving only oral doxycycline, liver and anti-anemia supplements, and injectable omeprazole, procaine, and benzathine penicillin. The same medications were given the next day, September 19 (day 11), with the addition of enalapril and amlodipine. The owner opted for additional tests. On September 20 (day 12), blood samples were obtained from the patient and sent to another veterinary clinic (GPY Veterinare Animale, Cebu City, Philippines) for PCR testing. The in-clinic PCR test (AimBio) revealed negative results for Ehrlichia canis, Anaplasma platys, Babesia canis, Babesia gibsoni, Mycoplasma haemocanis, and Hepatozoon sp. (Fig. 2) but positive for Rickettsia sp. (Fig. 3). Another fraction of the blood sample was further processed for DNA extraction and tested using standard PCR utilizing the gltA gene (Satoh et al., 2002) at the Institute for Molecular Genetics, Parasitology, and Vector-borne Diseases, Cebu Technological University, Cebu City, Philippines. Sequencing or species-level identification was not performed. PCR results revealed a positive band at the expected length (341 BP) (Fig. 4). A positive control was not included in the standard PCR run because Rickettsia DNA was unavailable at the local laboratory, which is a limitation of the test. However, the clear amplification of a band of appropriate size (341 bp) for the gltA gene is robust, supportive evidence. Contamination was also ruled out, as the negative control remained negative.
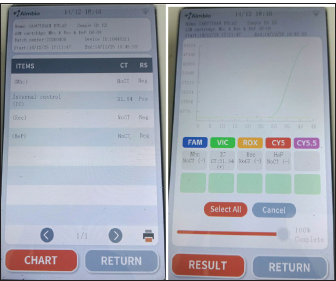
Fig. 2. In-clinic PCR showing negative results for Ehrlichia canis, Anaplasma platys, Babesia canis, Babesia gibsoni, and Leptospira spp.
Fig. 3. In-clinic PCR showing negative results for Mycoplasma haemocanis and Hepatozoon canis, and positive for Rickettsia spp. at Day 12.
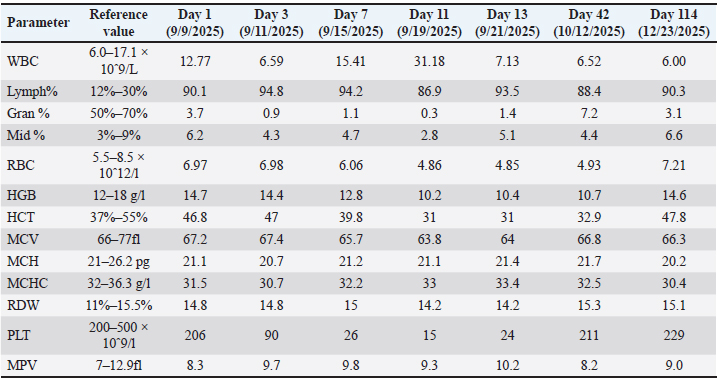
Fig. 4. Standard PCR gel showing a positive band for Rickettsia sp. (341 bp) (L=Ladder/Ladder, 1=Negative Control 1, 2=Patient Sample, 3=Negative Control 2). The patient was discharged on September 21, 2025 (Day 13). With the new findings, doxycycline, furosemide, a multiple-organ supplement (Organumshield), an anti-anemia supplement (Hemashield), and a Carica papaya-based supplement (Thrombocure) were prescribed for an additional three weeks. By October 12, 2025 (Day 42), the patient was re-evaluated. CBC showed normal platelet counts, but anemia persisted. The serum biochemical profile also showed normal values, indicating recovery from liver damage. After completing the prescribed medications, only the multiple-organ supplement and anti-anemia supplement were continued, with the addition of an immune supplement (Immunopetshield) (Table 3). By December 14, 2025 (Day 105) (Fig. 5), the patient was retested for Rickettsia sp. using the same in-clinic PCR for post-treatment evaluation. The results showed negative results. By December 23, 2025 (Day 114), CBC results showed a normal total white blood cell count, with no anemia or thrombocytopenia observed.
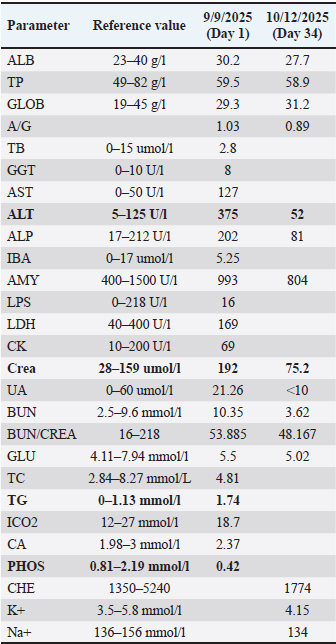
Fig. 5. In-clinic PCR showing negative results for Mycoplasma haemocanis and Hepatozoon canis, and positive for Rickettsia spp. at Day 105 Hematology and serum biochemistryElevated lymphocyte percentages were observed in all CBCs. Persistent thrombocytopenia and anemia were observed on days 3 and 11, respectively (Table 1). On day 11, leukocytosis was also observed (Table 1). On the other hand, increased alanine aminotransferase (ALT) (375 U/l), creatinine (192 umol/l), and triglyceride (1.74 0–1.13 mmol/l) and lowered serum phosphorus levels (0.42 mmol/l) were observed on day 1 (Table 2). At day 34, the ALT and creatinine values returned to normal. Table 1. Complete blood count results of a 3-year-old male Shih Tzu diagnosed with canine rickettsiosis in Cebu, Philippines.
Table 2. Serum biochemical profile of a three-year-old male Shih Tzu diagnosed with canine rickettsiosis in Cebu, Philippines.
Table 3. Treatment timeline and key diagnostic findings of a PCR-confirmed canine rickettsiosis case in Cebu, Philippines.
DiscussionThe present case highlights the critical challenges in diagnosing vector-borne diseases in resource-limited settings, particularly when advanced confirmatory tests, such as PCR, are often not readily available or not chosen by the animal’s owner. In such scenarios, veterinary clinicians frequently rely on rapid serological tests for initial diagnosis. While practical, these tests have significant interpretive limitations. The initial diagnoses of ehrlichiosis, babesiosis, and leptospirosis in the animal patient in this case were based on serological tests that detected antibodies. A key limitation of these tests is their inability to distinguish between active infection, previous exposure, and vaccine-induced immunity (Patterson et al., 2020; Sanchez-Vicente and Tokarz, 2023). Antibodies against pathogens such as Ehrlichia and Babesia spp. can persist for a long time even after an active infection resolves (Judy et al., 2023). In this case, the positive leptospirosis titer may have represented an anamnestic response to a prior vaccination rather than an active infection (Goy-Thollot et al., 2018; Sykes et al., 2023). The definitive diagnosis was established only by PCR testing on the 12th day of treatment, which showed the canine patient was negative for Ehrlichia, Babesia, and Leptospira spp., but positive for Rickettsia sp. This finding highlights a critical juncture in case management. PCR testing at the time of first presentation would have revealed results that would have changed the treatment course, making it more specific from the outset. On the other hand, the detection of the Rickettsia sp. pathogen using the proprietary Aimbio In-clinic PCR (with proprietary target gene) and the standard PCR targeting the gltA gene provides strong molecular evidence of the pathogen’s presence in Southern Philippines. Fortunately, doxycycline, which is used to treat ehrlichiosis and leptospirosis, is also the core treatment for rickettsiosis in dogs (Breitschwerdt et al., 1999). This drug was administered, even though rickettsiosis was not considered at the start. Hence, it was coincidental that the drug used also had an effect against Rickettsia sp. However, other medications, including clindamycin and penicillin, were initiated based on preliminary serological results (positive for babesiosis and leptospirosis) and would not have been indicated if the true, singular Rickettsia infection had been identified earlier. This case illustrates that while serology is a valuable initial screening tool, its results must be interpreted with caution, particularly in endemic areas. A positive result indicates evidence of exposure but not necessarily an active infection (Solano-Gallego et al., 2015; Robinson et al., 2019) in Philippine dogs with non-specific VBD signs. Where clinical response to initial empirical broad-spectrum treatment (e.g., doxycycline) is incomplete, or where cost is a limiting factor, molecular testing (PCR) should be considered to help narrow the diagnosis and avoid unnecessary or prolonged use of other antimicrobials. This becomes more challenging when clinical signs implicate multiple possible causative agents and serological test results support this. As in the present case, the clinical signs (jaundice, anemia, thrombocytopenia) were common to other probable causative agents, and the positive serological test results (Ehrlichia, Babesia, and Leptospira spp.) supported the initial diagnosis. As the presenting clinical signs of canine rickettsiosis are non-specific and closely resemble those of other tick-borne diseases (Pennisi et al., 2017; Silvestrini et al., 2023), its diagnosis is challenging, and confirmatory testing with PCR is recommended to establish a definite diagnosis. Rickettsia sp. has been detected in dogs and ticks in Manila, Philippines (Galay et al., 2018; Marquez et al., 2021), but the clinical details of infected dogs were not provided. The distribution of Rickettsia sp. in dogs may follow a pattern similar to that of Anaplasma platys and Ehrlichia canis, with its first molecular confirmation occurring only in a specific area in the Philippines (Cebu) (Ybañez et al., 2012), and it is now known to be widespread in the country. Currently, no rapid test kits are available for this pathogen in the Philippines. Hence, its confirmatory diagnosis in infected dogs is usually made using in-clinic PCR or standard PCR in the laboratory. As Rickettsia spp. are zoonotic, their detection is crucial as it can pose a health hazard to unsuspecting owners, especially when their dogs are infected. Some febrile human patients in the Philippines have been reported to have antibodies against SFG Rickettsia (SFGR) and typhus group Rickettsia (TGR) (Camer et al., 2003). On the other hand, the detection of Rickettsia spp. in cat fleas (Wolf and Reeves, 2012), cattle, water buffalo, and Rhipicephalus (Boophilus) microplus ticks (Galay et al., 2020) in the Philippines suggests its widespread presence in the area. Previous reports indicate the presence of Rickettsia spp., which were 99% identical with R. japonica and R. raoultii (Galay et al., 2018). Neighboring countries in Southeast Asia have already reported the detection of Rickettsia spp. in humans, dogs, cats, ticks, and fleas. These include Thailand (Mongkol et al., 2018), Fa-Ngoen et al., 2018, Foongladda et al., 2011) Vietnam (Parola et al., 2003; Nguyen et al., 2020; Nguyen et al. 2023; Do et al., 2024), Cambodia (Inpankaew et al., 2016), and Malaysia (Mokhtar and Tay, 2011; Khoo et al., 2020). These detections suggest that this pathogen may be endemic in Southeast Asia. Hence, routine surveillance is critical because of its zoonotic potential. ConclusionA canine rickettsiosis infection in a three-year-old Shi Tzu was molecularly confirmed using in-clinic qPCR and standard PCR. This is the first confirmed case of canine rickettsiosis in the southern Philippines. Serological tests for different pathogens were deemed necessary to establish clinical history, while PCR was informative for establishing the current disease status. Treatment must be carefully evaluated based on the presenting clinical signs and available diagnostic tests to ensure optimal recovery, while accounting for the limitations and advantages of the different test types. AcknowledgmentThe authors would like to thank the owner of “Ku Las” for his kind cooperation, Ms. Elmie S. Delan of Cebu Technological University for her assistance in the PCR procedures, Ms. Eljay Mae Bacalso for her assistance in the formatting of the final manuscript, and the staff of Vet Co. Animal Clinic and GPY Veterinare Animale Veterinary Clinic for their invaluable assistance. FundingThis research received no specific grant. Author’s contributionsAPY performed the molecular testing and mainly wrote the manuscript. He also monitored the patient and coordinated with the owner. WBC was the main attending veterinarian of the case and helped in providing information and improving the manuscript. RHDY, MBB, and VAM helped with the development, writing, and editing of the final manuscript. Conflict of interestThe authors declare that there is no conflict of interest. Data availabilityAll data supporting the findings of this study are available within the manuscript. ReferencesAlwasi, G.A., Alqahtani, A.M., Alalaliany, M.K.M., Saeed, M.S., Alshehri, A.A.H., Alqahtani, M.A.N., Asiri, Y.A.A., Alshehri, S.M.A. and Shukr, M.H. 2024. The role of laboratory testing in disease diagnosis: a comprehensive review. J. Int. Crisis. Risk. Commun. Res. 7(S11), 933. Blanda, V., D’Agostino, R., Giudice, E., Randazzo, K., La Russa, F., Villari, S., Vullo, S. and Torina, A. 2020. New real-time PCRs to differentiate Rickettsia spp. and Rickettsia conorii. Molecules 25(19), 4431. Breitschwerdt, E.B., Papich, M.G., Hegarty, B.C., Gilger, B., Hancock, S.I. and Davidson, M.G. 1999. Efficacy of doxycycline, azithromycin, or trovafloxacin for treatment of experimental Rocky Mountain spotted fever in dogs. Antimicrob. Agents. Chemother. 43(4), 813–821. Camer, G.A., Alejandria, M., Amor, M., Satoh, H., Muramatsu, Y., Ueno, H. and Morita, C. 2003. Detection of antibodies against spotted fever group Rickettsia (SFGR), typhus group Rickettsia (TGR), and Coxiella burnetii in human febrile patients in the Philippines. Jpn. J. Infect. Dis. 56(1), 26–28. Cevidanes, A., Di Cataldo, S., Muñoz-san Martín, C., Latrofa, M.S., Hernández, C., Cattan, P.E., Otranto, D. and Millán, J. 2022. Co-infection patterns of vector-borne zoonotic pathogens in owned free-ranging dogs in central Chile. Vet. Res. Commun. 47(2), 575. Choat, J., Yockey, B., Sheldon, S.W., Pappert, R., Petersen, J. and Dietrich, E.A. 2023. Development and validation of a real-time PCR test to detect Bartonella quintana in clinical samples. Diagn. Microbiol. Infect. Dis. 106(4), 116000. Do, T., Bui, L.K., Umemiya-Shirafuji, R., Inpankaew, T., Hasan, T., Zafar, I., Ma, Z., Hang, L., Mohanta, U.K., Amer, M., El-Sayed, S.A.E.S., Xuan, X. and Kamyingkird, K. 2024. The detection of zoonotic microorganisms in Rhipicephalus sanguineus (brown dog ticks) from Vietnam and the frequency of tick infestations in owned dogs. Front. Vet. Sci. 11, 1435441. Ebani, V.V., Nardoni, S. and Mancianti, F. 2023. Arthropod-borne pathogens in wild canids. Vet. Sci. 10(2), 165. Fa-Ngoen, C., Kaewmongkol, G., Inthong, N., Tanganuchitcharnchai, A., Abdad, M.Y., Siengsanan-Lamont, J., Blacksell, S.D. and Kaewmongkol, S. 2024. Serological detection of Rickettsia spp. and evaluation of blood parameters in pet dogs and cats from Bangkok and neighboring provinces. PLos One. 19(3), 1–13. Foongladda, S., Inthawong, D., Kositanont, U. and Gaywee, J. 2011. Rickettsia, Ehrlichia, Anaplasma, and Bartonella in ticks and fleas from dogs and cats in Bangkok. Vector-Borne Zoonotic Dis. 11(10), 1335–1341. Galay, R.L., Manalo, A.A.L., Dolores, S.L.D., Aguilar, I.P.M., Sandalo, K.A.C., Cruz, K.B., Divina, B.P., Andoh, M., Masatani, T. and Tanaka, T. 2018. Molecular detection of tick-borne pathogens in canine population and Rhipicephalus sanguineus (sensu lato) ticks from southern Metro Manila and Laguna, Philippines. Parasit. Vectors. 11(1). Galay, R.L., Talactac, M.R., Ambita-Salem, B.V., Chu, D.M.M., Costa, L.M.O.D., Salangsang, C.M.A., Caracas, D.K.B., Generoso, F.H., Babelonia, J.A., Vergano, J.L., Berana, L.C., Sandalo, K.A.C., Divina, B.P., Alvarez, C.R., Mago, E.R., Andoh, M. and Tanaka, T. 2020. Molecular detection of Rickettsia spp. and Coxiella burnetii in cattle, water buffalo, and Rhipicephalus (Boophilus) microplus ticks in Luzon Island of the Philippines. Trop. Med. Infect. Dis. 5(2), 54. Goy-Thollot, I., Djelouadji, Z., Nennig, M., Hazart, G. and Hugonnard, M. 2018. Screening for Leptospira DNA in blood and urine from 30 apparently healthy dogs. Revue Vét. Clin. 53(3), 79. Inpankaew, T., Hii, S.F., Chimnoi, W. and Traub, R.J. 2016. Canine vector-borne pathogens in semi-domesticated dogs residing in northern Cambodia. Parasit. Vectors. 9(1), 253. Judy, L., David, K., Peter, K. and Dhaval, S. 2023. Canine ehrlichiosis seropositivity and associated factors in Kenya and Tanzania: a retrospective study. BMC Vet. Res. 19(1). Khoo, J.J., Husin, N.A., Lim, F.S., Zulkifli, M.M.S., Azman, A.S., Mohd Azami, S.N.I., Mahfodz, N.H., Bell-Sakyi, L., Makepeace, B. and Abubakar, S. 2020. Rickettsial agents from fleas infesting stray dogs and cats in rural and urban communities in Malaysia. Int. J. Infect. Dis. 101, 529. Luong, N.H., Kamyingkird, K., Thammasonthijarern, N., Phasuk, J., Nimsuphan, B., Pattanatanang, K., Chimnoi, W., Kengradomkij, C., Klinkaew, N. and Inpankaew, T. 2023. Companion vector-borne pathogens and associated risk factors in apparently healthy pet animals (dogs and cats) in Khukhot City Municipality, Pathum Thani Province, Thailand. Pathogens 12(3), 391. Marquez, A.R.A., Eamens, K., Westman, M. and Šlapeta, J. 2021. Vector-borne pathogens in ticks and fleas of client-owned dogs in Metro Manila, Philippines. Parasitologia 1(4), 247–256. Mokhtar, A.S. and Tay, S.T. 2011. Molecular detection of Rickettsia felis, Bartonella henselae, and B. clarridgeiae in fleas from domestic dogs and cats in Malaysia. Am. J. Trop. Med. Hyg. 85(5), 931. Mongkol, N., Suputtamongkol, Y., Taweethavonsawat, P. and Foongladda, S. 2018. Molecular evidence of Rickettsia in human and dog blood in Bangkok. Vector-Borne. Zoonotic. Dis. 18(6), 297–302. Nguyen, V.L., Colella, V., Greco, G., Fang, F., Nurcahyo, W., Hadi, U.K., Venturina, V., Tong, K.B.Y., Tsai, Y.L., Taweethavonsawat, P., Tiwananthagorn, S., Tangtrongsup, S., Le, T.Q., Bui, K.L., Do, T., Watanabe, M., Rani, P.A.M.A., Dantas-Torres, F., Halos, L., Beugnet, F. and Otranto, D. 2020. Molecular detection of pathogens in ticks and fleas collected from companion dogs and cats in East and Southeast Asia. Parasit. Vectors 13(1), 420. Nguyen, V.T., Nguyen, H.Q., Nguyen, V.T. and Ng-Nguyen, D. 2023. Rickettsia felis and species of fleas parasitizing on household dogs in the central highlands of Vietnam. Comp. Immunol. Microbiol. Infect. Dis. 92, 101926. Parola, P., Cornet, J.P., Sanogo, Y.O., Miller, R.S., Thien, H.V., Gonzalez, J.P., Raoult, D., Telford, S.R. and Wongsrichanalai, C. 2003. Detection of Ehrlichia spp., Anaplasma spp., Rickettsia spp., and other eubacteria in ticks from the Thai-Myanmar border and Vietnam. J. Clin. Microbiol. 41(4), 1600–1608. Patterson, G., Tanhauser, M., Schmidt, P., Spangler, D., Faulkner, C., Faulkner, V., Kish, D., Gruszynski, K., Naikare, H., Coarsey, M.D. and Verma, A. 2020. Serosurvey of arthropod-borne diseases among shelter dogs in the Cumberland Gap Region of the United States. BMC. Vet. Res. 16(1). Pennisi, M.G., Hofmann-Lehmann, R., Radford, A.D., Tasker, S., Belák, S., Addie, D.D., Boucraut-Baralon, C., Egberink, H., Frymus, T., Gruffydd-Jones, T., Hartmann, K., Horzinek, M.C., Hosie, M.J., Lloret, A., Lutz, H., Marsilio, F., Thiry E Truyen. and Möstl, K. 2017. Anaplasma, Ehrlichia and Rickettsia species infections in cats: european guidelines from the ABCD on prevention and management. J. Feline Med. Surg. 19(5), 542–548. Ravishankar, V., Narayanan, S. and Shandil, R.K. 2024. Rickettsial infections: prevalence and diagnosis of scrub typhus in India. Front. Trop. Dis. 5, 1433013. Robinson, M.T., Satjanadumrong, J., Hughes, T., Stenos, J. and Blacksell, S.D. 2019. Diagnosis of spotted fever group Rickettsia infections: the Asian perspective. Epidemiol. Infect. 147, 286. Sanchez-Vicente, S. and Tokarz, R. 2023. Tick-borne co-infections: challenges in molecular and serologic diagnoses. Pathogens 12(11), 1371. Satoh, H., Motoi, Y., Camer, G.A., Inokuma, H., Izawa, M., Kiyuuna, T., Kumazawa, N., Muramatsu, Y., Ueno, H. and Morita, C. 2002. Characterization of spotted fever group rickettsiae detected in dogs and ticks in Okinawa, Japan. Microbiol. Immunol. 46(4), 257–263. Silvestrini, P., Lloyd-Bradley, B., Glanemann, B., Barker, E.N., Badham, H., Tappin, S., Pascual, M., Haines, A., Mas, A., Roura, X. and Piviani, M. 2023. Clinical presentation, diagnostic investigations, treatment protocols and outcomes of dogs diagnosed with tick-borne diseases living in the United Kingdom: 76 cases (2005–2019). J. Small. Anim. Pract. 64(6), 392–400. Solano-Gallego, L., Caprì, A., Pennisi, M.G., Caldin, M., Furlanello, T. and Trotta, M. 2015. Acute febrile illness is associated with Rickettsia spp infection in dogs. Parasit. Vectors 8(1), 216. Sykes, J.E., Francey, T., Schuller, S., Stoddard, R.A., Cowgill, L.D. and Moore, G.E. 2023. Updated ACVIM consensus statement on leptospirosis in dogs. J. Vet. Inter. Med. 37(6), 1966. Tian, Y., Kaufman, P.E. and Lord, C.C. 2024. Vector-borne Rickettsia pathogens. EDIS. 2, 1–6. Tsokana, C.N., Kapna, I. and Valiakos, G. 2022. Current data on Rickettsia felis occurrence in vectors, human and animal hosts in Europe: a scoping review. Microorganisms 10(12), 2491. Wolf, S.P. and Reeves, W.K. 2012. Rickettsia felis (Rickettsiales: rickettsiaceae) discovered in cat fleas (Siphonaptera: Pulicidae) in the Philippines. J. Entomol. Sci. 47(1), 95–96. Ybañez, A.P., Perez, Z.O., Gabotero, S.R., Yandug, R.T., Matsumoto, K. and Inokuma, H. 2012. First molecular detection of Ehrlichia canis and Anaplasma platys in ticks from dogs in Cebu, Philippines. Ticks. Tick-Borne. Dis. 3(3), 288. | ||
| How to Cite this Article |
| Pubmed Style Ybañez AP, Cardenas WBY, Ybañez RHD, Barrios MB, Morillo VA. First molecularly-confirmed canine rickettsiosis in Southern Philippines (Cebu): A case report. Open Vet. J.. 2026; 16(2): 1380-1388. doi:10.5455/OVJ.2026.v16.i2.54 Web Style Ybañez AP, Cardenas WBY, Ybañez RHD, Barrios MB, Morillo VA. First molecularly-confirmed canine rickettsiosis in Southern Philippines (Cebu): A case report. https://www.openveterinaryjournal.com/?mno=291702 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.54 AMA (American Medical Association) Style Ybañez AP, Cardenas WBY, Ybañez RHD, Barrios MB, Morillo VA. First molecularly-confirmed canine rickettsiosis in Southern Philippines (Cebu): A case report. Open Vet. J.. 2026; 16(2): 1380-1388. doi:10.5455/OVJ.2026.v16.i2.54 Vancouver/ICMJE Style Ybañez AP, Cardenas WBY, Ybañez RHD, Barrios MB, Morillo VA. First molecularly-confirmed canine rickettsiosis in Southern Philippines (Cebu): A case report. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 1380-1388. doi:10.5455/OVJ.2026.v16.i2.54 Harvard Style Ybañez, A. P., Cardenas, . W. B. Y., Ybañez, . R. H. D., Barrios, . M. B. & Morillo, . V. A. (2026) First molecularly-confirmed canine rickettsiosis in Southern Philippines (Cebu): A case report. Open Vet. J., 16 (2), 1380-1388. doi:10.5455/OVJ.2026.v16.i2.54 Turabian Style Ybañez, Adrian P., Wagner Bekim Y. Cardenas, Rochelle Haidee D. Ybañez, Maryneth B. Barrios, and Vernadyn A. Morillo. 2026. First molecularly-confirmed canine rickettsiosis in Southern Philippines (Cebu): A case report. Open Veterinary Journal, 16 (2), 1380-1388. doi:10.5455/OVJ.2026.v16.i2.54 Chicago Style Ybañez, Adrian P., Wagner Bekim Y. Cardenas, Rochelle Haidee D. Ybañez, Maryneth B. Barrios, and Vernadyn A. Morillo. "First molecularly-confirmed canine rickettsiosis in Southern Philippines (Cebu): A case report." Open Veterinary Journal 16 (2026), 1380-1388. doi:10.5455/OVJ.2026.v16.i2.54 MLA (The Modern Language Association) Style Ybañez, Adrian P., Wagner Bekim Y. Cardenas, Rochelle Haidee D. Ybañez, Maryneth B. Barrios, and Vernadyn A. Morillo. "First molecularly-confirmed canine rickettsiosis in Southern Philippines (Cebu): A case report." Open Veterinary Journal 16.2 (2026), 1380-1388. Print. doi:10.5455/OVJ.2026.v16.i2.54 APA (American Psychological Association) Style Ybañez, A. P., Cardenas, . W. B. Y., Ybañez, . R. H. D., Barrios, . M. B. & Morillo, . V. A. (2026) First molecularly-confirmed canine rickettsiosis in Southern Philippines (Cebu): A case report. Open Veterinary Journal, 16 (2), 1380-1388. doi:10.5455/OVJ.2026.v16.i2.54 |