| Research Article | ||
Open Vet. J.. 2026; 16(3): 1556-1564 Open Veterinary Journal, (2026), Vol. 16(3): 1556-1564 Research Article Prevalence of staphylococcal enterotoxin genes and phylogenetic analysis of coagulase-negative Staphylococcus aureus from buffalo subclinical mastitis milk in Nineveh Governorate, IraqMohammed Jasim Mohammed Awed*, Ayman Hani Taha and Omar Hashim SheetDepartment of Veterinary Public Health, College of Veterinary Medicine, University of Mosul, Mosul, Iraq *Corresponding Author: Mohammed Jasim Mohammed Awed. Department of Veterinary Public Health, College of Veterinary Medicine, University of Mosul, Mosul, Iraq. Email: mohammed.jassim.m [at] uomosul.edu.iq Submitted: 26/10/2025 Revised: 30/01/2026 Accepted: 10/02/2026 Published: 31/03/2026 © 2026 Open Veterinary Journal
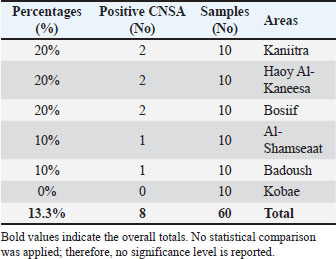
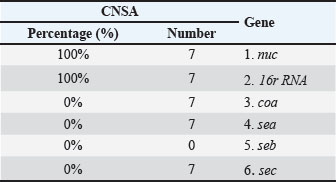
AbstractBackground: Staphylococcus bacteria-induced food poisoning is considered one of the most prevalent foodborne infections worldwide. The neutral pH of milk and abundance of nutrients support the growth of various bacteria. Aim: The goals of this study are to detect the nuc, coa, and 16S rRNA genes, find the sea, seb, and sec genes in coagulase-negative Staphylococcus aureus (CNSA), and compare the local strains with those from previous studies. Methods: Sixty samples of buffaloes with subclinical mastitis were obtained from numerous regions of Nineveh Province. Classical and molecular biology methods used for isolating and identifying of Staphylococcus aureus. Results: The results indicated that 13.3% (8/60) of buffalo milk contained isolated CNSA. While no CNSA isolates were discovered in Kobae, the highest percentage of CNSA isolates were observed in the Kaneitra Haoy, Al-Kaneesa, and Bosiif regions. Furthermore, the polymerase chain reaction (PCR) approach revealed that all CNSA isolates possessed the nuc and 16S rRNA genes (100%, 8/8); nevertheless, no CNSA isolates had the coa, sea, seb, or sec genes. The The National Center for Biotechnology Information GenBank database contains 7 newly discovered 16S rRNA gene sequences of S. aureus. These sequences are accessible with accession codes PV330711, PV330712, PV330713, PV330714, PV330715, PV330716, and PV330717. Furthermore, the newly isolated S. aureus sequences in this study are related to other S. aureus sequences isolated from various countries. Conclusion: CNSAs as mastitis-causing agents cannot be ignored as they can cause significant economic losses. The PCR method can be used to identify the virulence factors of CNSA isolates that help in determining the prevalence, severity, and implementation of control and prevention measures. Keywords: Buffalo milk, Coagulase negative S. aureus, Phylogenetic analysis, Staphylococcal enterotoxins genes. IntroductionBuffalo milk and its products are gaining more attention and value around the world (De Camargo et al., 2015). Milk and dairy products from cows and buffalo are key sources of protein in many developing countries (Preethirani et al., 2015). Buffaloes are generally classified into 2 main species, which are distinguished by their geographic distribution, physical traits, and genetic makeup. In Asia, buffaloes are the dominant livestock species, responsible for nearly 97% of the global milk output from buffaloes—roughly 90 million tons/year (Qar and Governorates, 2007). In Iraq, water buffalo (Bubalus bubalis) inhabit diverse environments, including marshlands, rural villages, and even some urban areas (Sharma et al., 2012). Buffalo milk plays a major commercial role in several regions of Iraq—particularly in Mosul—where it is widely used to produce dairy products (Alfekaiki, 2018). Nearly 20 factories generate approximately five tons of buffalo milk each day, with half of it being shipped to other regions of Iraq and the Kurdistan Region, playing a key role in boosting the local economy. Intramammary infections (IMIs) are primarily caused by bacteria such as Staphylococcus aureus, streptococci, coliforms, Mannheimia, Arcanobacterium pyogenes, and Pasteurella species. The dairy industry still faces major economic losses due to mastitis. Researchers have identified over 50 Staphylococcus species and subspecies responsible for staphylococcal mastitis. Across Asia, Staphylococcus aureus is identified as a primary pathogen linked to mastitis in both cattle and water buffaloes (Sharma and Maiti, 2010). Staphylococcus bacteria are categorized as either coagulase-negative or coagulase-positive in accordance with their capacity to coagulate plasma (Pyorala and Taponen, 2009). Coaguase-negative staphylococci (CNS) are the most widely occurring bacterial agents involved in subclinical mastitis among sheep and goats (Albenzio et al., 2002; Bergonier et al., 2003). Although classified as a coagulase-negative Staphylococcus aureus (CNSA) is capable of causing mastitis in animals and is often associated with poor treatment outcomes due to its non-contagious nature (Raad et al., 1998). In subclinical mastitis among sheep in Iraq, CNSA has been detected with a prevalence rate of 20% (Abdulrazzaq et al., 2025). The genotypic characteristics of S. aureus play a critical role in its pathogenicity and detection during diagnosis. Thermonuclease (nuc) is a well-known virulence factor specific to S. aureus and used for identifying the bacterium in various food samples (Hu et al., 2012). Although the precise role of the coagulase (coa) gene in IMI remains unclear, most S. aureus isolates obtained from these infections were capable of coagulating bovine plasma (Sutra and Poutrel, 1994). In addition to other virulence factors, S. aureus secretes numerous exoproteins, with staphylococcal enterotoxins (SEs) playing a significant role in food-borne diseases (Argudín et al., 2010). Traditional staphylococcal enterotoxins are believed to account for 95% of food poisoning cases, with the remaining 5% linked to more recently discovered SEs (Papadopoulos et al., 2019). The identification of CNS primarily relies on phenotypic biochemical reactions; however, misidentification can occur due to variations in the expression of these phenotypic traits (Zadoks and Watts, 2009). Owing to the time-consuming nature and inherent limitations of conventional culture techniques, molecular biology methods offer a faster and more reliable approach for detecting S. aureus, reducing the identification time from several days to just a few hours (Gao et al., 2011). This study aimed to isolate CNSA from buffaloes suffering from subclinical mastitis and identify virulence-related genes—nuc, coa, and 16S rRNA—along with enterotoxin genes (e.g., sea, seb, and sec). This study also seeks to compare local strains with those from earlier studies by constructing a phylogenetic tree to explore possible genetic relationships. Materials and MethodsSamples collectionThis study involved the collection of 60 samples from buffaloes with subclinical mastitis diagnosed using the California Mastitis Test, conducted between March and June 2025. The samples were evenly distributed, with 10 samples collected from each of the following regions in Nineveh Province: Kaniitra, Haoy Al-Kaneesa, Bosiif, Al-Shamseaat, Badoush, and Kobae. A total of 25 ml of milk samples was obtained from buffalo and placed into collecting tubes. To maintain sample integrity, the milk samples were transported in a chilled state using a cold box filled with dry ice (CO2) and delivered to the central laboratory. The enriched peptone water samples were incubated overnight at 37°C to increase the bacterial population (Quinn et al., 2011). Subsequently, milk samples were streaked onto blood agar and mannitol salt agar plates for selective isolation of staphylococcal species. Isolation and characterization of CNSACNSA isolates were identified through standard microbiological culture methods, assessing colony cellular structure, cell wall staining response, and catalase and coagulase test results (Quinn et al., 2002). DNA isolationAfter extracting the genomic Deoxyribonucleic Acid (DNA) from CNSA isolates, those that tested positive were incubated on mannitol salt agar for more than eight hours at 37°C. Genomic DNA extraction was carried out using the Genomic DNA Extraction Kit (Addbio, Korea), which is tailored for Gram-positive bacteria, following the manufacturer’s protocol. DNA quantity was assessed using a NanoDrop (Jenway, UK), with concentrations of CNSA isolates ranging from 50 μg/μl to 85 μg/μl. The extracted DNA was then held at –20°C for later analysis. Reaction of the PCRThe polymerase chain reaction (PCR) was used to detect nuc, coa, 16S rRNA, sea, seb, and sec genes in CNSA isolates. The expected amplicon sizes were: 166 bp for nuc (Graber et al., 2007), 674 bp for coa (Javid et al., 2018), 1403 bp for 16S rRNA (Onni et al., 2010), 219 bp for sea (Tsen and Chen, 1992), 478 bp for seb, and 257 bp for sec (Johnson et al., 1991) (Table 1). The PCR reaction was prepared in a 200-μl tube with a total volume of 50 μl (Biozym, Germany). Each reaction contained 8 μl of CNSA DNA template, 25 μl of GoTaq Green Master Mix (2×) (Promega, USA), 2 μl of each primer, and 13 μl of nuclease-free water (Promega, USA). Amplicons were examined by electrophoresis on a 2% agarose gel (Peqlab, Germany) using a 100 bp DNA ladder as a reference marker for fragment size. Table 1. PCR conditions and primer sequences used for detecting various genes in CNSA.
DNA sequencingAmplicons of the 7 target genes, which were confirmed to be positive for S. aureus through conventional PCR, were sent to Macrogen Inc. (Seoul, South Korea), a commercial sequencing service for purification and subsequent DNA sequencing. Nucleotide sequencing was performed on the 16S rRNA gene, and the resulting sequences were analyzed using the BLASTn program through the The National Center for Biotechnology Information (NCBI) database (http://www.ncbi.nlm.nih.gov) to identify similarities with known S. aureus 16S rRNA sequences deposited in GenBank. To further examine genetic relationships, multiple sequence alignment was carried out using the CLUSTALW algorithm within the NGPhylogeny.fr software package (https://ngphylogeny.fr/), facilitating precise comparisons of sequence similarity and variability among the isolates. Phylogenetic relationships were then inferred using the neighbor-joining method through the CLUSTALW tool hosted on the GenomeNet platform. To strengthen the accuracy and reliability of the phylogenetic tree, 500 S. aureus 16S rRNA gene sequences were used as an outgroup. This integrated methodology—encompassing gene purification, sequencing, and detailed bioinformatics analysis—aimed to clarify the genetic relationships and phylogenetic placement of S. aureus isolates collected from restaurant environments. Statistical analysisStatistical analysis was performed using JMP® 16.1 software (JMP®, 2021). The chi-square test revealed a significant difference in the distribution of CNSA isolates among different regions in Nineveh Governorate, with a p-value less than 0.05. Ethical approvalThe Institutional Animal Care and Use Committee (IACUC) of the College of Veterinary Medicine, University of Mosul, approved all animal procedures under approval number UM.Vet.2025.008 in date 15.02.2025. Sample collection was carried out with informed consent from the animal owners, and all protocols adhered strictly to ethical standards. ResultsColonies of CNSA that yielded strongly positive results exhibited a characteristic pink-red coloration on Mannitol Salt Agar. Furthermore, a series of biochemical tests—including a negative coagulase assay and a positive catalase test—confirmed the identification of CNSA isolates. Our findings indicated that 13.3% (8/60) of the CNSA isolates examined in this study exhibited a measurable incidence. The Kaneitra Haoy, Al-Kaneesa, and Bosiif regions exhibited the highest proportion of CNSA isolates from buffalo subclinical mastitis milk, with an incidence rate of 20% (2/10). Consequently, 10% (1 out of 10) of the samples from Al-Shamseaat and Badoush showed CNSA prevalence, whereas no CNSA isolates were detected in Kobae (Table 2). PCR analysis validated the results of conventional diagnostic techniques, revealing that all CNSA isolates (100%, 8/8) harbored the nuc gene (Fig. 1) (Table 2). Table 3 shows that all CNSA isolates possessed the 16S rRNA gene, with a detection rate of 100% (8/8) (Figs. 2 and 3). Furthermore, none of the CNSA isolates in the present study carried the coa, sea, seb, or sec genes (Figs. 3–6). Table 2. Occurrence rate of CNSA isolates in buffalo milk samples.
Table 3. Prevalence of target genes among CNSA isolates.
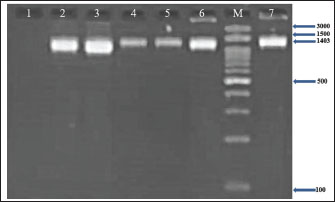
Fig. 1. Agarose gel electrophoresis (2%) showing the typical amplicon of the nuc gene product of CNSA isolates. The amplification of DNA appears as a ladder-like pattern. Lane 1 is negative control, Lanes 2–6 are represent positive isolates, Lane M are DNA Marker 100 bp ladder (Biozym Diagnostic), Lane 7 is the positive control.
Fig. 2. Agarose gel electrophoresis (2%) showing the typical amplicon of the 16S rRNA gene product of CNSA isolates. The amplification of DNA appears as a ladder-like pattern. Lane 1 is negative control, Lanes 2–6 are represent positive isolates, Lane M are DNA Marker 100 bp ladder (Biozym Diagnostic), Lane 7 is the positive control.
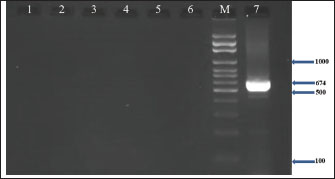
Fig. 3. Agarose gel electrophoresis (2%) showing the typical amplicon of the coa gene product of CNSA isolates. The amplification of DNA appears as a ladder-like pattern. Lane 1 is negative control, Lanes 2–6 are represent negative isolates, Lane M are DNA Marker 100 bp ladder (Biozym Diagnostic), Lane 7 is the positive control.
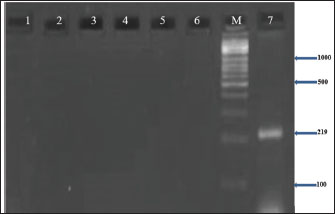
Fig. 4. Agarose gel electrophoresis (2%) showing the typical amplicon of the sea gene product of CNSA isolates. The amplification of DNA appears as a ladder-like pattern. Lane 1 is negative control, Lanes 2–6 are represent negative isolates, Lane M are DNA Marker 100 bp ladder (Biozym Diagnostic), Lane 7 is the positive control.
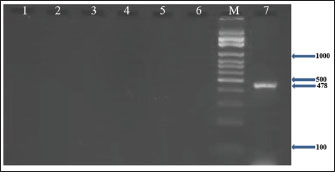
Fig. 5. Agarose gel electrophoresis (2%) showing the typical amplicon of the seb gene product of CNSA isolates. The amplification of DNA appears as a ladder-like pattern. Lane 1is negative control, Lanes 2–6 are represent negative isolates, Lane M are DNA Marker 100 bp ladder (Biozym Diagnostic), Lane 7 is the positive control.
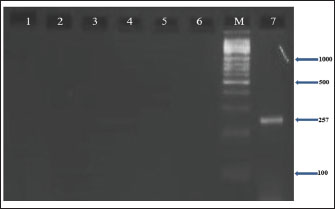
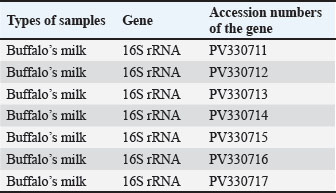
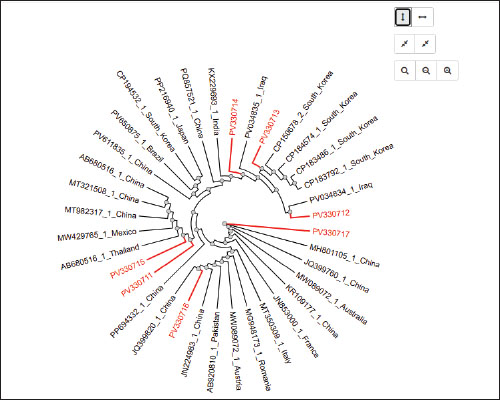
Fig. 6. Agarose gel electrophoresis (2%) showing the typical amplicon of the sec gene product of CNSA isolates. The amplification of DNA appears as a ladder-like pattern. Lane 1is negative control, Lanes 2–6 are represent negative isolates, Lane M are DNA Marker 100 bp ladder (Biozym Diagnostic), Lane 7 is the positive control. Based on the sequencing data generated in this study, BLASTn analysis was performed on 7 newly identified 16S rRNA gene sequences of S. aureus isolated from buffalo milk. As shown in Table 4, the sequences identified were sent to the NCBI GenBank database and are available under the sequence accession codes PV330711, PV330712, PV330713, PV330714, PV330715, PV330716, and PV330717. Additionally, the most probable estimation phylogenetic analysis using the NGPhylogeny software indicated that the native gene variants exhibited substantial divergence from previously reported sequences in the GenBank database (Fig. 7). Table 4. NCBI database accession numbers for S. aureus 16S rRNA gene sequences identified in buffalo milk samples.
Fig. 7. Phylogenetic clustering of Staphylococcus aureus isolates based on GenBank sequences. In addition, a relationship was observed between the PV330711 and PV330715 sequence types in this study with the AB680516, MT321508, and MT982317 sequence types from China, the MW429765 sequence types from Mexico, and the AB680516 sequence types from Thailand. Furthermore, the sequence type of this study, PV330712, is related to the sequence type PV034834 from Iraq, while the sequence types PV330715 and PV330713 are related to the sequence types from South Korea: CP150678, CP184574, CP183486, and CP183792. Moreover, the sequence type of this study, PV330716, is related to the JQ399820 sequence types from China, while the sequence type PV330717 is related to the MH801105, JQ399760, and KR109177 sequence types from China, the MW089072 sequence type from Australia, and the JN853000 sequence type from France (Fig. 6). DiscussionNumerous studies have revealed that S. aureus occurs in the environment, on the skin of humans, and on animals. Staphylococcus aureus is acknowledged as an important foodborne bacterium worldwide and causes food poisoning for consumers. Previous studies reported that S. aureus isolated from several kinds of food, such as broiler carcasses (Sheet et al., 2021a,b), meat and butchers’ shops (Sheet et al., 2023), local fish (Taha et al., 2024), and foods (Sheet et al., 2025). In addition, S. aureus has been identified as the principal etiological agent of all kinds of mastitis in animals, including bovine mastitis (Sheet, 2022), camel milk (Sheet et al., 2021a,b), goat subclinical mastitis milk (Taha et al., 2025), and sheep subclinical mastitis milk (Abdulrazzaq et al., 2025; Alalaf et al., 2025). The financial impact of mastitis disease on dairy farmers is significant on a global scale. This phenome is particularly significant because animals with non-clinical mastitis do not display obvious signs of infection (Momtaz et al., 2012). According to numerous earlier investigations, S. aureus, especially CNS infections, may give rise to increased inflammation (Poutrel et al., 1997), destruction of the udder tissue (Burriel, 1998), and loss of entire milk production in dairy animals (Gonzalo et al., 2002). The prevalence of S. aureus isolated from buffalo sub-clinical mastitis milk was 13.3%. The findings of this investigation were almost identical to those of a study conducted in Iraq, which found that 11.7% of sheep had subclinical mastitis-via CNSA (Abdulrazzaq et al., 2025). In Egypt, the percentage of CNSA in milk obtained from cows and buffaloes was 5.3% (2/38) (Abdel-Tawab et al., 2018), in Poland, the prevalence of CNS isolates was 18.54% (Krukowski et al., 2020), and in Germany, the prevalence was 46.8% (Tenhagen et al., 2009). Spatial variation, hygienic procedures in farm milk production facilities, and the type of techniques applied to detect CNSA in the samples are the main causes of the variations in CNSA prevalence rates. The phenotypic characterization of CNSA isolates and their isolation and identification from samples are carried out using traditional methods (Młynarczyk et al., 1998; Aarestrup et al., 1999). The biochemical techniques used to detect CNSA are not trustworthy for identifying the species and subspecies of bacteria or for providing detailed information about the genes in CNSA (Notarnicola et al., 1985). Molecular biology methods, including the PCR assay, are utilized to detect the coa gene, which aids in distinguishing CNSA from coagulase-positive Staphylococcus aureus (CPSA) (Luijendijk et al., 1996). The distribution of CNSA in milk and its products can happen through transportation, processing in dairy factories, storage in unsanitary environments, and milking on dairy farms under unhygienic conditions (Regasa et al., 2019). Furthermore, the finding of CNS in various buffalo body parts, including mouths, muzzles, and nostrils, as well as in the hands of prepared milkers, results in the spread of CNS throughout the environment and from dairy herds to their young through feeding or lactation (Pizauro et al., 2019). Furthermore, the PCR test confirmed that all CNSA isolates contained 100% of the nuc and 16S rRNA genes; however, none of them had the coa, sea, seb, or sec genes. According to prior studies, S. aureus has been determined to possess the specific-species nuc gene, which includes CPSA and CNSA (Tsen and Chen, 1992; Sheet, 2022; Sheet et al., 2023; Sheet et al., 2024). Furthermore, the coa gene was absent in every CNSA isolate, emphasizing its importance as a genetic indicator for differentiating between CNSA and CPSA. The findings of this study align with those of earlier research that revealed that no CNSA isolate from sheep subclinical mastitis contained the coa gene (Abdulrazzaq et al., 2025). Furthermore, no CNSA isolates possessed the sea gene in the present study, which is consistent with a prior study conducted in India that demonstrated that S. aureus isolates from cow mastitis lacked the sea gene; nonetheless, seb and sec genes were detected in 9.09% and 1.82% of isolates, respectively (Neelam et al., 2022). In addition, S. aureus isolates from buffalo in Bangladesh carried the sea and sec genes in 11.9% and 7.4% of isolates, respectively (Hoque et al., 2022). This study discovered seven novel sequences (accession numbers PV330711–PV330717) that revealed varying degrees of sequence similarity to S. aureus that were already submitted to GenBank from across the world. The degree of sequence variation indicates that most current sequences are either evolutionarily conserved staphylococcal gene variants or uniformly circulating, indicating shared ancestral sources or broad transmission throughout CNSA populations. Several factors, such as geographic dispersion, ecological niche, and selective pressures, affect the variety of isolates (Hoque et al., 2022; Taha et al., 2024). ConclusionIn Iraq’s Nineveh Province, the buffalo is acknowledged as one of the most important ruminants. It generates vast amounts of milk with a high fat content, which is used to make cream. Staphylococcus aureus (CPSA and CNSA) present a serious threat to human and animal health worldwide and hinder the production of milk and its derivatives through food poisoning caused by staphylococcal enterotoxins. A variety of factors contribute to the high prevalence of CNSA infections, particularly poor agricultural methods, disregard for fundamental hygiene precautions, and the haphazard use of medications without first contacting a veterinarian. Furthermore, the virulence factors of CNSA isolated from buffalo milk exhibit genetic heterogeneity depending on the various geographical areas and environmental conditions worldwide. Sustained investigation and monitoring are essential to track and employ the contemporary method for quickly identifying and assessing CNSA strains, as well as to create efficient management plans for them in dairy farms and facilities to avoid contamination and minimize their effects on the general public and dairy production. AcknowledgmentWe would like to sincerely thank the College of Veterinary Medicine, University of Mosul, for providing the essential facilities and equipment required to conduct this study. Conflict of interestThe authors have no conflicts of interest to declare. FundingThe research is fully self-funded by the authors. Authors’ contributionOmar H.S. designed the study. Ayman H.T. and Mohammed J.M. conducted DNA isolation and PCR. All authors contributed to manuscript preparation and approved the final version. Data availabilityThe data generated in this study are included in the manuscript. ReferencesAbdel-Tawab, A.A., Darwish, S.F., El –Hofy, F.I. and. Shoieb, E.M. 2018. Phenotypic and genotypic characterization of coagulase negative Staphylococcus aureus isolated from different sources. Benha. Vet. Med. J. 34(3), 129–149. Aarestrup, F.M., Larsen, H.D., Eriksen, N.H.R., Elsberg, C.S. and Jensen, N.E. 1999. Frequency of α-and β-haemolysin in Staphylococcus aureus of bovine and human origin: a comparison between pheno-and genotype and variation in phenotypic expression. Acta. Pathol. Microbiol. Immunol. Scand. 107(1-6), 425–430. Available via https://pubmed.ncbi.nlm.nih.gov/10230698/ Abdulrazzaq, K.M., Taha, A.H. and Sheet, O.H. 2025. Phylogenetic analysis and genotypic characterization of coagulase-negative Staphylococcus aureus isolates from sheep subclinical mastitis milk in Nineveh governorate, Iraq. Iraqi. J. Vet. Sci. 39(3), 555–562; doi:10.33899/ijvs.2025.158348.4186 Alalaf, A.S., Taha, A.H. and Sheet, O.H. 2025. Genotypic characteristics and phylogenetic tree analysis of Staphylococcus aureus isolates from sheep subclinical mastitis milk. Open. Vet. J. 15(1), 289; doi:10.5455/OVJ.2025.v15.i1.27 Albenzio, M., Taibi, L., Muscio, A. and Sevi, A. 2002. Prevalence and etiology of subclinical mastitis in intensively managed flocks and related changes in the yield and quality of ewe milk. Small. Ruminant. Res. 43(3), 219–226; doi:10.1016/S0921-4488(02)00022-6 Alfekaiki, D.F. 2018. Characteristics of fat milk Iraqi buffalo (Bubalus bubalis). Res. J. Pharm. Tech. 11(10), 4349–4356; doi: 10.5958/0974-360X.2018.00796.5 Argudín, M.A., Mendoza, M.C. and Rodicio, M.R. 2010. Food poisoning and Staphylococcus aureus enterotoxins. Toxins 2(7), 1751–1773; doi:10.3390/toxins2071751 Bergonier, D., De Crémoux, R., Rupp, R., Lagriffoul, G. and Berthelot, X. 2003. Mastitis of dairy small ruminants. Vet. Res. 34(5), 689–716; doi:10.1051/vetres:2003030 Burriel, A.R. 1998. Isolation of coagulase-negative staphylococci from the milk and environment of sheep. J. Dairy. Res. 65(1), 139–142; doi:10.1017/s0022029997002689 De Camargo, G., Aspilcueta-Borquis, R., Fortes, M., Porto-Neto, R., Cardoso, D., Santos, D., Lehnert, S., Reverter, A., Moore, S. and Tonhati, H. 2015. Prospecting major genes in dairy buffaloes. BMC Genomics 16(1), 872; doi:10.1186/s12864-015-1986-2 Gao, J., Ferreri, M., Liu, X.Q., Chen, L.B., Su, J.L. and Han, B. 2011. Development of multiplex polymerase chain reaction assay for rapid detection of Staphylococcus aureus and selected antibiotic resistance genes in bovine mastitic milk samples. J. Vet. Diagn. Invest. 23(5), 894–901; doi:10.1177/1040638711416964 Gonzalo, C., Ariznabarreta, A., Carriedo, J.A. and San Primitivo, F. 2002. Mammary pathogens and their relationship to somatic cell count and milk yield losses in dairy ewes. J. Dairy Sci. 85(6), 1460–1467. Graber, H.U., Casey, M.G., Naskova, J., Steiner, A. and Schaeren, W. 2007. Development of a highly sensitive and specific assay to detect Staphylococcus aureus in bovine mastitic milk. J. Dairy. Sci. 90(10), 4661–4669; doi:10.3168/jds.2006-902 Hoque, M.N., Talukder, A.K., Saha, O., Hasan, M.M., Sultana, M., Rahman, A.A. and Das, Z.C. 2022. Antibiogram and virulence profiling reveals multidrug resistant Staphylococcus aureus as the predominant aetiology of subclinical mastitis in riverine buffaloes. Vet. Med. Sci. 8(6), 2631–2645; doi:10.1002/vms3.942 Hu, Y., Xie, Y., Tang, J. and Shi, X. 2012. Comparative expression analysis of two thermostable nuclease genes in Staphylococcus aureus. Foodborne Pathog. Dis. 9(3), 265–271; doi:10.1089/fpd.2011.1033 Javid, F., Taku, A., Bhat, M.A., Badroo, G.A., Mudasir, M. and Sofi, T.A. 2018. Molecular typing of Staphylococcus aureus based on coagulase gene. Vet. World. 11(4), 423–430; doi:10.14202/vetworld.2018.423-430 JMP®, Pro 16.1. Cary, NC: SAS Institute Inc, 1989-2021. Johnson, W.M., Tyler, S.D., Ewan, E.P., Ashton, F.E., Pollard, D.R. and Rozee, K.R. 1991. Detection of genes for enterotoxins, exfoliative toxins, and toxic shock syndrome toxin 1 in Staphylococcus aureus by the polymerase chain reaction. J. Clin. Microbial. 29(3), 426–430; doi:10.1128/jcm.29.3.426-430.1991 Krukowski, H., Lassa, H., Zastempowska, E., Smulski, S. and Bis-Wencel, H. 2020. Etiological agents of bovine mastitis in Poland. Med. Weter. 76(4), 221–225; doi:10.21521/mw.6339 Luijendijk, A., Van Belkum, A., Verbrugh, H. and Kluytmans, J. 1996. Comparison of five tests for identification of Staphylococcus aureus from clinical samples. J. Clin. Microbiol. 34(9), 2267–2269; doi:10.1128/jcm.34.9.2267-2269.1996 Młynarczyk, G., Kochman, M., Ławrynowicz, M., Fordymacki, P., Młynarczyk, A. and Jeljaszewicz, J. 1998. Coagulase-negative variants of methicillin-resistant Staphylococcus aureus subsp. aureus strains isolated from hospital specimens. Zentralbl. Bakteriol. 288(3), 373–381; doi:10.1016/s0934-8840(98)80010-8 Momtaz, H., Farzan, R., Rahimi, E., Safarpoor Dehkordi, F. and Souod, N. 2012. Molecular characterization of Shiga Toxin-Producing Escherichia coli isolated from ruminant and donkey raw milk samples and traditional dairy products in Iran. Sci. World J. 2012(1), 231342; doi: 10.1100/2012/231342 Neelam., Jain, V.K., Singh, M., Joshi, V.G., Chhabra, R., Singh, K. and Rana, Y.S. 2022. Virulence and antimicrobial resistance gene profiles of Staphylococcus aureus associated with clinical mastitis in cattle. PLos One 17(5), 264762; doi:10.1371/journal.pone.0264762 Notarnicola, S.M., Zamarchi, G.R. and Onderdonk, A.B. 1985. Misidentification of mucoid variants of Staphylococcus aureus by standard laboratory techniques. J. Clin. Microbial. 22(3), 459–461; doi:10.1128/jcm.22.3.459-461.1985 Onni, T., Sanna, G., Cubeddu, G.P., Marogna, G., Lollai, S., Leori, G. and Tola, S. 2010. Identification of coagulase-negative staphylococci isolated from ovine milk samples by PCR–RFLP of 16S rRNA and gap genes. Vet. Microbial. 144(3-4), 347–352. Papadopoulos, P., Angelidis, A.S., Papadopoulos, T., Kotzamanidis, C., Zdragas, A., Papa, A., Filioussis, G. and Sergelidis, D. 2019. Staphylococcus aureus and methicillin-resistant S. aureus (MRSA) in bulk tank milk, livestock and dairy-farm personnel in north-central and north-eastern Greece: prevalence, characterization and genetic relatedness. Food Microbiol. 84, 103249. Pizauro, L.J., De Almeida, C.C., Soltes, G.A., Slavic, D., De Ávila, F.A., Zafalon, L.F. and MacInnes, J.I. 2019. Detection of antibiotic resistance, mecA, and virulence genes in coagulase-negative Staphylococcus spp. from buffalo milk and the milking environment. J. Dairy Sci. 102(12), 11459–11464; doi:10.3168/jds.2018-15920 Poutrel, B., De Crémoux, R., Ducelliez, M. and Verneau, D. 1997. Control of intramammary infections in goats: impact on somatic cell counts. J. Anim. Sci. 75(2), 566–570; doi:10.2527/1997.752566x Preethirani, P.L., Isloor, S., Sundareshan, S., Nuthanalakshmi, V., Deepthikiran, K., Sinha, A.Y., Rathnamma, D., Nithin Prabhu, K., Sharada, R., Mukkur, T.K. and Hegde, N.R. 2015. Isolation, biochemical and molecular identification, and in-vitro antimicrobial resistance patterns of bacteria isolated from bubaline subclinical mastitis in South India. PLos One 10(11), e0142717. Pyorala, S. and Taponen, S. 2009. Coagulase-negative staphylococci—Emerging mastitis pathogens. Vet. Microbiol. 134(1-2), 3–8; doi:10.1016/j.vetmic.2008.09.015 Qar , T. and Governorates, M. 2007. Water buffalo in the Iraqi Marshes. Nature. Available via http://www.natureiraq.org/uploads/9/2/7/0/9270858/status_report_buffalothiqar.pdf Quinn, P.J., Markey, B.K., Carter, M.E., Donnelly, W.J.C., Leonard, F.C. and Maguire, D. 2002. Veterinary Microbiology and Microbial Diseases. 1st ed. Chichester, UK: Blackwell Science Ltd. Available via https://www.wiley.com/en-us/Veterinary+Microbiology+and+ Microbial+Disease%2C+ 2nd +Edition-p-9781405158237 Quinn, P.J., Markey, B.K., Leonard, F.C., Hartigan, P., Fanning, S. and Fitzpatrick, E. 2011. Veterinary microbiology and microbial disease. Chichester, UK: John Wiley&Sons. Available via https://www.wiley.com/en-us/ Veterinary+Microbiology+and+Microbial+Disease Raad, I., Alrahwan, A. and Rolston, K. 1998. Staphylococcus epidermidis: emerging resistance and need for alternative agents. Rev. Infect. Dis. 26(5), 1182–1187; doi:10.1086/520285 Regasa, S., Mengistu, S. and Abraha, A. 2019. Milk safety assessment, isolation, and antimicrobial susceptibility profile of Staphylococcus aureus in selected dairy farms of Mukaturi and Sululta town, Oromia region, Ethiopia. Vet. Med. Int. 2019(1), 3063185; doi:10.1155/2019/3063185 Sharma, N. and Maiti, S.K. 2010. Prevalence and etiology of sub-clinical mastitis in cows. Indian J. Vet. Res. 19(2). Available via https://www.cabidigitallibrary. org/doi/pdf/10.5555/20113162136 Sharma, N., Rho, G.J., Hong, Y.H., Kang, T.Y., Lee, H.K., Hur, T.Y. and Jeong, D.K. 2012. Bovine mastitis: an Asian perspective. Asian. J. Anim. Vet. Adv. 7(6), 454–476; doi:10.3923/ajava.2012.454.476 Sheet, O.H. 2022. Molecular detection of mecA gene in methicillin-resistant Staphylococcus aureus isolated from dairy mastitis in Nineveh governorate, Iraq. Iraqi J. Vet. Sci. 36(4), 939–943; doi:10.33899/ijvs.2022.132643 Sheet, O.H., Al-Mahmood, O.A., Othamn, S.M., Al-Sanjary, R.A., Alsabawi, A.H. and Abdulhak, A.A. 2023. Detection of positive mecA Staphylococcus aureus isolated from meat and butchers’ shops by using PCR technique in Mosul city. Iraqi. J. Vet. Sci. 37(4), 865–870; doi:10.33899/ijvs.2023.136964.2632 Sheet, O.H., Al-Mahmood, O.A., Taha, A.H., Alsanjary, R.A., Plötz, M. and Abdulmawjood, A.A. 2025. Molecular detection of Methicillin-resistant Staphylococcus aureus isolated from foods in Germany using LAMP assay. Egypt. J. Vet. Sci. 56(6), 1153–1160; doi:10.21608/EJVS.2024.281105.1981 Sheet, O.H., Al-Mahmood, Y.S., Taha, A.H. and Abdulmawjood, A.A. 2024. Detection the spa type of methicillin-resistant Staphylococcus aureus isolated from local Basturma in Mosul city, Iraq. Iraqi. J. Vet. Sci. 38(4), 739–745; doi:10.33899/ijvs.2024.148254.3569 Sheet, O.H., Hussein, S.A. and Al-Chalaby, A.Y. 2021a. Detection of methicillin-resistant Staphylococcus aureus from broiler carcasses in Mosul city. Iraqi. J. Vet. Sci. 35(3), 489–493; doi:10.33899/ijvs.2020.127052 Sheet, O.H., Jwher, D.M., Al-Sanjary, R.A. and Alajami, A.D. 2021b. Direct detection of Staphylococcus aureus in camel milk in the Nineveh Governorate by using the PCR technique. Iraqi J. Vet. Sci. 35(4), 669–672; doi:10.33899/ijvs.2020.127725.1524 Sutra, L. and Poutrel, B. 1994. Virulence factors involved in the pathogenesis of bovine intramammary infections due to Staphylococcus aureus. J. Med. Microbiol. 40(2), 79–89; doi:10.1099/00222615-40-2-79 Taha, A.H., Al-Mahmood, O.A., Sheet, O.H., Hamed, A.A., Al-Sanjary, R.A. and Abdulmawjood, A.A. 2024. Molecular detection of methicillin resistant Staphylococcus aureus isolated from local fish in Mosul city. Iraqi J. Vet. Sci. 38(2), 437–441; doi:10.33899/ijvs.2023.142707 Taha, A.H., Sheet, O.H. and Jwher, D.M. 2025. Molecular detection and phylogenetic diversity of Staphylococcus aureus isolated from goat subclinical mastitis in Nineveh governorate. Iraqi J. Vet. Sci. 39(1), 71–79; doi:10.33899/ijvs.2024.152066.3791 Tenhagen, B.A., Hansen, I., Reinecke, A. and Heuwieser, W. 2009. Prevalence of pathogens in milk samples of dairy cows with clinical mastitis and in heifers at first parturition. J. Dairy Res. 76(2), 179–187; doi:10.1017/S0022029908003786 Tsen, H.Y. and Chen, T.R. 1992. Use of the polymerase chain reaction for specific detection of type A, D and E enterotoxigenic Staphylococcus aureus in foods. Appl. Microbiol. Biotechnol. 37, 685–690. Zadoks, R. and Watts, J. 2009. Species identification of coagulase-negative staphylococci: genotyping is superior to phenotyping. Vet. Microbial. 134(1-2), 20–28. | ||
| How to Cite this Article |
| Pubmed Style Awed MJM, Taha AH, Sheet OH. Prevalence of staphylococcal enterotoxin genes and phylogenetic analysis of coagulase-negative Staphylococcus aureus from buffalo subclinical mastitis milk in Nineveh Governorate, Iraq. Open Vet. J.. 2026; 16(3): 1556-1564. doi:10.5455/OVJ.2026.v16.i3.13 Web Style Awed MJM, Taha AH, Sheet OH. Prevalence of staphylococcal enterotoxin genes and phylogenetic analysis of coagulase-negative Staphylococcus aureus from buffalo subclinical mastitis milk in Nineveh Governorate, Iraq. https://www.openveterinaryjournal.com/?mno=292729 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.13 AMA (American Medical Association) Style Awed MJM, Taha AH, Sheet OH. Prevalence of staphylococcal enterotoxin genes and phylogenetic analysis of coagulase-negative Staphylococcus aureus from buffalo subclinical mastitis milk in Nineveh Governorate, Iraq. Open Vet. J.. 2026; 16(3): 1556-1564. doi:10.5455/OVJ.2026.v16.i3.13 Vancouver/ICMJE Style Awed MJM, Taha AH, Sheet OH. Prevalence of staphylococcal enterotoxin genes and phylogenetic analysis of coagulase-negative Staphylococcus aureus from buffalo subclinical mastitis milk in Nineveh Governorate, Iraq. Open Vet. J.. (2026), [cited March 31, 2026]; 16(3): 1556-1564. doi:10.5455/OVJ.2026.v16.i3.13 Harvard Style Awed, M. J. M., Taha, . A. H. & Sheet, . O. H. (2026) Prevalence of staphylococcal enterotoxin genes and phylogenetic analysis of coagulase-negative Staphylococcus aureus from buffalo subclinical mastitis milk in Nineveh Governorate, Iraq. Open Vet. J., 16 (3), 1556-1564. doi:10.5455/OVJ.2026.v16.i3.13 Turabian Style Awed, Mohammed Jasim Mohammed, Ayman Hani Taha, and Omar Hashim Sheet. 2026. Prevalence of staphylococcal enterotoxin genes and phylogenetic analysis of coagulase-negative Staphylococcus aureus from buffalo subclinical mastitis milk in Nineveh Governorate, Iraq. Open Veterinary Journal, 16 (3), 1556-1564. doi:10.5455/OVJ.2026.v16.i3.13 Chicago Style Awed, Mohammed Jasim Mohammed, Ayman Hani Taha, and Omar Hashim Sheet. "Prevalence of staphylococcal enterotoxin genes and phylogenetic analysis of coagulase-negative Staphylococcus aureus from buffalo subclinical mastitis milk in Nineveh Governorate, Iraq." Open Veterinary Journal 16 (2026), 1556-1564. doi:10.5455/OVJ.2026.v16.i3.13 MLA (The Modern Language Association) Style Awed, Mohammed Jasim Mohammed, Ayman Hani Taha, and Omar Hashim Sheet. "Prevalence of staphylococcal enterotoxin genes and phylogenetic analysis of coagulase-negative Staphylococcus aureus from buffalo subclinical mastitis milk in Nineveh Governorate, Iraq." Open Veterinary Journal 16.3 (2026), 1556-1564. Print. doi:10.5455/OVJ.2026.v16.i3.13 APA (American Psychological Association) Style Awed, M. J. M., Taha, . A. H. & Sheet, . O. H. (2026) Prevalence of staphylococcal enterotoxin genes and phylogenetic analysis of coagulase-negative Staphylococcus aureus from buffalo subclinical mastitis milk in Nineveh Governorate, Iraq. Open Veterinary Journal, 16 (3), 1556-1564. doi:10.5455/OVJ.2026.v16.i3.13 |